Comparative review of nutrition for PSSM versus recurrent exertional rhabdomyolysis
Two horses can both "tie up" and need completely different feeding plans. Recurrent Exertional Rhabdomyolysis is a calcium-handling problem in the muscle fiber, not a glycogen storage problem like PSSM. The high-fat low-sugar ration that controls PSSM is unhelpful for an RER horse, and the small-meal low-grain low-stress protocol that controls RER will leave a PSSM horse undertreated. This review tells the two apart so the diet matches the disease. Read with review 12 (when the diagnosis is unclear).
Research thesis
This comparative systematic review argues that PSSM and Recurrent Exertional Rhabdomyolysis are different diseases that share clinical signs but respond to different feeding protocols, not only because PSSM is a glycogen storage disorder of skeletal muscle while RER is a calcium handling disorder of the muscle fiber, but also because the high-fat low-NSC ration that controls PSSM is unhelpful for an RER horse, and the small-meal, low-stress, low-grain protocol that controls RER leaves a PSSM horse undertreated; by reviewing controlled trials, breed-stratified cohorts, and discipline-specific data across Thoroughbred, Standardbred, Quarter Horse, and Warmblood populations, this review clarifies when a horse with exertional rhabdomyolysis should be managed as PSSM, as RER, or with a hybrid protocol, and identifies the diagnostic markers that meaningfully separate the two.
Evidence visualizations & full pages
Evidence figures (3)
Figures referenced by the review's Figure N: captions. Click any chart for a full-size view. They are also rendered inline below their respective captions in the full review text further down.
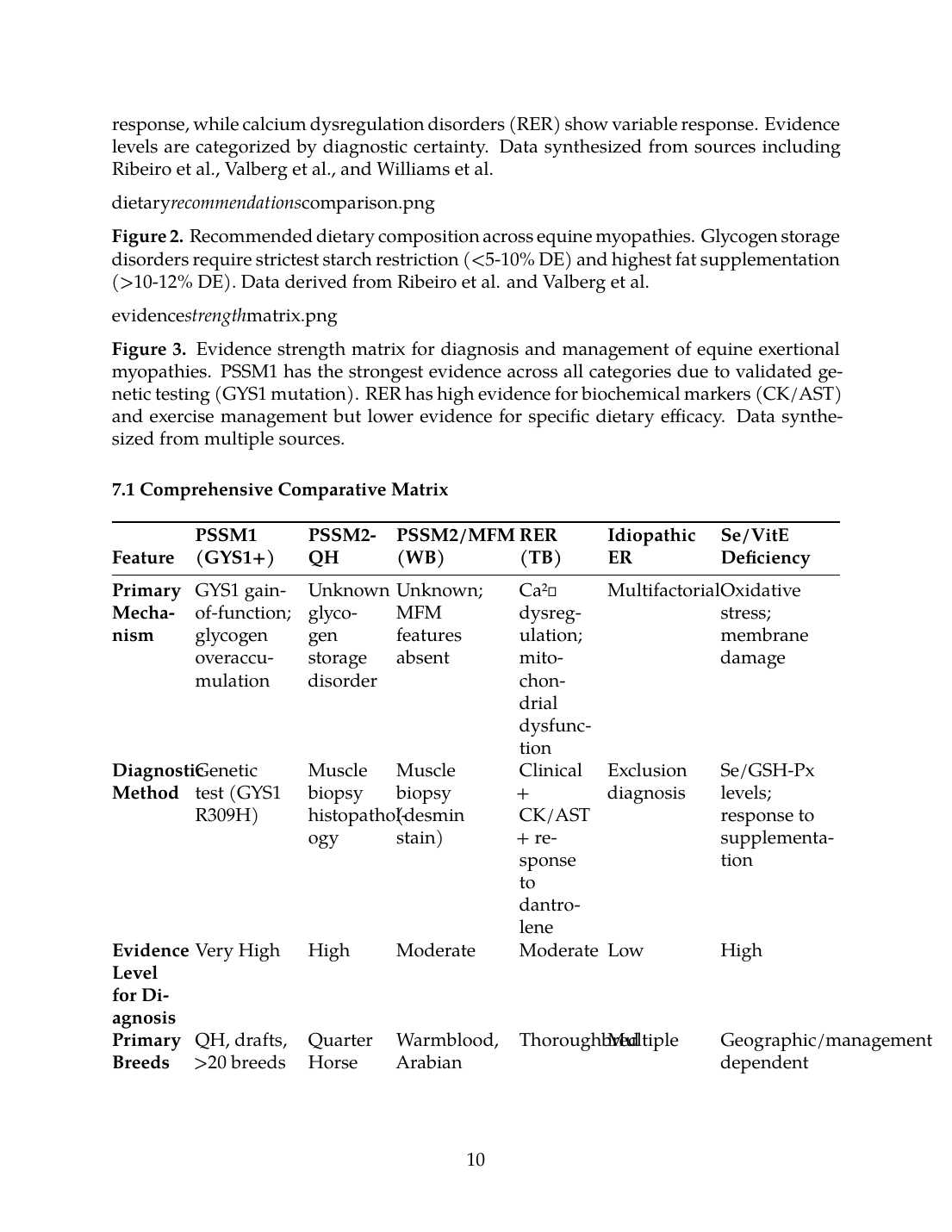
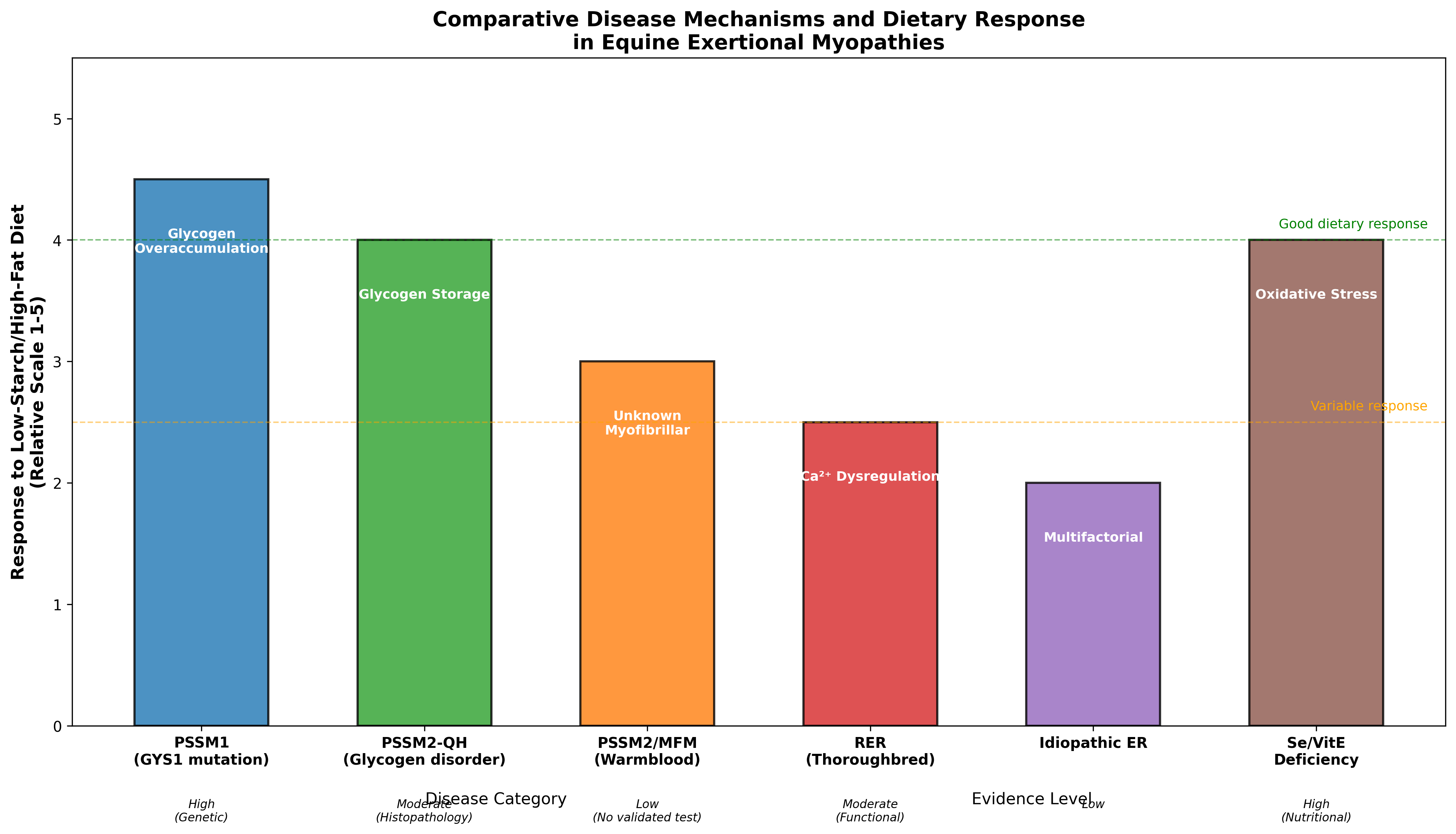 Figure 1. Comparative disease mechanisms and dietary response across equine exertional myopathies. Glycogen storage disorders (PSSM1, PSSM2-QH) show the strongest dietary response, while calcium dysregulation
Figure 1. Comparative disease mechanisms and dietary response across equine exertional myopathies. Glycogen storage disorders (PSSM1, PSSM2-QH) show the strongest dietary response, while calcium dysregulation
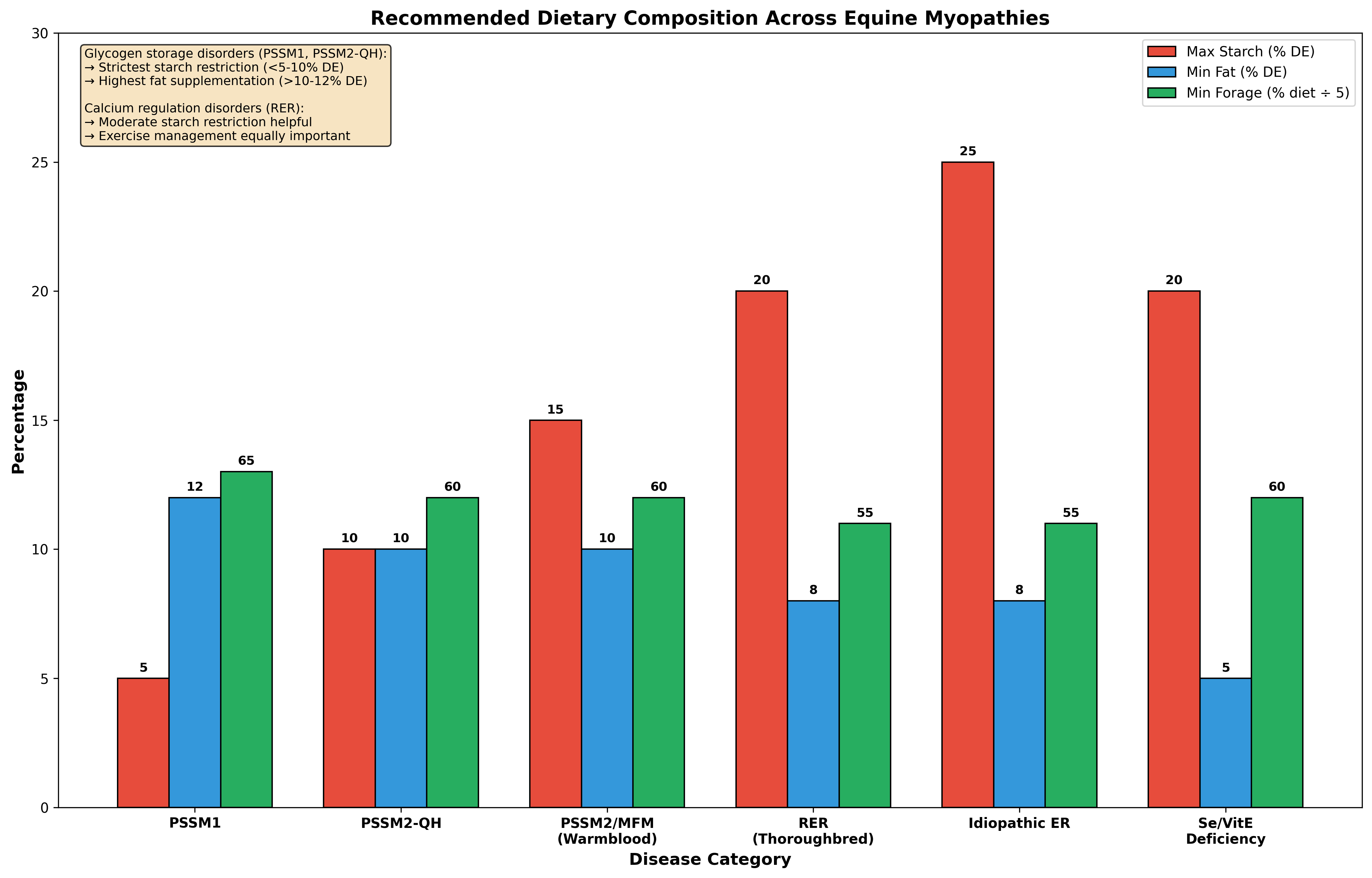 Figure 1. Comparative disease mechanisms and dietary response across equine exertional myopathies. Glycogen storage disorders (PSSM1, PSSM2-QH) show the strongest dietary response, while calcium dysregulation
Figure 1. Comparative disease mechanisms and dietary response across equine exertional myopathies. Glycogen storage disorders (PSSM1, PSSM2-QH) show the strongest dietary response, while calcium dysregulation
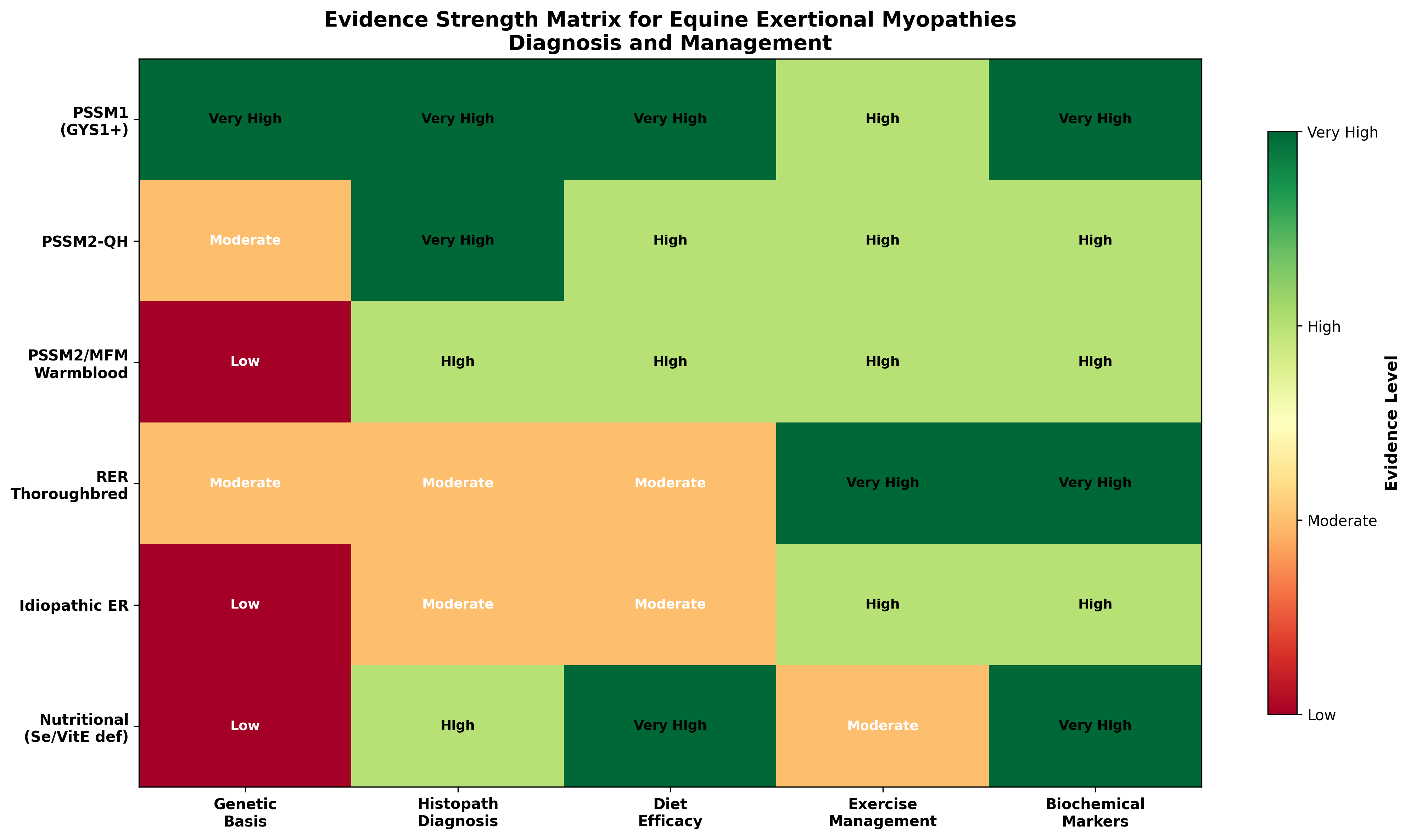 Figure 1. Comparative disease mechanisms and dietary response across equine exertional myopathies. Glycogen storage disorders (PSSM1, PSSM2-QH) show the strongest dietary response, while calcium dysregulation
Figure 1. Comparative disease mechanisms and dietary response across equine exertional myopathies. Glycogen storage disorders (PSSM1, PSSM2-QH) show the strongest dietary response, while calcium dysregulation
Original review pages (PDF render)
Download
Full review text
Nutrition for PSSM versus recurrent exertional rhabdomyolysis
Comparative Systematic Review: Nutritional Strategies for Polysaccharide Storage Myopathy Versus Recurrent Exertional Rhabdomyolysis in Horses
1. Introduction and Scope
1.1 Overview of Equine Exertional Myopathies
Equine exertional myopathies encompass a heterogeneous group of muscle disorders characterized by exercise-induced muscle damage, elevated serum creatine kinase (CK) and aspartate aminotransferase (AST) activities, and clinical signs ranging from muscle stiffness to severe rhabdomyolysis. Genetic selection in horses has concentrated heritable diseases affecting skeletal muscle metabolism and function across breeds [1]. The central challenge for clinicians and nutritionists lies in determining whether dietary recommendations should be disease-specific or represent generalized "tying-up" management strategies applicable across all exertional myopathy phenotypes.
This review addresses this pivotal question by systematically comparing nutritional interventions across distinct diagnostic categories: Type 1 polysaccharide storage myopathy (PSSM1), Type 2 PSSM (including PSSM2-QH in Quarter Horses and PSSM2/MFM in Warmbloods), recurrent exertional rhabdomyolysis (RER) in Thoroughbreds, idiopathic rhabdomyolysis, and exertional myopathies in performance horses. The review examines whether low-starch/high-fat dietary recommendations have different mechanistic targets and evidence strengths across these conditions.
1.2 Classification of Equine Exertional Myopathies
The classification of equine exercise-associated myopathies has evolved substantially. Computational clustering methods have identified phenotypic patterns supporting multiple disease subtypes, with routine muscle histopathology being helpful in sub-categorizing phenotypes [2]. PSSM1 represents an autosomal dominant glycogen storage disorder caused by a founder R309H mutation in the glycogen synthase gene (GYS1), affecting more than 20 breeds [3]. In contrast, PSSM2 in Quarter Horses (PSSM2-QH) is a novel glycogen storage disorder not resulting from mutations in genes currently known to cause muscle glycogenoses in other species [4]. Recurrent exertional rhabdomyolysis in Thoroughbreds appears to involve intramuscular calcium dysregulation and mitochondrial dysfunction, representing a fundamentally different pathophysiology from glycogen storage disorders [5].
1.3 Objectives of This Review
This systematic review aims to: (1) extract and compare diagnostic categories, diet composition, exercise programs, breed predispositions, sex effects, temperament associations, management conditions, serum biomarkers, recurrence rates, and performance outcomes across myopathy types; (2) analyze whether low-starch/high-fat recommendations have different mechanisms or evidence strength across PSSM and non-PSSM rhabdomyolysis; and (3) produce a comparative matrix of disease mechanism, dietary target, evidence level, and management implications.
---
2. Disease Mechanisms and Pathophysiology
2.1 Type 1 Polysaccharide Storage Myopathy (PSSM1)
PSSM1 is characterized by a gain-of-function mutation in the GYS1 gene (R309H substitution) that results in enhanced glycogen synthesis and accumulation of abnormal, amylase-resistant polysaccharide in skeletal muscle fibers [6]. The mutation increases glycogen synthase activity, leading to excessive glycogen storage with characteristic subsarcolemmal aggregates and cytoplasmic inclusions visible on PAS-amylase histochemical staining. In affected Percheron horses, positive homozygous dominant mutations have been documented with associated severe rhabdomyonecrosis, myoglobin nephrosis, and increased susceptibility to anesthetic complications [6].
The pathology demonstrates incomplete dominance, with homozygous horses showing greater subclinical severity than heterozygotes. Subsarcolemmal and cytoplasmic inclusions correlate with clinical severity, suggesting that physical disruption by accumulated material contributes to the myopathic phenotype [7]. Resting and post-exercise CK and AST activities are significantly higher in homozygotes (median resting CK 364 U/L) compared to heterozygotes (301 U/L) and controls (260 U/L), with significant differences between groups [7].
In Haflinger and Noriker horses with the GYS1 mutation, surface electromyography reveals reduced density of muscle activity in genetically affected animals even without overt clinical signs, indicating that the mutation reduces functional muscle fiber recruitment [8]. Gene expression profiling in PSSM muscles reveals mitochondrial dysfunctions, glycogenesis inhibition, and chronic muscle hypoxia as key pathological features [9].
2.2 Type 2 Polysaccharide Storage Myopathy in Quarter Horses (PSSM2-QH)
PSSM2-QH represents a distinct glycogen storage disorder characterized by intermediate histopathological scores and glycogen concentrations compared to PSSM1 and controls. Histopathological analysis shows amylase-resistant polysaccharide scores (PSSM2-QH: 1.4±0.6, PSSM1-QH: 2.1±0.3, control-QH: 0±0) and elevated glycogen concentrations (PSSM2-QH: 129±62, PSSM1-QH: 175±9, control-QH: 80±27 mmol/kg) with significant differences among groups [4]. The abnormal polysaccharide in PSSM2-QH has a less filamentous ultrastructure than PSSM1-QH, and phosphorylase and phosphofructokinase activities are normal, suggesting a different underlying mechanism.
Epidemiological analysis reveals that 17 of 30 PSSM2-QH horses with available pedigrees descended from one of three stallions within four generations, suggesting a hereditary component [4]. However, examination of 29 predicted high or moderate impact genetic variants in 12 candidate genes associated with glycogenoses revealed none unique to PSSM2-QH, indicating the genetic basis remains unknown.
Commercial genetic tests for variants in MYOT (P2), FLNC (P3a, P3b), and MYOZ3 (P4) genes marketed for PSSM2/MFM diagnosis show no significant association with histopathological diagnosis. These variants would improperly diagnose PSSM2/MFM in 57% of healthy Quarter Horses and fail to diagnose PSSM2 in 40% of horses with histopathological evidence [10]. Histopathological features of myofibrillar myopathy are absent in Quarter Horses diagnosed with PSSM2 by biopsy, distinguishing PSSM2-QH from the MFM phenotype seen in Warmbloods [11].
2.3 Type 2 PSSM and Myofibrillar Myopathy in Warmbloods
In Warmblood horses, PSSM2 and myofibrillar myopathy (MFM) represent overlapping diagnoses with unclear genetic etiology. Commercial genetic test variants (P2, P3a/b, P4) in genes associated with human MFM were not significantly associated with histopathological diagnosis in Warmblood and Arabian horses [12]. Importantly, all P variants were present in early domestic horses (400-5500 years old) and P2 was present in Przewalski horses, suggesting these are ancient variants not specifically linked to disease pathogenesis.
RNA sequencing of 16 MFM candidate genes revealed 426 variants, including 26 missense variants, but none were associated with the MFM phenotype. Candidate gene differential expression and muscle fiber specific force did not differ between MFM Warmbloods (143.1±34.7 kPa) and non-MFM Warmbloods (140.2±43.7 kPa) [13]. The lack of validated genetic markers distinguishes PSSM2/MFM in Warmbloods as a syndrome requiring histopathological diagnosis rather than genetic testing.
2.4 Recurrent Exertional Rhabdomyolysis in Thoroughbreds
RER in Thoroughbreds involves fundamentally different molecular mechanisms centered on intracellular calcium dysregulation. Proteomic and transcriptomic analysis of gluteal muscle reveals 125 differentially expressed proteins in RER-susceptible versus control horses, with 52 upregulated proteins primarily involving calcium regulation (N=11), including RYR1, calmodulin, calsequestrin, and calpain [5]. Concurrently, 73 downregulated proteins were largely mitochondrial (N=45), impacting the electron transport system (28 proteins), with decreased mitochondrial heat shock proteins and contractile proteins including calcium-binding proteins.
Importantly, 812 differentially expressed genes were identified in RER-susceptible horses, involving the electron transfer system, mitochondrial transcription/translational response, and the pro-apoptotic calcium-activated mitochondrial membrane transition pore (SLC25A27, BAX, ATP5 subunits) [5]. When RER-susceptible horses were treated with dantrolene, which slows sarcoplasmic reticulum calcium release, they showed no differential gene expression compared to control horses. This finding strongly supports that RER arises from excessive sarcoplasmic reticulum calcium release with subsequent mitochondrial buffering of excessive myoplasmic calcium.
Calcium homeostasis studies using skin-derived myotubes transformed with MyoD have been developed to study RYR1 function and assess responses to dantrolene, providing insights into calcium handling defects underlying RER [14]. This fundamentally different mechanism—calcium dysregulation rather than glycogen storage—has important implications for dietary management approaches.
2.5 Idiopathic and Nutritional Myopathies
Idiopathic exertional rhabdomyolysis encompasses cases without identified genetic or metabolic etiology. In Standardbred horses, all 36 RER-susceptible horses tested were negative for the R309H GYS1 mutation, yet displayed similar risk factors to Thoroughbreds [15]. Female horses were at significantly greater risk (odds ratio 7.1) and nervous horses were at greater risk (odds ratio 7.9) than calm horses.
Nutritional myopathies, particularly selenium and vitamin E deficiency, cause distinct pathology. White muscle disease in foals from selenium-deficient regions presents with muscle weakness, inability to rise, elevated muscle enzymes, and low glutathione peroxidase (GSH-Px) activity [16]. In adult horses, nutritional muscular dystrophy involving masticatory and skeletal muscles can occur with selenium deficiency, demonstrated by marked CK and AST elevations [17].
A severe form of recurrent exertional rhabdomyolysis occurring enzootically in Transylvania demonstrates the role of selenium deficiency, with affected horses showing significantly lower GSH-Px levels (P=0.006) and good correlation between erythrocyte selenium concentration and GSH-Px (r=0.777, P<0.001) [18]. This highlights that some geographically localized rhabdomyolysis may have nutritional rather than genetic etiology.
---
3. Evidence for Low-Starch, High-Fat Dietary Interventions
3.1 Mechanistic Rationale for Dietary Modification in Glycogen Storage Disorders
The fundamental rationale for low-starch, high-fat diets in PSSM derives from the goal of reducing substrate for glycogen synthesis while providing alternative energy sources. In PSSM horses, a diet with <5% digestible energy (DE) from starch and >12% DE from fat significantly reduces serum CK activity compared to higher starch diets [19]. The study using four isocaloric diets (21.2%, 14.8%, 8.4%, and 3.9% DE from starch) demonstrated that log CK activity was higher with diets A, B, and C versus the lowest starch diet D.
Postprandial insulin and glucose responses were higher for high-starch diets, while daily insulin was higher and free fatty acid (FFA) availability lower on high-starch diets [19]. Although muscle glycogen and abnormal polysaccharide content remained high on both extreme diets, the clinical benefit of low starch diets appears to derive from increased FFA availability for muscle metabolism rather than glycogen reduction. Muscle oxidative capacity and lipid stores were low in PSSM horses on all diets, suggesting that fat supplementation may help overcome inherently low capacity for lipid utilization.
Long-chain fats are the best dietetic for PSSM, as demonstrated in a study comparing triheptanoin (odd-chain fatty acid) to long-chain fat supplementation. Triheptanoin had detrimental effects reflecting decreased NEFA availability, increased insulin-stimulated glycogen synthesis, and potential inhibition of lipid oxidation [20].
3.2 Diet and Exercise Response in PSSM2 Warmblood Horses
A retrospective questionnaire study of Warmblood horses with PSSM2 (2008-2016) evaluated responses to low-starch/fat-supplemented diet and exercise regimes [21]. Abnormal traits reported by owners began at approximately 6 years of age and included performance decline, reluctance to collect, and difficulty going forward in over 50% of horses. With diet and exercise modification, 80% of owners reported overall improvement with significant decreases in the proportion showing performance decline and rhabdomyolysis.
However, 53% still did not advance as expected, and performance issues persisted in one-third of horses [21]. Median glycogen concentrations did not differ between horses with and without persistent signs, but horses with the highest glycogen were significantly more likely to show signs than those with lower concentrations. This indicates that low-starch/high-fat diets can improve but do not eliminate the under-saddle characteristics of PSSM2, suggesting different or additional mechanisms beyond glycogen accumulation.
3.3 Dietary Evidence in Recurrent Exertional Rhabdomyolysis
For Thoroughbred RER, the dietary evidence focuses more on caloric balance than specific macronutrient composition. Rations high in grain and formulated to exceed daily energy requirements may increase episodes of rhabdomyolysis in susceptible horses [22]. This suggests that total energy intake relative to expenditure, rather than starch content specifically, drives episodes in RER-susceptible Thoroughbreds.
Survey data from New Zealand trainers managing 1,771 Thoroughbred and Standardbred horses reveals practical challenges [23]. The overall crude prevalence of trainer-identified RER was 8.4%, with 98% of horses fed premixed feeds. Of horses displaying RER signs, only 7% received dietary starch at the recommended level of less than 20% DE, and 50% were fed specialty "tie-up" feeds while 64% still received standard racing feed concurrently. Average starch levels were 33.4±1.1% DE and fat levels 17.5±0.6% DE.
Early case series established that feeding a low-carbohydrate, high-fat diet decreased severity of exercise-induced muscle injury in horses with exertional rhabdomyolysis [24]. All 19 horses had evidence of chronic myopathic changes and abnormal glycogen accumulation; 11 also had complex polysaccharide accumulation. After 3-6 months on low-starch/high-fat diet, 16 horses had no episodes of rhabdomyolysis, with postexercise CK significantly lower than initial values. This study predates the genetic distinction between PSSM and RER, so the mixed population may explain the generally favorable response.
3.4 Dietary Cation-Anion Balance in RER
Research has examined whether manipulating dietary cation-anion balance (DCAB) affects mineral homeostasis in RER-susceptible Thoroughbreds. The DCAB strongly affects plasma and urine pH and fractional excretion of sodium, potassium, chloride, and phosphorus in both clinically normal horses and those with RER [25]. However, this approach has not demonstrated clear clinical benefit in preventing episodes.
---
4. Comparative Analysis of Breed, Sex, and Temperament Effects
4.1 Breed Predispositions
Breed-specific differences in exertional myopathy prevalence and type reflect both genetic factors and selection pressures. The GYS1 mutation causing PSSM1 has been identified in Quarter Horse-related breeds, draft breeds including Percherons and Norikers, and various other breeds [3]. In Noriker draft horse stallions, 38% carried the mutation, with heterozygous horses showing significantly lower scores in driving ability, drafting manner, kindness, and discipline during heavy-load lodging [26].
In Standardbred racehorses, the annual incidence of exertional rhabdomyolysis susceptibility was 6.4% (95% CI 4.6-8.2%) per 100 horses [15]. All tested RER-susceptible Standardbreds were negative for the GYS1 mutation, indicating a distinct condition. Interestingly, RER-susceptible Standardbreds performed better from standstill starts (p=0.04) and had a higher percentage of wins (p=0.006) than controls, suggesting that if the disorder has a genetic basis, improved performance may maintain it in the population.
The Mangalarga Marchador breed in Brazil showed no occurrence of the GYS1 mutation despite myopathy being commonly observed clinically, suggesting different underlying causes [27]. Commercial genetic tests for P2, P3, P4 variants show breed-specific allele frequencies in Warmbloods (controls/cases): 8%/15% (P2), 5%/6% (P3a/b), 9%/13% (P4); in Arabians: 12%/17% (P2), 2%/2% (P3a/b), 7%/12% (P4) [12].
4.2 Sex Effects
Female sex represents a consistent risk factor across multiple forms of exertional rhabdomyolysis. In Thoroughbreds, two-year-old females were most frequently affected, and females showed an odds ratio of 7.1 (95% CI 2.1-23.4) compared to males [28]. Similarly, in Standardbreds, female horses were at significantly greater risk than males (odds ratio 7.1; 95% CI 2.1-23.4; p=0.001) [15].
Investigation of muscle enzyme activity relative to estrous cycle stage in Italian Standardbred mares revealed that during diestrus, RER horses had significantly higher resting and postexercise AST activity compared to controls, with different patterns than during estrus [29]. During estrus, 17β-estradiol and AST activity were significantly negatively correlated in control but not RER mares, suggesting possible hormonal influences on susceptibility.
4.3 Temperament and Management Factors
Temperament represents a modifiable risk factor particularly relevant for RER in Thoroughbreds. Nervous or extremely nervous horses were 5.4 times more likely to develop exertional rhabdomyolysis than calm horses, and horses with some form of lameness were 4.2 times more likely to have episodes [28]. Episodes commonly developed in susceptible horses on a day of rest prior to exercise or when galloped rather than receiving routine exercise.
Case management in an Argentine mare with exertional rhabdomyolysis demonstrated the integrated approach: movement restriction, analgesic/anti-inflammatory treatment, vitamin E/selenium supplementation, and gentle walking exercise with low carbohydrate diet during recovery, with muscle function gradually returning over 4 weeks [30].
---
5. Selenium and Vitamin E in Exertional Myopathies
5.1 Nutritional Myopathies: Distinct from Glycogen Storage Disorders
Selenium and vitamin E deficiency cause nutritional myopathy distinct from glycogen storage disorders but presenting with overlapping clinical signs. White muscle disease in foals occurs in selenium-deficient regions, with affected animals showing muscle weakness, elevated CK and AST, low GSH-Px, and necropsy findings of skeletal muscle necrosis [16]. The Netherlands contains sufficiently selenium-deficient regions to predispose foals to WMD, especially when fed locally harvested roughage.
Selenium deficiency may contribute to acquired recurrent exertional rhabdomyolysis syndromes in specific geographic regions. In Transylvania, an enzootic equine recurrent rhabdomyolysis syndrome affects 17-23% of horses at high altitude settlements versus <2% in neighboring valley villages [18]. Hay from affected areas had lower selenium content, and affected horses had significantly lower GSH-Px levels. This represents a potentially preventable form of recurrent rhabdomyolysis with selenium supplementation rather than starch restriction as the primary intervention.
5.2 Antioxidant Status in Performance Horses
Selenium supplementation augments calcium release from sarcoplasmic reticulum and improves skeletal muscle performance, with effects accompanied by increased selenoprotein N expression, which could improve oxidative stress tolerance during sustained contraction [31]. Selenium has global impacts beyond antioxidant function, influencing mitochondrial capacity and muscular health through selenoproteins H, N, W, and O, which exhibit varying mitochondrial and/or calcium homeostasis influences [32].
Coenzyme Q10 supplementation (1600 mg/d) in Thoroughbred horses showed altered mitochondrial substrate utilization with enhanced respiration via complex II and decreased reliance on complex I, supporting alternative energy pathways during exercise [33]. This demonstrates that antioxidant supplementation can modify muscle energetics independent of starch/fat macronutrient ratios.
5.3 Interaction Between Selenium Status and Myopathy Susceptibility
Dietary selenium imbalances can manifest with diverse clinical signs including tremors and shivering (deficiency) or reproductive dysfunction (toxicity) [34]. Zinc, copper, and selenium deficiencies in pregnant broodmares are subclinical but prevalent, with all studied mares showing zinc concentrations below reference range [35]. Selenium is critical for spermatozoa function and affects motility and oocyte fusion through lipid peroxidation effects [36].
In animal nutrition broadly, selenium deficiency manifests differently across species: in foals, WMD and yellow fat disease; in calves, lambs, and kids, WMD; in pigs, vitamin E/selenium deficiency syndrome [37]. Prevention requires adequate supplementation, with forms including sodium selenite and organic selenium (selenomethionine) showing different tissue distribution and retention patterns [38].
---
6. Management Integration: Exercise, Diet, and Pharmacological Approaches
6.1 Exercise Programs Across Disease Categories
Regular exercise is recommended for PSSM and shows benefit independent of diet in reducing clinical signs. In PSSM2 Warmblood horses, combined diet and exercise management led to 80% owner-reported improvement [21]. However, the relative contribution of each intervention is difficult to separate in retrospective studies.
For RER in Thoroughbreds, management minimizing excitability, particularly when tailored to each horse, may be effective in controlling episodes [28]. Episodes commonly develop during irregular exercise patterns—either after rest days or with sudden increase in intensity. The multifactorial nature of poor performance was confirmed in Standardbred racehorses, with myopathies along with metabolic equivalents, dynamic upper airway obstructions, and gastric ulceration representing main diseases affecting fitness [39].
6.2 Pharmacological Interventions: Dantrolene and Calcium Regulation
Dantrolene, which slows sarcoplasmic reticulum calcium release, is used prophylactically for RER in Thoroughbreds. The finding that RER-susceptible horses treated with dantrolene showed no differential gene expression compared to control horses strongly supports excessive sarcoplasmic reticulum calcium release as the central mechanism [5]. This pharmacological response clearly distinguishes RER from glycogen storage disorders, where dietary intervention rather than calcium channel modulation is the primary approach.
6.3 Integrated Management Recommendations
Dietary management should be disease-specific when diagnosis permits. For documented PSSM1 (GYS1-positive), strict low-starch (<5% DE) and high-fat (>12% DE) diets are evidence-based [19],[3]. For PSSM2-QH diagnosed by histopathology, low starch/fat supplementation combined with regular exercise provides improvement though not complete resolution [4]. For RER in Thoroughbreds, caloric management avoiding excess energy intake, consistent exercise routines, and temperament management appear more important than strict macronutrient ratios, with dantrolene as adjunctive therapy [22],[5].
For nutritional myopathies, selenium and vitamin E supplementation with appropriate attention to geographic risk factors is the primary intervention [18],[16]. Carbohydrate metabolism differences are secondary considerations in these cases.
---
7. Comparative Disease-Diet-Evidence Matrix

Figure 1. Comparative disease mechanisms and dietary response across equine exertional myopathies. Glycogen storage disorders (PSSM1, PSSM2-QH) show the strongest dietary response, while calcium dysregulation disorders (RER) show variable response. Evidence levels are categorized by diagnostic certainty. Data synthesized from sources including Ribeiro et al., Valberg et al., and Williams et al.

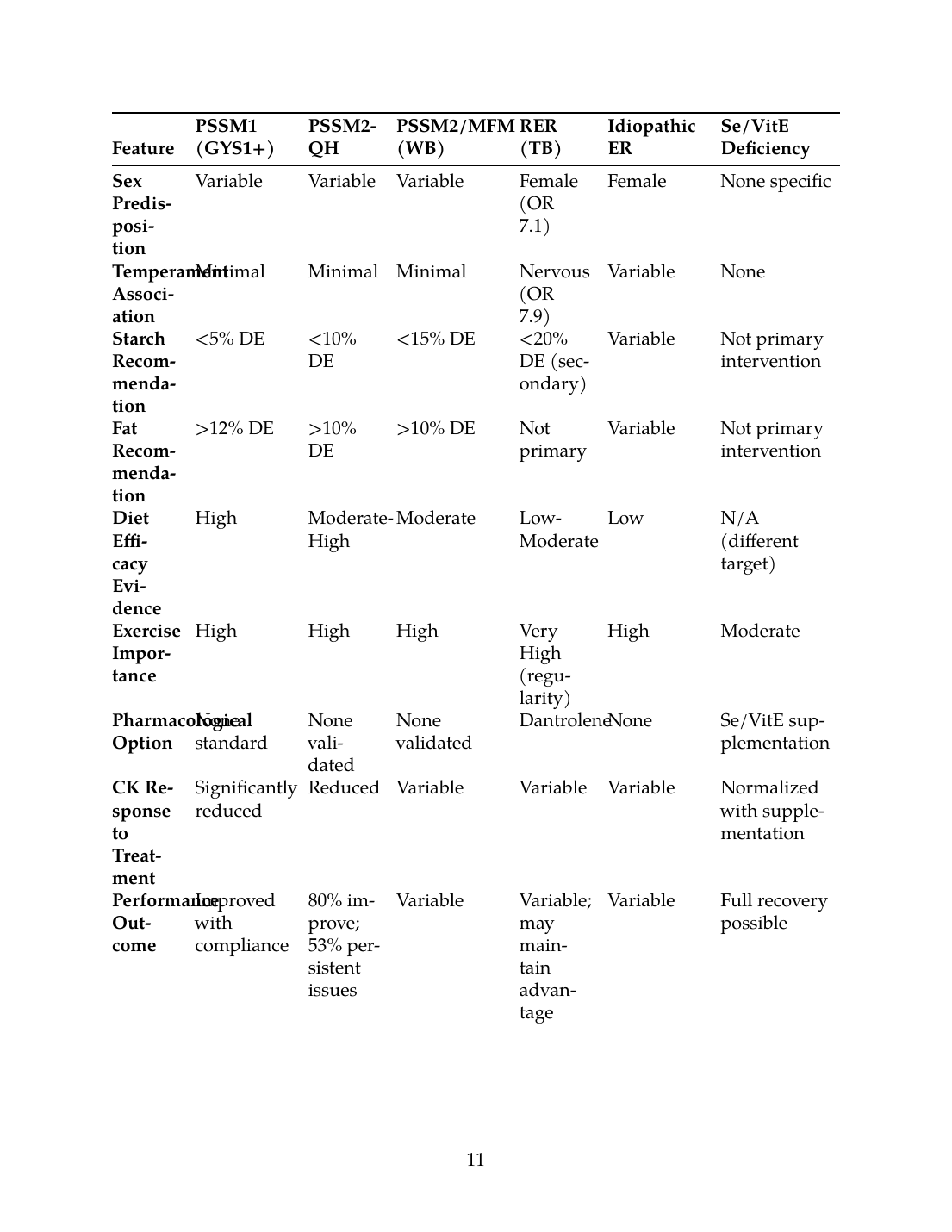
Figure 2. Recommended dietary composition across equine myopathies. Glycogen storage disorders require strictest starch restriction (<5-10% DE) and highest fat supplementation (>10-12% DE). Data derived from Ribeiro et al. and Valberg et al.

Figure 3. Evidence strength matrix for diagnosis and management of equine exertional myopathies. PSSM1 has the strongest evidence across all categories due to validated genetic testing (GYS1 mutation). RER has high evidence for biochemical markers (CK/AST) and exercise management but lower evidence for specific dietary efficacy. Data synthesized from multiple sources.
7.1 Comprehensive Comparative Matrix
| Feature | PSSM1 (GYS1+) | PSSM2-QH | PSSM2/MFM (WB) | RER (TB) | Idiopathic ER | Se/VitE Deficiency |
|---|---|---|---|---|---|---|
| Primary Mechanism | GYS1 gain-of-function; glycogen overaccumulation | Unknown glycogen storage disorder | Unknown; MFM features absent | Ca²⁺ dysregulation; mitochondrial dysfunction | Multifactorial | Oxidative stress; membrane damage |
| Diagnostic Method | Genetic test (GYS1 R309H) | Muscle biopsy histopathology | Muscle biopsy (desmin stain) | Clinical + CK/AST + response to dantrolene | Exclusion diagnosis | Se/GSH-Px levels; response to supplementation |
| Evidence Level for Diagnosis | Very High | High | Moderate | Moderate | Low | High |
| Primary Breeds | QH, drafts, >20 breeds | Quarter Horse | Warmblood, Arabian | Thoroughbred | Multiple | Geographic/management dependent |
| Sex Predisposition | Variable | Variable | Variable | Female (OR 7.1) | Female | None specific |
| Temperament Association | Minimal | Minimal | Minimal | Nervous (OR 7.9) | Variable | None |
| Starch Recommendation | <5% DE | <10% DE | <15% DE | <20% DE (secondary) | Variable | Not primary intervention |
| Fat Recommendation | >12% DE | >10% DE | >10% DE | Not primary | Variable | Not primary intervention |
| Diet Efficacy Evidence | High | Moderate-High | Moderate | Low-Moderate | Low | N/A (different target) |
| Exercise Importance | High | High | High | Very High (regularity) | High | Moderate |
| Pharmacological Option | None standard | None validated | None validated | Dantrolene | None | Se/VitE supplementation |
| CK Response to Treatment | Significantly reduced | Reduced | Variable | Variable | Variable | Normalized with supplementation |
| Performance Outcome | Improved with compliance | 80% improve; 53% persistent issues | Variable | Variable; may maintain advantage | Variable | Full recovery possible |
| Recurrence Risk | Low with dietary compliance | Moderate (1/3 persistent) | Moderate | Moderate-High | High | Low with supplementation |
7.2 Key Distinguishing Features
The matrix reveals critical distinctions:
- Glycogen storage disorders (PSSM1, PSSM2-QH) have the strongest evidence for dietary intervention, with specific starch thresholds (<5% DE for PSSM1) and documented reduction in postexercise CK activity [19].
- Calcium dysregulation disorders (RER) respond to dantrolene treatment with normalization of gene expression profiles [5], suggesting pharmacological management may be more targeted than dietary modification.
- PSSM2/MFM in Warmbloods lacks validated diagnostic testing; commercial genetic tests do not correspond to histopathological diagnosis [12], complicating treatment targeting.
- Nutritional myopathies require selenium/vitamin E supplementation rather than macronutrient modification, representing a fundamentally different intervention [18].
---
8. Conclusions and Implications
8.1 Disease-Specific Versus Generalized Recommendations
This comparative review demonstrates that low-starch/high-fat dietary recommendations are disease-specific for glycogen storage myopathies, with the strongest evidence base for PSSM1, where dietary modification directly addresses the pathophysiological mechanism of enhanced glycogen synthesis driven by insulin stimulation. For PSSM2-QH, similar recommendations provide clinical improvement, though underlying mechanisms differ and complete resolution is uncommon.
For recurrent exertional rhabdomyolysis in Thoroughbreds, dietary recommendations represent generalized tying-up management strategies rather than mechanism-specific interventions. The central pathophysiology involves intracellular calcium dysregulation rather than glycogen accumulation [5], explaining why dietary modification alone shows inconsistent efficacy. Caloric management avoiding excess energy, consistent exercise programming, temperament management, and dantrolene prophylaxis address the actual disease mechanisms more directly than strict macronutrient ratios.
8.2 Implications for Clinical Practice
Clinicians should pursue definitive diagnosis before implementing dietary interventions:
- For PSSM1: Genetic testing (GYS1) provides definitive diagnosis, and dietary modification (<5% DE starch, >12% DE fat) with regular exercise is evidence-based with high response rates [3],[19].
- For PSSM2-QH: Muscle biopsy with histopathological evaluation remains necessary given the lack of validated genetic tests [10]. Low-starch/fat-supplemented diets combined with regular exercise improve 80% of cases but do not eliminate all signs [21].
- For RER in Thoroughbreds: Management should focus on caloric balance, exercise regularity, temperament management, and dantrolene prophylaxis rather than strict starch restriction [28],[5].
- For nutritional myopathies: Geographic risk assessment, selenium/vitamin E status testing, and appropriate supplementation address the underlying deficiency [18],[16].
8.3 Future Research Directions
Critical gaps remain in understanding:
- The genetic basis of PSSM2-QH despite its hereditary pattern [4]
- Why PSSM2/MFM commercial genetic tests show no association with histopathological diagnosis [12]
- The relative contributions of diet versus exercise in clinical improvement
- Optimal integration of pharmacological (dantrolene) and nutritional approaches for RER
- Gene-environment interactions affecting individual responses to dietary intervention
The evidence clearly supports disease-specific rather than blanket dietary recommendations, with glycogen storage disorders warranting strict starch restriction while calcium dysregulation disorders require alternative management strategies. This differentiation is essential for optimizing treatment outcomes across the spectrum of equine exertional myopathies.
References
References
Numbered in order of first citation, IEEE style. Each entry links to the original source via DOI or publisher URL.
- J. R. Mickelson and S. J. Valberg, “The Genetics of Skeletal Muscle Disorders in Horses,” Annual Review of Animal Biosciences, Sep. 2014. doi: 10.1146/annurev-animal-022114-110653. View source
- V. Lindsay‐McGee, C. Massey, Y. T. Li, E. L. Clark, A. Psifidi, and R. J. Piercy, “Characterisation of phenotypic patterns in equine exercise‐associated myopathies,” Equine Veterinary Journal, Jul. 2024. doi: 10.1111/evj.14128. View source
- A. Firshman and S. Valberg, “Polysaccharide Storage Myopathy.,” The Veterinary clinics of North America. Equine practice, Jan. 2025. doi: 10.1016/j.cveq.2024.11.004. View source
- S. Valberg, Z. J. Williams, C. Finno, A. Schultz, D. Velez-Irizarry, M. Henry, K. Gardner, and J. Petersen, “Type 2 polysaccharide storage myopathy in Quarter Horses is a novel glycogen storage disease causing exertional rhabdomyolysis.,” Equine Veterinary Journal, Aug. 2022. doi: 10.1111/evj.13876. View source
- K. Aldrich, D. Velez-Irizarry, C. Fenger, M. Schott, and S. Valberg, “Pathways of calcium regulation, electron transport, and mitochondrial protein translation are molecular signatures of susceptibility to recurrent exertional rhabdomyolysis in Thoroughbred racehorses,” PLoS ONE, Feb. 2021. doi: 10.1371/journal.pone.0244556. View source
- G. Fredo, D. M. Bassuíno, M. V. Bianchi, D. J. Z. Delfiol, A. S. Borges, S. P. Pavarini, L. Sonne, and D. Driemeier, “Pathological findings of post-anesthetic myopathy associated with type 1 polysaccharide storage myopathy in a Percheron horse,” LA Referencia (Red Federada de Repositorios Institucionales de Publicaciones Científicas), Jan. 2018. doi: None. View source
- R. J. Naylor, L. Livesey, J. Schumacher, N. Henke, C. Massey, K. V. Brock, M. Fernández-Fuente, and R. J. Piercy, “Allele Copy Number and Underlying Pathology Are Associated with Subclinical Severity in Equine Type 1 Polysaccharide Storage Myopathy (PSSM1),” PLoS ONE, Jul. 2012. doi: 10.1371/journal.pone.0042317. View source
- R. Zsoldos, N. Khayatzadeh, J. Soelkner, U. Schroeder, C. Hahn, and T. Licka, “Comparison of gluteus medius muscle activity in Haflinger and Noriker horses with polysaccharide storage myopathy,” Journal of animal physiology and animal nutrition, Feb. 2021. doi: 10.1111/jpn.13504. View source
- É. Barrey, E. Mucher, N. Jeansoule, T. Larcher, L. Guigand, B. Herszberg, S. Chaffaux, G. Guérin, X. Mata, P. Benech, M. Canale, O. Alibert, P. Maltere, and X. Gidrol, “Gene expression profiling in equine polysaccharide storage myopathy revealed inflammation, glycogenesis inhibition, hypoxia and mitochondrial dysfunctions,” BMC Veterinary Research, Jan. 2009. doi: 10.1186/1746-6148-5-29. View source
- S. Valberg, M. Henry, K. L. Herrick, D. Velez-Irizarry, C. Finno, and J. Petersen, “Absence of myofibrillar myopathy in Quarter Horses with a histopathological diagnosis of type 2 polysaccharide storage myopathy and lack of association with commercial genetic tests,” Equine Veterinary Journal, Mar. 2022. doi: 10.1111/evj.13574. View source
- S. J. Valberg, M. L. Henry, K. Herrick, D. Velez‐Irizarry, C. J. Finno, and J. L. Petersen, “Absence of myofibrillar myopathy in Quarter Horses with a histopathological diagnosis of type 2 polysaccharide storage myopathy and lack of association with commercial genetic tests,” Equine Veterinary Journal, Mar. 2022. doi: 10.1111/evj.13574. View source
- S. Valberg, C. Finno, M. Henry, M. Schott, D. Velez-Irizarry, S. Peng, E. McKenzie, and J. Petersen, “Commercial genetic testing for type 2 polysaccharide storage myopathy and myofibrillar myopathy does not correspond to a histopathological diagnosis,” Equine Veterinary Journal, Jun. 2020. doi: 10.1111/evj.13345. View source
- Z. J. Williams, D. Velez-Irizarry, J. Petersen, J. Ochala, C. Finno, and S. Valberg, “Candidate gene expression and coding sequence variants in Warmblood horses with myofibrillar myopathy.,” Equine Veterinary Journal, May 2020. doi: 10.1111/evj.13286. View source
- M. Fernández-Fuente, C. M. Terracciano, P. Martín‐Duque, S. Brown, G. Vassaux, and R. J. Piercy, “Calcium Homeostasis in Myogenic Differentiation Factor 1 (MyoD)-Transformed, Virally-Transduced, Skin-Derived Equine Myotubes,” PLoS ONE, Aug. 2014. doi: 10.1371/journal.pone.0105971. View source
- C. Isgren, M. Upjohn, M. Fernández-Fuente, C. Massey, G. Pollott, K. Verheyen, and R. Piercy, “Epidemiology of Exertional Rhabdomyolysis Susceptibility in Standardbred Horses Reveals Associated Risk Factors and Underlying Enhanced Performance,” PLoS ONE, Jul. 2010. doi: 10.1371/journal.pone.0011594. View source
- C. Delesalle, M. de Bruijn, S. Wilmink, H. Vandendriessche, G. Mol, B. Boshuizen, L. Plancke, and G. Grinwis, “White muscle disease in foals: focus on selenium soil content. A case series,” BMC Veterinary Research, May 2017. doi: 10.1186/s12917-017-1040-5. View source
- K. Schefer, R. Hagen, S. K. Ringer, and C. C. Schwarzwald, “Laboratory, Electrocardiographic, and Echocardiographic Detection of Myocardial Damage and Dysfunction in an Arabian Mare with Nutritional Masseter Myodegeneration,” Journal of Veterinary Internal Medicine, Sep. 2011. doi: 10.1111/j.1939-1676.2011.00787.x. View source
- C. Kósa, K. Nagy, O. Szenci, B. Vincze, E. Andrásofszky, R. Szép, Á. Keresztesi, M. Mircean, M. Taulescu, and O. Kutasi, “The role of selenium and vitamin E in a Transylvanian enzootic equine recurrent rhabdomyolysis syndrome,” Acta Veterinaria Hungarica, Sep. 2021. doi: 10.1556/004.2021.00031. View source
- W. P. Ribeiro, S. Valberg, J. D. Pagan, and B. Gustavsson, “The effect of varying dietary starch and fat content on serum creatine kinase activity and substrate availability in equine polysaccharide storage myopathy.,” Journal of Veterinary Internal Medicine, Nov. 2004. doi: 10.1111/j.1939-1676.2004.tb02637.x. View source
- L. A. Borgia, S. J. Valberg, M. E. McCue, J. D. Pagan, and C. R. Roe, “Effect of dietary fats with odd or even numbers of carbon atoms on metabolic response and muscle damage with exercise in Quarter Horse–type horses with type 1 polysaccharide storage myopathy,” American Journal of Veterinary Research, Mar. 2010. doi: 10.2460/ajvr.71.3.326. View source
- Z. J. Williams, M. Bertels, and S. J. Valberg, “Muscle glycogen concentrations and response to diet and exercise regimes in Warmblood horses with type 2 Polysaccharide Storage Myopathy,” PLoS ONE, Sep. 2018. doi: 10.1371/journal.pone.0203467. View source
- J. M. MacLeay, S. J. Valberg, J. D. Pagan, J. Xue, F. D. D. L. Côrte, and J. M. Roberts, “Effect of ration and exercise on plasma creatine kinase activity and lactate concentration in Thoroughbred horses with recurrent exertional rhabdomyolysis,” American Journal of Veterinary Research, Nov. 2000. doi: 10.2460/ajvr.2000.61.1390. View source
- L. Wood, B. Lancaster, and C. Rogers, “The feeding and management of Thoroughbred and Standardbred Racehorses displaying clinical signs of Recurrent Exertional Rhabdomyolysis,” Edinburgh Research Explorer (University of Edinburgh), Oct. 2019. doi: None. View source
- B. A. Valentine, H. F. Hintz, K. M. Freels, A. J. Reynolds, and K. Thompson, “Dietary control of exertional rhabdomyolysis in horses,” Journal of the American Veterinary Medical Association, May 1998. doi: 10.2460/javma.1998.212.10.1588. View source
- E. C. McKenzie, S. J. Valberg, S. Godden, J. D. Pagan, G. P. Carlson, J. M. MacLeay, and F. D. DeLaCorte, “Plasma and urine electrolyte and mineral concentrations in Thoroughbred horses with recurrent exertional rhabdomyolysis after consumption of diets varying in cation-anion balance,” American Journal of Veterinary Research, Jul. 2002. doi: 10.2460/ajvr.2002.63.1053. View source
- T. Druml, G. Grilz-Seger, and Г. Брем, “Effect of the glycogen synthase 1 (&lt;i&gt;GYS1)&lt;/i&gt; mutation on performance traits in 169 Noriker draft horse stallions – a retrospective study,” Archives animal breeding/Archiv für Tierzucht, Nov. 2016. doi: 10.5194/aab-59-453-2016. View source
- N. V. D. Silva, A. L. H. Albuquerque, J. P. O. Oliveira Filho, A. S. Borges, and D. Delfiol, “Study of the mutation causing type 1 polysaccharide storage myopathy in a Mangalarga Marchador population used in breeding programs,” Acta Veterinaria Brasilica, Mar. 2023. doi: 10.21708/avb.2023.17.1.11422. View source
- J. M. MacLeay, S. A. Sorum, S. J. Valberg, W. Marsh, and M. D. Sorum, “Epidemiologic analysis of factors influencing exertional rhabdomyolysis in Thoroughbreds,” American Journal of Veterinary Research, Dec. 1999. doi: 10.2460/ajvr.1999.60.12.1562. View source
- M. Pasolini, R. Pezzella, P. Santoro, N. Cocchia, M. Greco, C. D. Prete, G. D. Valle, and L. Auletta, “Correlation Between Serum Activity of Muscle Enzymes and Stage of the Estrous Cycle in Italian Standardbred Horses Susceptible to Exertional Rhabdomyolysis.,” Journal of Equine Veterinary Science, Sep. 2020. doi: 10.1016/j.jevs.2020.103175. View source
- M. U. Ememe and R. E. Edeh, “A Case of Exertional Rhabdomyolysis in an 11-year-old Argentine Mare,” Sahel Journal of Veterinary Sciences, Dec. 2022. doi: 10.54058/saheljvs.v19i4.313. View source
- D. Bodnár, O. Ruzsnavszky, T. Oláh, B. Dienes, I. Balatoni, É. Ungvári, I. Benkő, B. Babka, J. Prokisch, L. Csernoch, and P. Szentesi, “Dietary selenium augments sarcoplasmic calcium release and mechanical performance in mice,” Nutrition & Metabolism, Nov. 2016. doi: 10.1186/s12986-016-0134-6. View source
- L. T. Wesolowski, P. L. Semanchik, and S. H. White-Springer, “Beyond antioxidants: Selenium and skeletal muscle mitochondria,” Frontiers in Veterinary Science, Dec. 2022. doi: 10.3389/fvets.2022.1011159. View source
- M. L. Henry, L. T. Wesolowski, J. D. Pagan, J. L. Simons, S. J. Valberg, and S. H. White-Springer, “Impact of Coenzyme Q10 Supplementation on Skeletal Muscle Respiration, Antioxidants, and the Muscle Proteome in Thoroughbred Horses,” Antioxidants, Jan. 2023. doi: 10.3390/antiox12020263. View source
- T. Danese and E. Valle, “Dietary intervention in selenium imbalances: a case series,” UK-Vet Equine, Mar. 2026. doi: 10.12968/ukve.2025.0006. View source
- W. Kędzierski, A. Chałabis‐Mazurek, Z. Bełkot, I. Janczarek, and S. Kowalik, “Zinc, copper and selenium deficiencies in broodmares in south-eastern Poland,” Polish Journal of Veterinary Sciences, Sep. 2025. doi: 10.24425/pjvs.2025.156074. View source
- E. Wiedosari and Y. Sani, “The Role of Selenium in Controlling Reproductive Disorder in Beef Cattle,” Indonesian Bulletin of Animal and Veterinary Sciences, Mar. 2022. doi: 10.14334/wartazoa.v32i1.2883. View source
- B. Hosnedlova, M. Kepinska, S. Skalickova, C. Fernandez, B. Ruttkay-Nedecky, T. Malevu, J. Sochor, M. Baroň, M. Melčová, J. Žídková, and R. Kizek, “A Summary of New Findings on the Biological Effects of Selenium in Selected Animal Species—A Critical Review,” International Journal of Molecular Sciences, Oct. 2017. doi: 10.3390/ijms18102209. View source
- E. Reinoso-Maset, M. Falk, A. Bernhoft, C. Ersdal, T. Framstad, H. Fuhrmann, B. Salbu, and M. Oropeza-Moe, “Selenium Speciation Analysis Reveals Improved Antioxidant Status in Finisher Pigs Fed l-Selenomethionine, Alone or Combined with Sodium Selenite, and Vitamin E,” Biological Trace Element Research, Dec. 2022. doi: 10.1007/s12011-022-03516-9. View source
- C. M. L. Feudo, L. Stucchi, B. Conturba, G. Stancari, E. Zucca, and F. Ferrucci, “Medical causes of poor performance and their associations with fitness in Standardbred racehorses,” Journal of Veterinary Internal Medicine, May 2023. doi: 10.1111/jvim.16734. View source
This review is one of 34 in the Foxwatch research corpus. Methodology: PRISMA 2020-style systematic literature review against PubMed, CAB Abstracts, Scopus, Web of Science, and equine veterinary conference proceedings.