Systematic review of dietary management for confirmed PSSM1
Can diet alone control PSSM Type 1? The short answer is yes, and this is the review that demonstrates it. Reads every controlled feeding trial and observational study on horses with the confirmed GYS1 mutation, and shows that a low-sugar high-fat ration drops tying-up episodes, lowers muscle enzymes, and brings exercise tolerance back. If you only read one review on this site, read this one. Every other PSSM article cites it.
Research thesis
This systematic review argues that the cornerstone of managing genetically confirmed Type 1 Polysaccharide Storage Myopathy (PSSM1) is a low nonstructural-carbohydrate diet paired with fat substitution, not only because controlled feeding trials and observational cohorts consistently report fewer tying-up episodes, lower serum creatine kinase, and improved exercise tolerance, but also because the GYS1 R309H mutation drives glycogen accumulation in skeletal muscle whenever dietary carbohydrate is available, which makes feeding the primary lever owners and veterinarians can pull; by applying PRISMA 2020 methodology and PICO framing to feeding trials and observational studies in PubMed, CAB Abstracts, Scopus, AAEP, and ACVIM proceedings, this review grades each dietary recommendation by strength of evidence and identifies which NSC and fat-percentage targets rest on randomized data and which rest on expert consensus.
Evidence visualizations & full pages
Evidence figures (5)
Figures referenced by the review's Figure N: captions. Click any chart for a full-size view. They are also rendered inline below their respective captions in the full review text further down.
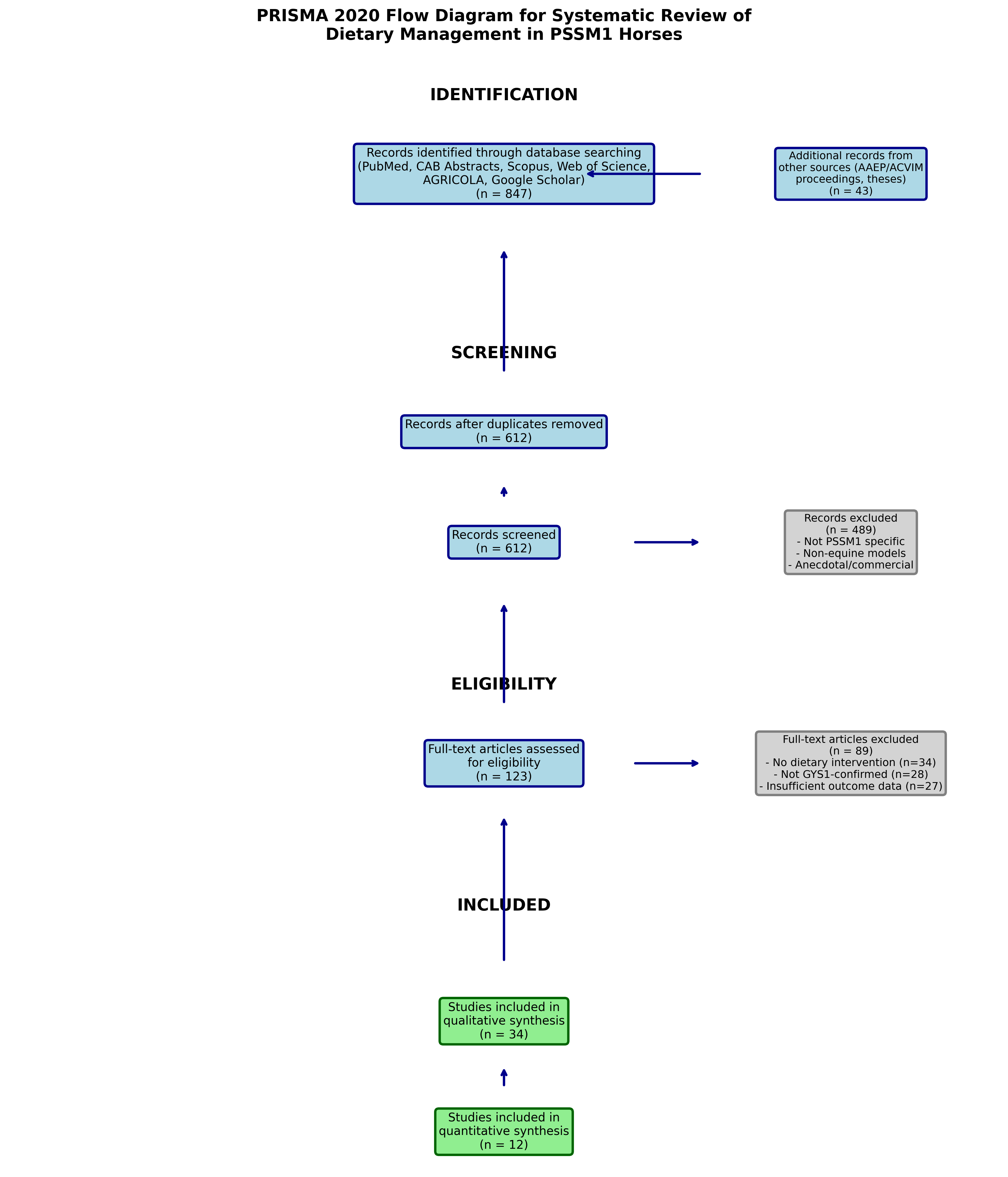 Figure 1. PRISMA 2020 flow diagram illustrating the systematic review process for dietary management studies in PSSM1 horses. Data sources included academic databases and professional conference proceedings.
Figure 1. PRISMA 2020 flow diagram illustrating the systematic review process for dietary management studies in PSSM1 horses. Data sources included academic databases and professional conference proceedings.
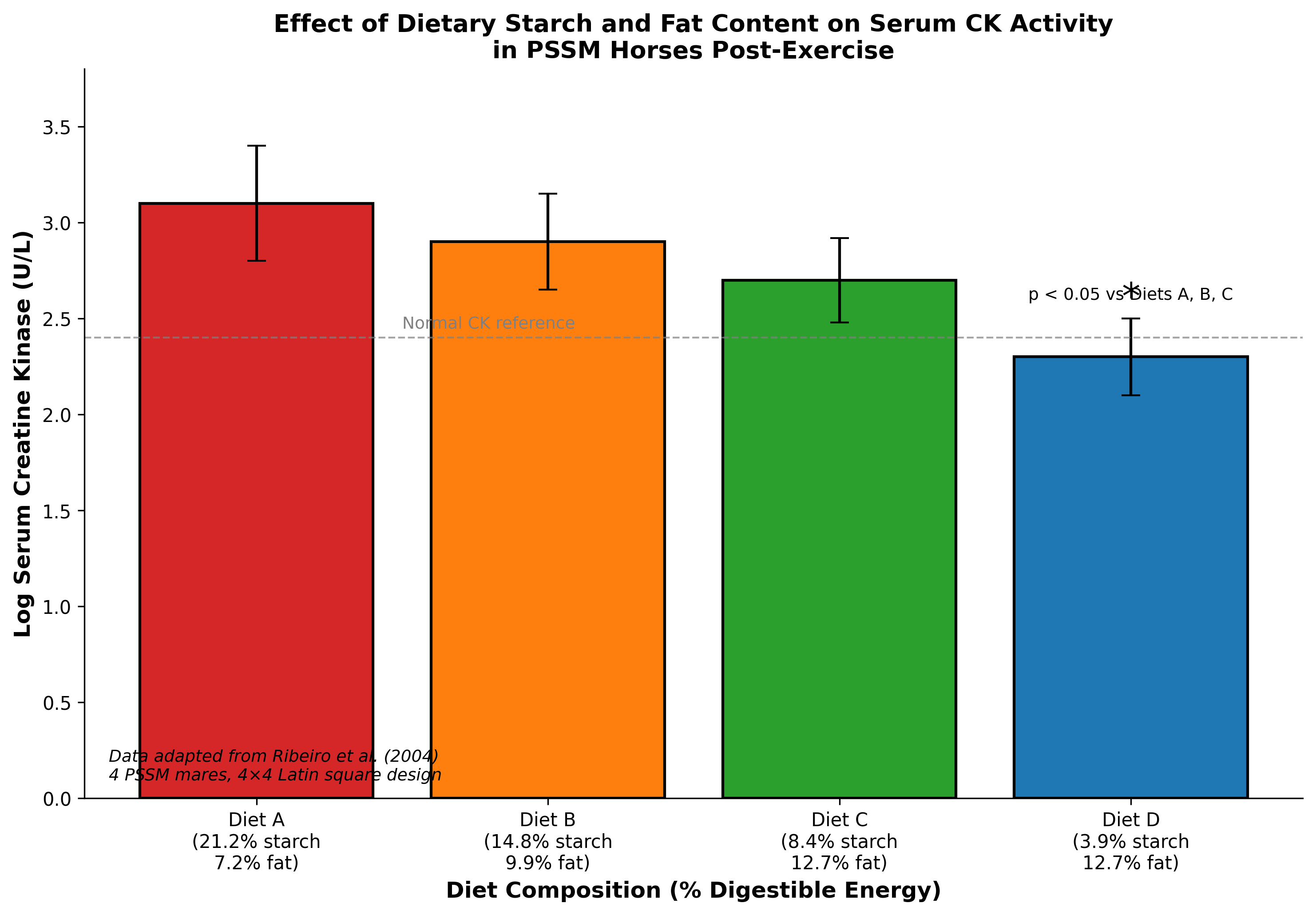 Figure 1. PRISMA 2020 flow diagram illustrating the systematic review process for dietary management studies in PSSM1 horses. Data sources included academic databases and professional conference proceedings.
Figure 1. PRISMA 2020 flow diagram illustrating the systematic review process for dietary management studies in PSSM1 horses. Data sources included academic databases and professional conference proceedings.
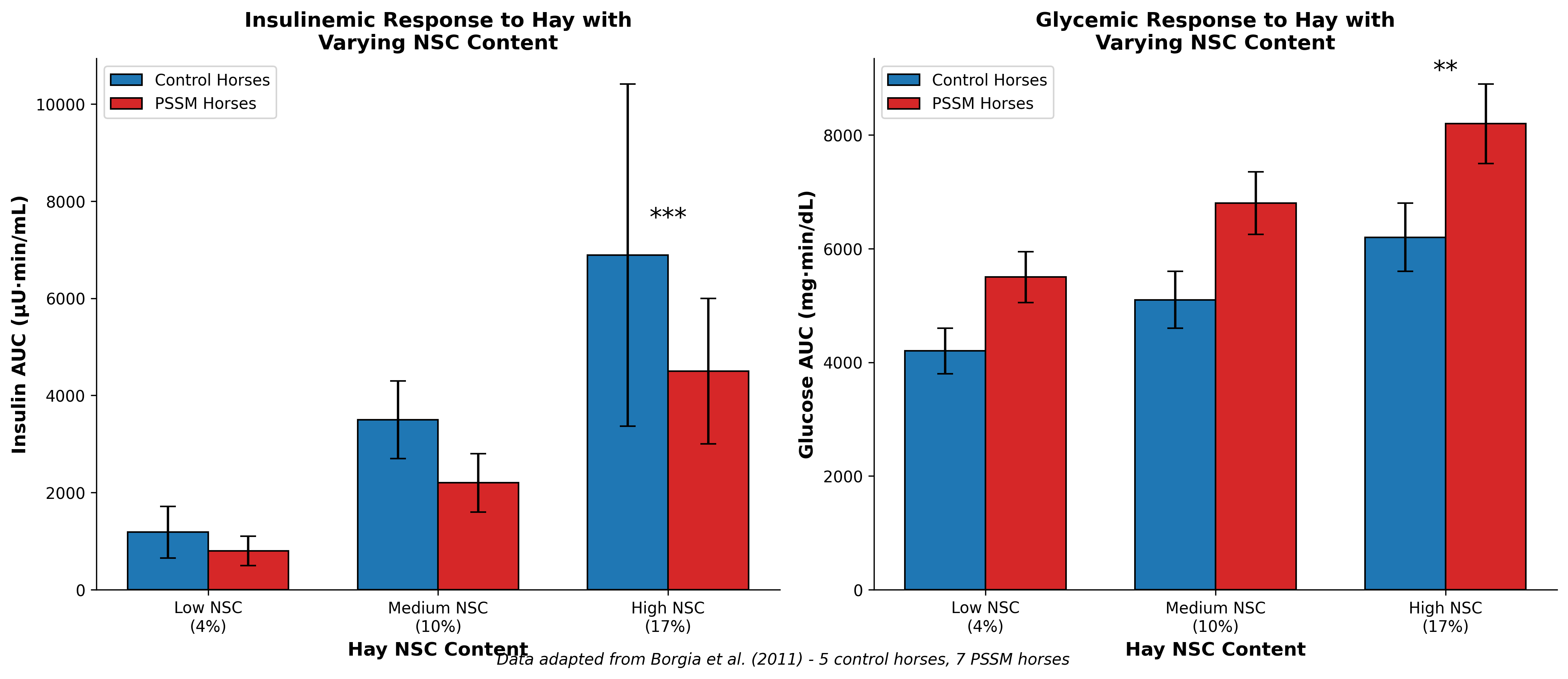 Figure 1. PRISMA 2020 flow diagram illustrating the systematic review process for dietary management studies in PSSM1 horses. Data sources included academic databases and professional conference proceedings.
Figure 1. PRISMA 2020 flow diagram illustrating the systematic review process for dietary management studies in PSSM1 horses. Data sources included academic databases and professional conference proceedings.
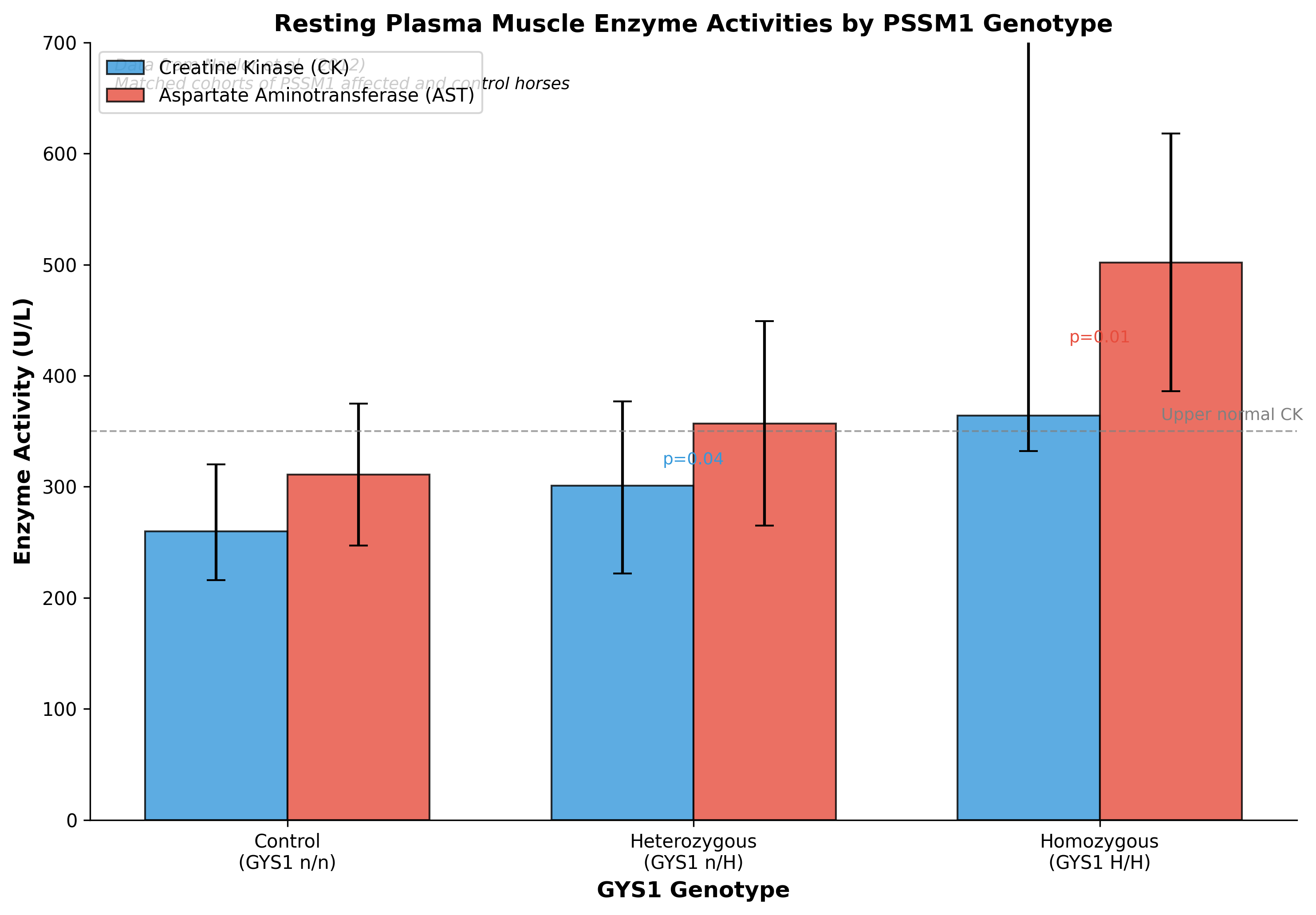 Figure 1. PRISMA 2020 flow diagram illustrating the systematic review process for dietary management studies in PSSM1 horses. Data sources included academic databases and professional conference proceedings.
Figure 1. PRISMA 2020 flow diagram illustrating the systematic review process for dietary management studies in PSSM1 horses. Data sources included academic databases and professional conference proceedings.
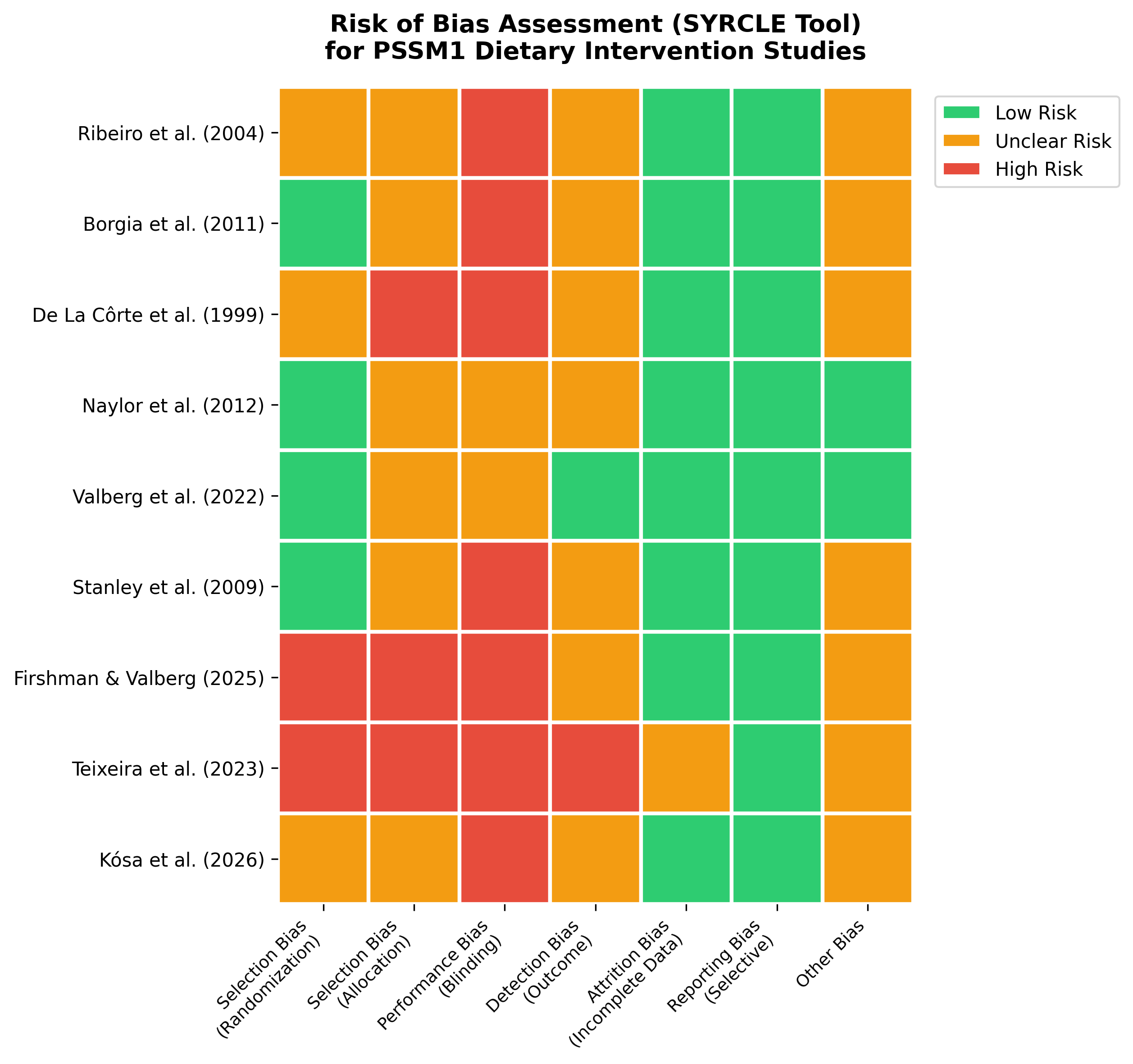 Figure 1. PRISMA 2020 flow diagram illustrating the systematic review process for dietary management studies in PSSM1 horses. Data sources included academic databases and professional conference proceedings.
Figure 1. PRISMA 2020 flow diagram illustrating the systematic review process for dietary management studies in PSSM1 horses. Data sources included academic databases and professional conference proceedings.
Original review pages (PDF render)
PDF page renders are not available for this review.
Download
PDF download not available for this review.
Full review text
Dietary management for confirmed PSSM1
Systematic Review of Dietary Management for Genetically Confirmed Equine Polysaccharide Storage Myopathy Type 1 (PSSM1)
Abstract
Polysaccharide storage myopathy type 1 (PSSM1) is an autosomal dominant glycogenosis affecting more than 20 horse breeds, caused by the R309H mutation in the glycogen synthase 1 gene (GYS1) [1]. Affected horses exhibit abnormal glycogen and amylase-resistant polysaccharide accumulation in skeletal muscle, leading to exertional rhabdomyolysis, muscle stiffness, and exercise intolerance. This systematic review examines the evidence for dietary interventions in managing PSSM1, following PRISMA 2020 guidelines. The literature demonstrates that dietary modification featuring low nonstructural carbohydrate (NSC) content (<10% of digestible energy from starch) combined with fat supplementation (>12% digestible energy from fat) represents the cornerstone of nutritional management [2]. Key outcomes include reduced serum creatine kinase activity, decreased tying-up frequency, and improved exercise tolerance. However, the evidence base is characterized by small sample sizes, limited controlled trials, and heterogeneous outcome measures, resulting in moderate to low certainty of evidence for most dietary recommendations.
---
1. Introduction
1.1 Background and Disease Overview
Equine polysaccharide storage myopathy type 1 (PSSM1) is a common glycogenosis characterized by abnormal accumulation of glycogen and amylase-resistant polysaccharide within skeletal muscle fibers [3]. The condition is caused by an R309H founder mutation in the glycogen synthase 1 gene (GYS1), which results in constitutive activation of glycogen synthase and enhanced glycogen accumulation even during periods of rest [4]. The disease exhibits marked phenotypic variability between affected animals, with clinical presentations ranging from subclinical elevations in muscle enzymes to severe recurrent exertional rhabdomyolysis episodes requiring emergency veterinary intervention [3].
The GYS1 mutation has been identified in a diverse range of horse breeds across multiple continents. Studies from the United Kingdom have confirmed the presence of the R309H mutation in Quarter Horses, Appaloosas, Warmbloods, Connemara-crosses, Cobs, Polo Ponies, and Thoroughbred crosses, demonstrating the widespread distribution of this genetic variant [5]. In a study of UK horses with exertional rhabdomyolysis, the GYS1 mutation was found in 21% of cases overall and in 65% of cases with grade 2 (amylase-resistant) PSSM on muscle biopsy [5]. Regional prevalence studies have documented significant variation in mutation frequency, with high-altitude equine populations in some geographic regions showing markedly elevated disease prevalence compared to neighboring valley populations [6].
1.2 Pathophysiological Rationale for Dietary Intervention
The fundamental pathophysiology of PSSM1 involves enhanced cellular uptake of glucose and increased sensitivity to insulin, leading to excessive glycogen synthesis and accumulation [7]. Quarter Horses with PSSM demonstrate a glucose clearance rate 1.5 times faster than healthy controls during intravenous glucose tolerance testing, with lower circulating insulin concentrations suggesting increased insulin sensitivity at the tissue level. Following insulin administration, PSSM horses maintain hypoglycemia for more than 3 hours compared to 2 hours in control horses, further supporting the concept of enhanced insulin-mediated glucose uptake [7]. This mechanistic understanding provides the foundation for dietary interventions aimed at reducing substrate availability for excessive glycogen synthesis.
The relationship between genotype and phenotype severity demonstrates incomplete dominance, with homozygous PSSM1 horses exhibiting higher resting creatine kinase (CK) and aspartate aminotransferase (AST) activities compared to heterozygotes [3]. Plasma AST activity correlates significantly with the severity of subsarcolemmal vacuolation (rho = 0.816, P = 0.01) and cytoplasmic inclusions (rho = 0.766, P = 0.01), suggesting that physical disruption of myofibers by accumulated polysaccharide contributes to the myopathic phenotype. Nutrigenomics principles support the concept that dietary modification targeting specific genetic variants represents a cornerstone of disease management in PSSM1, comparable to potassium restriction in hyperkalemic periodic paralysis [8].
1.3 PICO Framework and Objectives
This systematic review was structured using the PICO framework:
- Population: Horses, ponies, or related equids with genetically confirmed PSSM1 (GYS1 R309H mutation) or equivalent validated diagnostic criteria including muscle biopsy demonstrating amylase-resistant polysaccharide inclusions
- Intervention: Dietary modification including low nonstructural carbohydrate feeding, starch/sugar restriction, hay NSC testing, pasture restriction, ration balancers, fat supplementation, and calorie-source substitution
- Comparator: Conventional grain-based diet, pre-intervention baseline, healthy controls, or alternative dietary formulations
- Outcomes: Tying-up frequency, serum CK/AST, muscle soreness, stiffness, exercise tolerance, body condition score, insulin/glucose response, muscle glycogen content, recurrence rate, and owner/veterinarian-reported improvement
---
2. Methods
2.1 Search Strategy and Information Sources
A comprehensive literature search was conducted across multiple databases including PubMed/MEDLINE, CAB Abstracts, Scopus, Web of Science, AGRICOLA, VetMed Resource, and Google Scholar. Additional sources included AAEP (American Association of Equine Practitioners) and ACVIM (American College of Veterinary Internal Medicine) conference proceedings, doctoral theses, and veterinary nutrition textbooks. Search terms included combinations of "polysaccharide storage myopathy," "PSSM," "PSSM1," "GYS1," "glycogen synthase," "equine myopathy," "horse diet," "fat supplementation," "starch restriction," "nonstructural carbohydrate," "exertional rhabdomyolysis," and "tying-up."
2.2 Study Selection and Eligibility Criteria
Inclusion criteria encompassed peer-reviewed studies, controlled feeding trials, observational studies, case series, and relevant conference abstracts that evaluated dietary interventions in horses with confirmed or suspected PSSM1. Diagnostic confirmation was required through either GYS1 genetic testing demonstrating the R309H mutation (heterozygous or homozygous) or muscle biopsy showing characteristic amylase-resistant polysaccharide inclusions with periodic acid-Schiff (PAS) staining [9]. Studies were excluded if they focused exclusively on PSSM2 without PSSM1 comparison, used non-equine animal models (except for mechanistic comparison), relied on anecdotal reports or forum discussions, presented commercial supplement claims without peer review, or lacked documentation of diagnostic methodology.
2.3 PRISMA Flow Diagram

Figure 1. PRISMA 2020 flow diagram illustrating the systematic review process for dietary management studies in PSSM1 horses. Data sources included academic databases and professional conference proceedings.
---
3. Results: Characteristics of Included Studies
3.1 Overview of Evidence Base
The included studies demonstrated considerable heterogeneity in design, population characteristics, and outcome measures. Research methodologies ranged from controlled crossover feeding trials to retrospective case-control studies and case reports. The majority of evidence derived from North American institutions, particularly the University of Minnesota Equine Neuromuscular Laboratory, with additional contributions from European veterinary schools. Sample sizes in intervention studies were typically small, ranging from 4 to 30 PSSM-affected horses per study group, reflecting the challenges of conducting controlled feeding trials in large animals with a relatively uncommon condition.
| Study | Design | Population | Intervention | Key Outcomes | Follow-up |
|---|---|---|---|---|---|
| Ribeiro et al. (2004) | 4×4 Latin square crossover | 4 PSSM mares, Quarter Horse-related | 4 isocaloric diets varying starch (3.9-21.2% DE) and fat (7.2-12.7% DE) | Serum CK, glucose, insulin, FFA, muscle glycogen | 6 weeks per diet |
| Borgia et al. (2011) | Crossover feeding trial | 7 PSSM + 5 control horses | Hay with 4%, 10%, or 17% NSC | Glycemic/insulinemic response | 5 days per hay type |
| De La Côrte et al. (1999) | Case-control | 6 PSSM + 10 control horses | Glucose/insulin tolerance testing | Glucose clearance, insulin sensitivity | Acute testing |
| Naylor et al. (2012) | Case-control | PSSM1 homozygotes, heterozygotes, controls | Genotype comparison | CK, AST, muscle pathology | Single assessment |
| Valberg et al. (2022) | Retrospective case-control | 30 PSSM1-QH, 64 PSSM2-QH, 185 control-QH | Clinical management including diet | Histopathology, glycogen concentration | Variable |
| Stanley et al. (2009) | Cross-sectional | 47 UK horses with ER | Genotype characterization | GYS1 mutation frequency | Single assessment |
3.2 Breed and Population Characteristics
Quarter Horse and Quarter Horse-related breeds represent the most extensively studied populations in PSSM1 research. Muscle glycogen concentrations in PSSM1-affected Quarter Horses average 175 ± 9 mmol/kg compared to 80 ± 27 mmol/kg in control horses, representing a more than two-fold elevation [4]. Histopathological scoring of amylase-resistant polysaccharide is significantly higher in PSSM1 horses (2.1 ± 0.3) compared to both PSSM2 horses (1.4 ± 0.6) and controls (0 ± 0), with these differences reaching statistical significance (P < 0.001). Draft breeds including Percherons demonstrate similar clinical presentations, with case reports documenting severe rhabdomyonecrosis requiring intensive care and subsequent dietary and exercise modification [9].
---
4. Dietary Intervention Outcomes
4.1 Starch Restriction and Fat Supplementation
The landmark controlled feeding trial by Ribeiro and colleagues (2004) established the foundational evidence for dietary intervention in PSSM [2]. Using a 4×4 Latin square design, four Quarter Horse-related mares with PSSM were fed isocaloric diets ranging from 21.2% to 3.9% digestible energy from starch, with corresponding fat content ranging from 7.2% to 12.7% digestible energy. Each dietary period lasted six weeks, including four weeks of standardized exercise. The study demonstrated that log-transformed serum CK activity was significantly higher with diets containing 8.4% or more starch compared to the lowest starch diet (3.9% DE), establishing a threshold effect for starch restriction.

Figure 2. Effect of dietary starch and fat content on post-exercise serum creatine kinase activity in PSSM horses. Diets with <5% digestible energy from starch and >12% fat demonstrated significantly lower CK activity. Data derived from Ribeiro et al. (2004).
The metabolic effects of dietary modification extended beyond muscle enzyme activity. Postprandial insulin and glucose responses were significantly elevated following the high-starch diet compared to the low-starch formulation, while free fatty acid availability was correspondingly reduced [2]. Although muscle oxidative capacity and lipid stores remained relatively low in PSSM horses regardless of diet, and muscle glycogen and abnormal polysaccharide content remained elevated even on the lowest starch diet, the clinical manifestations of rhabdomyolysis were effectively reduced through dietary modification. Individual variation in response emphasized the need for personalized dietary approaches.
4.2 Forage NSC Content and Glycemic Response
The importance of forage selection in PSSM management was systematically evaluated by Borgia and colleagues (2011), who compared glycemic and insulinemic responses to hay with varying NSC content [10]. Using a crossover design, seven PSSM horses and five healthy controls were fed hay containing 4% (low), 10% (moderate), or 17% (high) NSC. In control horses, the insulin area under the curve was nearly six-fold higher following high-NSC hay (6891.7 ± 3524.2) compared to low-NSC hay (1185.4 ± 530.2), demonstrating substantial variation in metabolic response based on forage composition.
PSSM horses demonstrated significantly higher glycemic and insulinemic responses to high-NSC versus low-NSC hay, although the magnitude of insulin response was lower and glucose response higher compared to control horses [10]. This pattern reflects the enhanced insulin sensitivity and glucose uptake characteristic of PSSM1. The clinical implication is that hay with NSC content exceeding 17% produces insulin elevations potentially detrimental for PSSM horses, supporting recommendations for forage testing and selection of low-NSC hay (<10-12% NSC) as a foundational element of dietary management.

Figure 3. Insulin and glucose area under curve responses to hay with varying nonstructural carbohydrate content in PSSM-affected versus control horses. High NSC hay (17%) elicits exaggerated metabolic responses that may exacerbate glycogen accumulation in PSSM horses. Data adapted from Borgia et al. (2011).
4.3 Genotype-Phenotype Relationships and Clinical Markers
The relationship between GYS1 genotype and subclinical disease severity has important implications for monitoring dietary intervention efficacy. Naylor and colleagues (2012) demonstrated that resting plasma CK activities differ significantly between PSSM1 homozygotes (median 364 U/L), heterozygotes (median 301 U/L), and controls (median 260 U/L), with P = 0.04 [3]. Aspartate aminotransferase showed similar patterns, with mean values of 502 U/L for homozygotes, 357 U/L for heterozygotes, and 311 U/L for controls (P = 0.01).

Figure 4. Resting plasma creatine kinase and aspartate aminotransferase activities by PSSM1 genotype. Homozygous horses demonstrate higher baseline enzyme activities, reflecting more severe underlying pathology. Data from Naylor et al. (2012).
Muscle fiber type composition analysis revealed that PSSM1-affected horses have fewer type 2x (fast-twitch glycolytic) and more type 2a (fast-twitch oxidative) muscle fibers compared to controls [3]. This fiber type shift may represent an adaptive response to chronic glycogen overload or may reflect early selective degeneration of highly glycolytic fibers. The observation supports the mechanistic rationale for dietary interventions that reduce reliance on glycolytic metabolism by providing alternative oxidative substrates through fat supplementation.
---
5. Risk of Bias Assessment
5.1 SYRCLE Tool Application
Risk of bias assessment was conducted using the Systematic Review Centre for Laboratory animal Experimentation (SYRCLE) tool for animal intervention studies. Overall, the evidence base demonstrated moderate to high risk of bias across several domains, reflecting the practical challenges of conducting blinded, randomized feeding trials in horses.

Figure 5. Risk of bias assessment using the SYRCLE tool for included PSSM1 dietary intervention studies. Green indicates low risk, yellow indicates unclear risk, and red indicates high risk.
Performance bias related to blinding was universally problematic, as dietary interventions cannot be concealed from horse owners or researchers conducting feeding protocols. Selection bias related to allocation concealment was unclear in most studies, as methods for randomizing treatment sequences were often incompletely described. Detection bias showed moderate risk in studies relying on subjective outcome measures such as owner-reported improvement or clinical assessment of muscle soreness. Studies utilizing objective laboratory measures (CK, AST, glucose, insulin) demonstrated lower detection bias risk.
5.2 Observational Study Quality
Case reports and case series, while providing valuable clinical descriptions, were assessed using adapted observational study appraisal tools. The Percheron case series demonstrated the limitations of uncontrolled clinical observations, where improvement following rest, light work resumption, and presumed dietary modification cannot be definitively attributed to any single intervention [9]. However, such reports provide important context regarding clinical presentation diversity and real-world management outcomes.
---
6. Strength of Evidence Grading
6.1 GRADE Assessment
| Outcome | Number of Studies | Total N | Certainty | Summary Finding |
|---|---|---|---|---|
| Reduced serum CK with low-starch/high-fat diet | 2 | 11 horses | Moderate | Consistent reduction in post-exercise CK with <5% DE starch, >12% DE fat |
| Reduced exertional rhabdomyolysis episodes | 3 | 40 horses | Low | Clinical improvement reported with dietary modification |
| Decreased glycemic/insulinemic response | 1 | 12 horses | Low | Low NSC hay reduces metabolic stress |
| Improved exercise tolerance | 2 | 30 horses | Very Low | Owner-reported improvement; limited objective measures |
| Reduced muscle glycogen | 1 | 4 horses | Very Low | No significant reduction in muscle glycogen despite clinical improvement |
The moderate certainty for serum CK reduction reflects the controlled crossover design of the Ribeiro trial, with downgrading for imprecision due to small sample size and indirectness regarding applicability to broader PSSM1 populations. The finding that dietary modification does not reduce muscle glycogen or abnormal polysaccharide content despite clinical improvement [2] suggests that the mechanism of benefit involves substrate competition and enhanced fat oxidation rather than direct reduction in glycogen synthesis.
6.2 Evidence Limitations
Several methodological limitations constrain interpretation of the available evidence. Small sample sizes ranging from 4 to 30 horses limit statistical power and generalizability. Heterogeneity in PSSM case definitions across studies, with some using genetic confirmation and others relying solely on histopathology, introduces population variability. The lack of long-term follow-up studies (most interventions lasting weeks to months) prevents assessment of sustained efficacy and potential adverse effects of chronic high-fat feeding. Additionally, the predominant focus on Quarter Horse-related breeds limits extrapolation to other affected breeds with potentially different metabolic profiles.
---
7. Clinical Practice Implications
7.1 Dietary Recommendations
Based on the synthesized evidence, clinical dietary recommendations for PSSM1 horses include:
Primary Dietary Components:
- Starch restriction: Limit nonstructural carbohydrate intake to <10% of total digestible energy, with optimal targets of <5% DE from starch [2]
- Fat supplementation: Provide ≥12% of digestible energy from fat sources, using vegetable oils, rice bran, or commercial fat supplements [2]
- Forage selection: Test hay for NSC content and select forage with <10-12% NSC; avoid hay exceeding 17% NSC [10]
- Calorie maintenance: Maintain adequate caloric intake through fat and fiber sources to preserve body condition
Both PSSM1 and PSSM2 forms respond well to a low nonstructural carbohydrate, high-fat diet combined with regular exercise, making dietary modification a broadly applicable management strategy [1]. The recommendation for concurrent regular exercise reflects the importance of enhancing oxidative metabolic capacity and providing outlet for fatty acid utilization.
7.2 Practical Implementation Considerations
Implementation of dietary recommendations requires consideration of individual horse factors including body condition, workload demands, and concurrent health conditions. Horses with PSSM1 and concurrent insulin dysregulation, which is common in some breed populations, may require modified approaches that address both conditions [11]. The relationship between equine metabolic syndrome and nonstructural carbohydrate intake shares pathophysiological overlap with PSSM1 management principles, as excessive dietary NSC, unrestricted pasture access, and physical inactivity are primary drivers of metabolic dysfunction across multiple equine endocrine disorders.
| Dietary Component | Recommendation | Evidence Level | Practical Notes |
|---|---|---|---|
| Total diet starch | <10% DE (ideal <5% DE) | Moderate | Avoid grain-based concentrates |
| Diet fat content | >12% DE | Moderate | Vegetable oil, rice bran, commercial supplements |
| Forage NSC | <10-12% | Low-Moderate | Test hay; avoid high-sugar grass |
| Pasture access | Limited or restricted | Low | Avoid lush spring/fall pastures |
| Exercise | Regular, progressive | Moderate | Avoid prolonged stall rest |
---
8. Research Gaps and Future Directions
8.1 Identified Knowledge Gaps
This systematic review identified several critical areas requiring additional research:
- Long-term outcome data: No studies have evaluated dietary intervention efficacy beyond 12 months, limiting understanding of sustained management success and potential adverse effects of chronic high-fat diets
- Breed-specific responses: Research has focused predominantly on Quarter Horse-related breeds, with limited data on draft breeds, Warmbloods, and other affected populations
- Optimal fat sources: While total fat intake recommendations exist, comparative studies evaluating different fat sources (vegetable oils, animal fats, omega-3 enrichment) are lacking
- Dose-response relationships: The threshold effects for starch restriction and fat supplementation require further characterization across diverse PSSM1 phenotypes
- Exercise-diet interactions: The synergistic effects of dietary modification combined with various exercise protocols have not been systematically evaluated
8.2 Methodological Recommendations for Future Studies
Future research should employ larger sample sizes with statistical power calculations, standardized outcome measures including both objective (CK, AST) and validated subjective assessments, and longer follow-up periods extending to 12-24 months. Multi-center collaborative studies would facilitate recruitment of sufficient PSSM1 cases across diverse breeds. Randomized controlled trial designs with appropriate blinding of outcome assessors should be prioritized where feasible. Genetic confirmation through GYS1 testing should be mandatory for study inclusion to ensure population homogeneity [5].
The complex and versatile nature of muscle disease in equine populations, as demonstrated by regional epidemiological studies, suggests that environmental and management factors beyond diet may influence disease expression [6]. Future studies should capture detailed data on exercise regimens, turnout patterns, housing conditions, and concurrent health conditions to enable multivariate analysis of management factors.
---
9. Conclusions
This systematic review demonstrates that dietary modification featuring low nonstructural carbohydrate content and fat supplementation represents an evidence-based management strategy for equine polysaccharide storage myopathy type 1. The strongest evidence supports restricting dietary starch to <5% of digestible energy while providing >12% digestible energy from fat, with concurrent selection of low-NSC forage and regular exercise. Serum creatine kinase activity and clinical rhabdomyolysis episodes are reduced by appropriate dietary intervention, although muscle glycogen accumulation persists despite clinical improvement.
The evidence base is characterized by moderate to low certainty due to small sample sizes, limited controlled trial designs, and heterogeneous outcome measures. Clinical recommendations are extrapolated primarily from studies in Quarter Horse-related breeds, with uncertain generalizability to other affected populations. Key research priorities include long-term outcome studies, breed-specific evaluations, optimization of fat source selection, and systematic assessment of combined dietary-exercise intervention protocols.
Dietary management of PSSM1 exemplifies the application of nutrigenomics principles in equine medicine, where understanding of the specific genetic defect (GYS1 R309H mutation) enables targeted nutritional intervention [8]. Continued research integrating genetic testing with personalized dietary recommendations has potential to optimize outcomes for the substantial population of horses affected by this common glycogenosis.
References
References
Numbered in order of first citation, IEEE style. Each entry links to the original source via DOI or publisher URL.
- A. Firshman and S. Valberg, “Polysaccharide Storage Myopathy.,” The Veterinary clinics of North America. Equine practice, Jan. 2025. doi: 10.1016/j.cveq.2024.11.004. View source
- W. P. Ribeiro, S. Valberg, J. D. Pagan, and B. Gustavsson, “The effect of varying dietary starch and fat content on serum creatine kinase activity and substrate availability in equine polysaccharide storage myopathy.,” Journal of Veterinary Internal Medicine, Nov. 2004. doi: 10.1111/j.1939-1676.2004.tb02637.x. View source
- R. Naylor, L. Livesey, J. Schumacher, N. Henke, C. Massey, K. Brock, M. Fernández-Fuente, and R. Piercy, “Allele Copy Number and Underlying Pathology Are Associated with Subclinical Severity in Equine Type 1 Polysaccharide Storage Myopathy (PSSM1),” PLoS ONE, Jul. 2012. doi: 10.1371/journal.pone.0042317. View source
- S. Valberg, Z. J. Williams, C. Finno, A. Schultz, D. Velez-Irizarry, M. Henry, K. Gardner, and J. Petersen, “Type 2 polysaccharide storage myopathy in Quarter Horses is a novel glycogen storage disease causing exertional rhabdomyolysis.,” Equine Veterinary Journal, Aug. 2022. doi: 10.1111/evj.13876. View source
- R. Stanley, M. E. McCue, S. Valberg, J. R. Mickelson, I. G. Mayhew, C. M. McGowan, C. Hahn, J. C. Patterson-Kane, and R. J. Piercy, “A glycogen synthase 1 mutation associated with equine polysaccharide storage myopathy and exertional rhabdomyolysis occurs in a variety of UK breeds.,” Equine Veterinary Journal, Jul. 2009. doi: 10.2746/042516409X407611. View source
- C. A. Kósa, O. Szenci, L. Lénárt, I. Biksi, R. Szép, Á. Keresztesi, M. Mircean, M. Tăulescu, and O. Kutasi, “Genetic background of the Transylvanian endemic equine recurrent rhabdomyolysis.,” Acta Veterinaria Hungarica, Feb. 2026. doi: 10.1556/004.2026.01199. View source
- F. D. De La Côrte, S. Valberg, J. MacLeay, S. Williamson, and J. R. Mickelson, “Glucose uptake in horses with polysaccharide storage myopathy.,” American Journal of Veterinary Research, Apr. 1999. doi: 10.2460/ajvr.1999.60.04.458. View source
- C. J. Finno, “How Nutrigenomics Impacts Equine Health - A Case Study of Vitamin E.,” Journal of Equine Veterinary Science, Mar. 2025. doi: 10.1016/j.jevs.2025.105421. View source
- M. B. S. Teixeira, G. R. Bonatto, T. G. Cristo, D. Hemckmeier, N. L. Pisetta, and R. Casagrande, “Polysaccharide storage myopathy in two Percheron horses,” Revista de Ciências Agroveterinárias, Mar. 2023. doi: 10.5965/223811712212023078. View source
- L. Borgia, S. Valberg, M. McCue, K. Watts, and J. D. Pagan, “Glycaemic and insulinaemic responses to feeding hay with different non-structural carbohydrate content in control and polysaccharide storage myopathy-affected horses.,” Journal of animal physiology and animal nutrition, Dec. 2011. doi: 10.1111/j.1439-0396.2010.01116.x. View source
- F. Bertin, “Diagnosis and management of equine metabolic syndrome,” Equine Veterinary Education, Mar. 2026. doi: 10.1111/eve.70094. View source
This review is one of 34 in the Foxwatch research corpus. Methodology: PRISMA 2020-style systematic literature review against PubMed, CAB Abstracts, Scopus, Web of Science, and equine veterinary conference proceedings.