Integrated review of horses with both suspected PSSM and EPM
Some horses have both diseases. The high-fat low-sugar ration that controls PSSM raises the gastric ulcer and antioxidant-demand concerns most relevant to EPM recovery. Antiprotozoal treatment changes muscle protein turnover in ways the single-disease guidance does not anticipate. This review reconciles the two protocols so you do not solve one disease and worsen the other. Pairs with reviews 11 (EPM nutrition) and 22 (EPM rehab).
Research thesis
This systematic review argues that horses with concurrent PSSM and EPM are a clinically distinct population whose feeding priorities partly conflict and require explicit reconciliation, not two protocols running side by side, not only because the high-fat low-NSC ration optimal for PSSM raises gastric ulcer and antioxidant-demand concerns most relevant to EPM recovery, but also because antiprotozoal treatment and prolonged stall rest interact with insulin response and muscle protein turnover in ways single-disease protocols do not anticipate; by reviewing case series, retrospective cohorts, and the supportive-care literature across co-occurring populations, this review constructs an integrated feeding protocol that resolves the conflict and identifies the points where veterinary judgment is required.
Evidence visualizations & full pages
Evidence figures (4)
Figures referenced by the review's Figure N: captions. Click any chart for a full-size view. They are also rendered inline below their respective captions in the full review text further down.
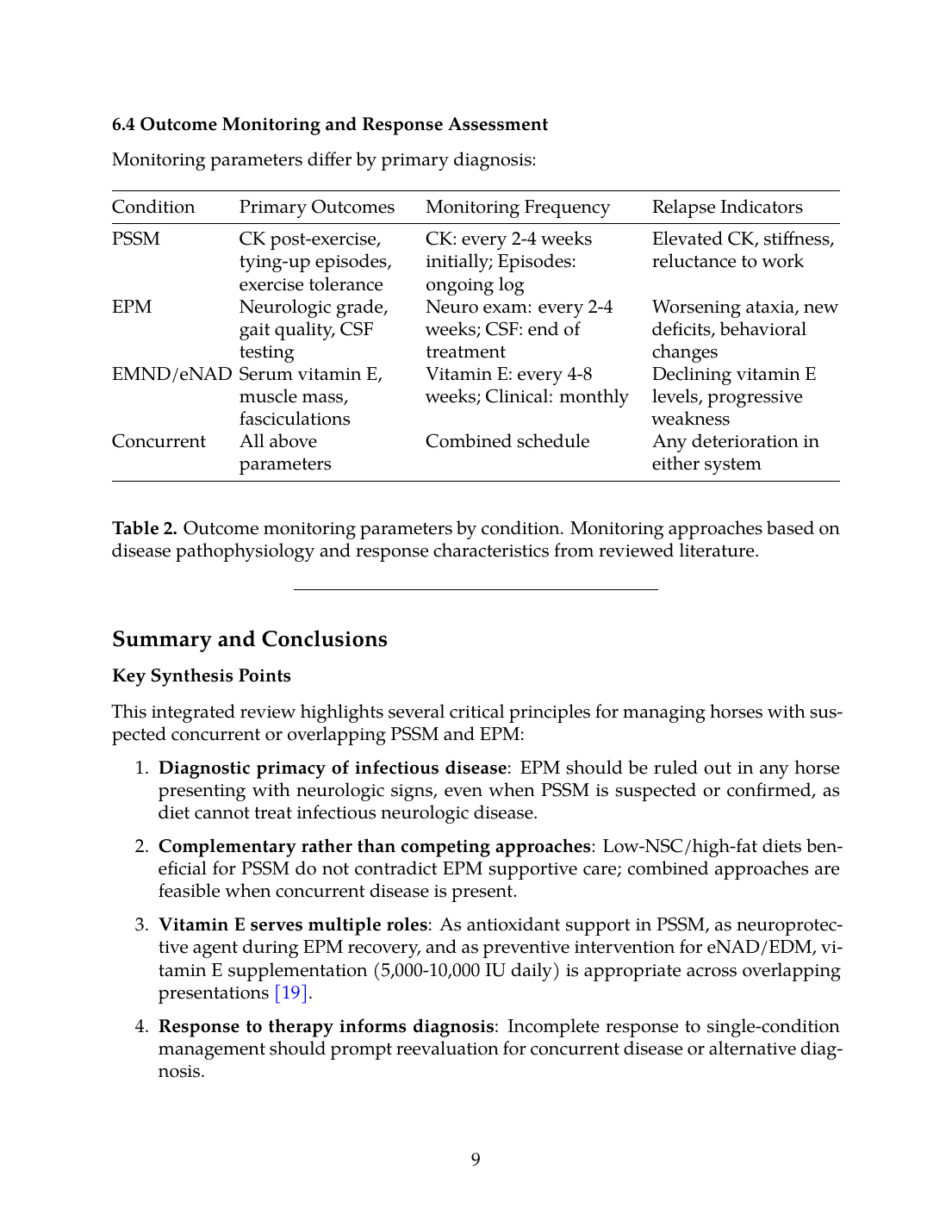
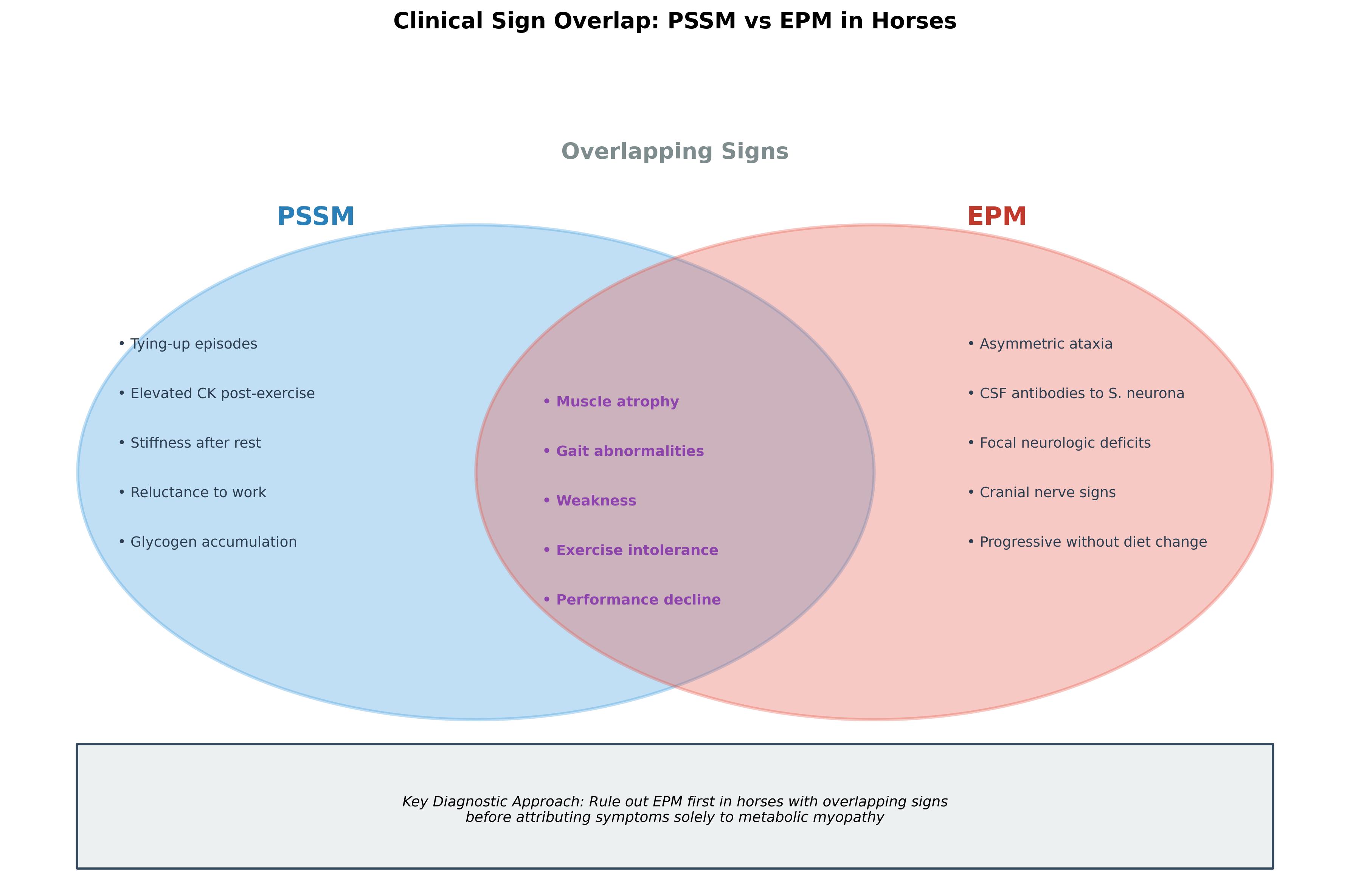 Figure 1. Clinical sign overlap between PSSM and EPM in horses, illustrating the diagnostic challenge posed by shared presentations and the importance of ruling out infectious neurologic disease before attribu
Figure 1. Clinical sign overlap between PSSM and EPM in horses, illustrating the diagnostic challenge posed by shared presentations and the importance of ruling out infectious neurologic disease before attribu
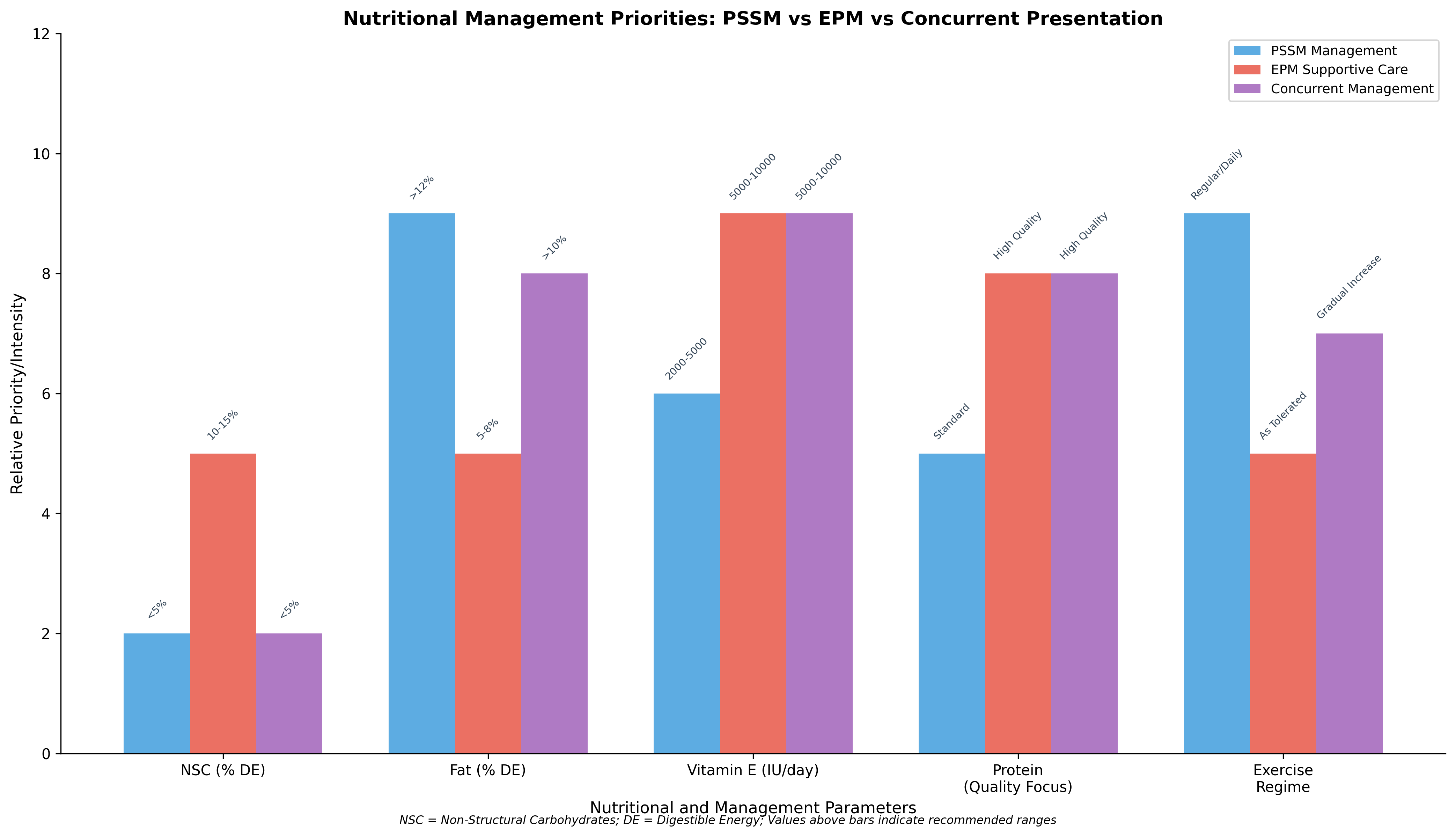 Figure 1. Clinical sign overlap between PSSM and EPM in horses, illustrating the diagnostic challenge posed by shared presentations and the importance of ruling out infectious neurologic disease before attribu
Figure 1. Clinical sign overlap between PSSM and EPM in horses, illustrating the diagnostic challenge posed by shared presentations and the importance of ruling out infectious neurologic disease before attribu
 Figure 1. Clinical sign overlap between PSSM and EPM in horses, illustrating the diagnostic challenge posed by shared presentations and the importance of ruling out infectious neurologic disease before attribu
Figure 1. Clinical sign overlap between PSSM and EPM in horses, illustrating the diagnostic challenge posed by shared presentations and the importance of ruling out infectious neurologic disease before attribu
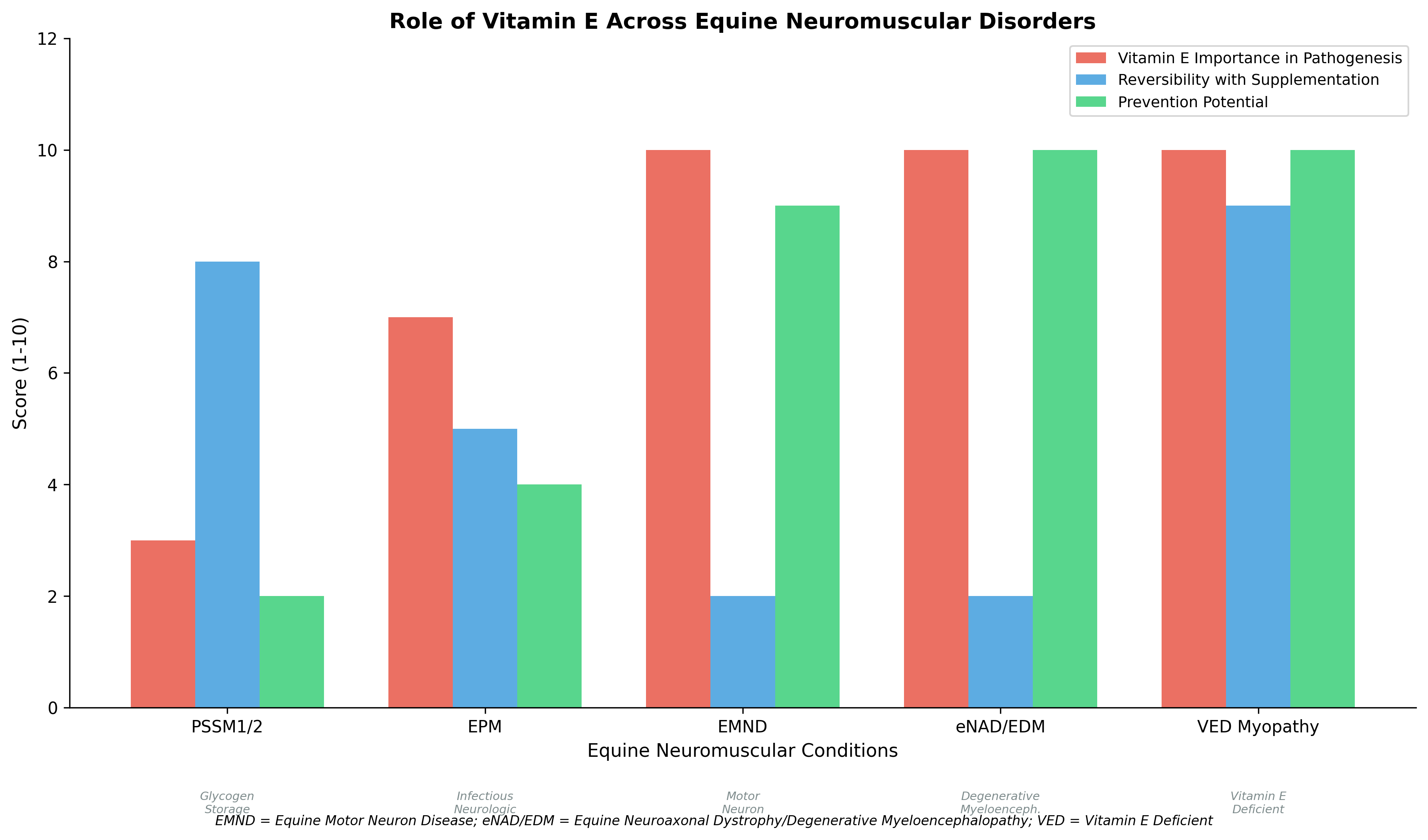 Figure 1. Clinical sign overlap between PSSM and EPM in horses, illustrating the diagnostic challenge posed by shared presentations and the importance of ruling out infectious neurologic disease before attribu
Figure 1. Clinical sign overlap between PSSM and EPM in horses, illustrating the diagnostic challenge posed by shared presentations and the importance of ruling out infectious neurologic disease before attribu
Original review pages (PDF render)
Download
Full review text
Horses with both suspected PSSM and EPM
Integrated Review of Horses with Both Suspected PSSM and EPM: A Clinical-Decision Synthesis
1. Introduction and Background
1.1 Clinical Significance of Overlapping Presentations
Horses presenting with concurrent muscle dysfunction and neurologic signs pose significant diagnostic challenges for equine practitioners. Polysaccharide storage myopathy (PSSM) and equine protozoal myeloencephalitis (EPM) represent two distinct pathophysiological processes—one metabolic/genetic and one infectious—that share several clinical features including muscle atrophy, gait abnormalities, exercise intolerance, and performance decline. Type 1 PSSM (PSSM1) is an autosomal dominant glycogen storage disorder affecting more than 20 breeds of horses, caused by the R309H mutation in the GYS1 gene and characterized by abnormal amylase-resistant polysaccharide accumulation in skeletal muscle [1]. EPM, caused primarily by Sarcocystis neurona, is a debilitating neurologic disease affecting horses across the Americas, with ongoing challenges in diagnosis and treatment that negatively impact prognosis [2]. The potential for misdiagnosis is substantial when clinicians encounter horses with overlapping symptomatology, particularly as both conditions can manifest with generalized weakness, reluctance to work, and progressive decline.
1.2 Rationale for Integrated Review
Direct evidence of PSSM-EPM comorbidity in the veterinary literature remains limited, necessitating a synthesis approach that draws from parallel evidence streams. This review integrates findings from PSSM nutritional management studies, EPM diagnostic and therapeutic research, and vitamin E-related neuromuscular disorders to construct a clinically actionable framework. The goal is to provide differential-diagnosis-aware feeding and management recommendations that avoid over-attributing neurologic signs to dietary management failures when infectious disease is present, while simultaneously optimizing metabolic support for muscle function.
1.3 Scope and Objectives
This systematic review examines: (1) diagnostic criteria distinguishing PSSM from EPM; (2) clinical sign overlap and misdiagnosis risk; (3) nutritional interventions including NSC restriction, fat supplementation, vitamin E, and protein support; (4) rehabilitation approaches; and (5) outcome monitoring for horses with suspected concurrent or sequential diagnoses. The ultimate objective is a differential-diagnosis-aware feeding framework applicable to clinical practice.

Figure 1. Clinical sign overlap between PSSM and EPM in horses, illustrating the diagnostic challenge posed by shared presentations and the importance of ruling out infectious neurologic disease before attributing signs solely to metabolic myopathy.
---
2. Diagnostic Criteria and Disease Characterization
2.1 Polysaccharide Storage Myopathy: Types and Diagnosis
PSSM encompasses two distinct subtypes with different diagnostic approaches. PSSM1 is definitively diagnosed through genetic testing for the GYS1 R309H mutation, which has been identified across a variety of breeds including Quarter Horses, Warmbloods, draft breeds, and their crosses [3]. The GYS1 mutation was found in 24/37 (65%) of grade 2 PSSM cases in UK horses, demonstrating that while it is an important cause of exertional rhabdomyolysis, it does not account for all forms of PSSM. Genetic testing is recommended in cases of exertional rhabdomyolysis prior to or in combination with muscle biopsy [3]. The mutation prevalence varies significantly by breed, with studies showing particularly high frequencies in draft breeds—90% in Percheron horses and 49.9% in Russian heavy draft horses [4].
PSSM2 presents a more complex diagnostic picture. Type 2 polysaccharide storage myopathy in Quarter Horses has been characterized as a novel glycogen storage disease distinct from PSSM1, with intermediate histopathological scores for amylase-resistant polysaccharide (1.4 ± 0.6 compared to 2.1 ± 0.3 in PSSM1) and glycogen concentrations (129 ± 62 versus 175 ± 9 mmol/kg in PSSM1) [5]. Importantly, commercial genetic tests for variants in MYOT, FLNC, and MYOZ3 genes have been evaluated and found to have no association with histopathological diagnosis of PSSM2, with these variants improperly diagnosing PSSM2 in 57% of healthy Quarter Horses while failing to diagnose 40% of horses with histopathological evidence of the condition [6]. This underscores that muscle biopsy remains the gold standard for PSSM2 diagnosis.
2.2 Equine Protozoal Myeloencephalitis: Diagnostic Approaches
EPM diagnosis relies on clinical presentation, immunodiagnostics performed on serum and cerebrospinal fluid (CSF), and exclusion of other neurological disorders [7]. Real-time PCR detection of S. neurona DNA in CSF represents a valuable complement to traditional immunodiagnostic approaches. In a study of 210 horses with suspected neurologic disease, 25 horses tested positive for S. neurona by rtPCR only, while 30 had intrathecally-derived antibodies only (serum:CSF ratio ≤64 by IFAT), and 13 tested positive by both methods. Horses with prior anti-protozoal treatment were more likely to test positive solely by IFAT, while treatment-naive horses were more likely to test positive by rtPCR [7].
The diagnostic picture is further complicated by evidence of multiple potential causative agents. Research has demonstrated intrathecal production of anti-Toxoplasma gondii antibodies in 10% of EPM-suspect horses, indicating that horses may present with EPM-like clinical signs while testing negative for the primary causative agents [8]. Elevated soluble CD14 concentrations in CSF samples from EPM-affected horses compared to controls represent a potential inflammatory biomarker that may complement clinical evaluation and standard antibody testing [2].
2.3 Vitamin E-Related Differential Diagnoses
Several vitamin E-deficient conditions must be considered in the differential diagnosis of horses with muscle atrophy and gait abnormalities. Equine neuroaxonal dystrophy/equine degenerative myeloencephalopathy (eNAD/EDM) is an inherited neurodegenerative disease associated with vitamin E deficiency in the first year of life and represents the second most common cause of spinal ataxia in horses euthanized for neurologic disease [9]. Clinical signs include symmetric ataxia (>grade 2/5), wide-base stance at rest, and proprioceptive defects. Conclusive diagnosis requires postmortem histologic evaluation, though research on antemortem biomarkers is ongoing [9].
Equine motor neuron disease (EMND) is a neurodegenerative disorder of the somatic lower motor neurons resulting in diffuse neuromuscular disease in adult horses, with vitamin E deficiency established as a significant risk factor [10]. Clinical features include weakness, weight loss, muscle atrophy, and muscle fasciculations [11]. The median time to develop EMND in vitamin E-deficient horses was 38.5 months in a field trial, demonstrating the cumulative nature of oxidative damage [12].
---
3. Clinical Sign Overlap and Misdiagnosis Risk
3.1 Shared Clinical Manifestations
The clinical overlap between PSSM and EPM centers on several key presentations that complicate differential diagnosis. Both conditions can manifest with muscle atrophy, exercise intolerance, reluctance to work, and gait abnormalities. In PSSM2 Warmblood horses, abnormal exercise responses reported by owners began at approximately 6 years of age and included decline in performance, reluctance to collect, and reluctance to go forward in over 50% of horses [13]. These nonspecific signs of reduced willingness to work and performance decline mirror early presentations of EPM, where subtle clinical signs originally considered unimportant may progress to obvious neurologic signs [14].
Exertional rhabdomyolysis, the hallmark of PSSM, occurs predominantly in performance horses—barrel racing and working cow/roping types in the case of PSSM2-QH [5]. However, horses with neurologic disease may also demonstrate exercise-related deterioration, and muscle enzyme elevations can occur secondary to recumbency or abnormal gait patterns in EPM cases. The distinction requires careful characterization of whether muscle damage is primary (metabolic) or secondary (neurogenic).
3.2 Distinguishing Features and Red Flags
Key distinguishing features favor EPM when asymmetric neurologic deficits are present, when ataxia is the predominant presentation, or when cranial nerve involvement is documented. EPM-affected horses demonstrate asymmetric ataxia and focal neurologic deficits that would be atypical for PSSM. Conversely, classic tying-up episodes with elevated serum creatine kinase, particularly occurring post-exercise with subsequent resolution, strongly suggest myopathic rather than neurogenic disease. Research has demonstrated that serum CK activity is significantly higher with high-starch diets compared to low-starch/high-fat diets in PSSM horses, providing both a diagnostic marker and treatment response indicator [15].
Red flags for concurrent disease include: partial response to appropriate single-condition therapy; progression of neurologic signs despite optimal PSSM dietary management; or muscle enzyme normalization without corresponding improvement in gait quality. Horses with EMND present with a combination of neurologic and muscular signs—weakness, muscle atrophy, and muscle fasciculations—that may initially be confused with either PSSM or EPM [11].
3.3 Breed and Risk Factor Considerations
Breed predisposition provides important diagnostic context. PSSM1 affects draft breeds with particularly high frequency, while Quarter Horses and related breeds are affected by both PSSM1 and PSSM2 [4]. EPM, by contrast, lacks strong breed predisposition but is associated with geographic exposure to the definitive host (opossum) and environmental risk factors. Horses with restricted access to pasture and genetic predispositions, such as Morgan horses, are particularly susceptible to vitamin E deficiency-related neuromuscular diseases [16].
Management factors significantly influence disease risk. Horses maintained without access to green pasture face elevated risk for vitamin E deficiency-related conditions including EMND. Feeding complete pelleted feed as the sole concentrate source, without pasture access, was associated with significantly increased EMND risk, while horses exercised on green pasture or in grass paddocks were less likely to develop EMND [17]. This nutritional history may help distinguish primary PSSM from vitamin E-related conditions in the differential.
---
4. Nutritional Management Strategies
4.1 Non-Structural Carbohydrate (NSC) Restriction for PSSM
The cornerstone of PSSM dietary management is restriction of non-structural carbohydrates. Research has demonstrated that diets with less than 5% digestible energy (DE) from starch combined with greater than 12% DE from fat can reduce exertional rhabdomyolysis in PSSM horses, potentially by increasing availability of free fatty acids for muscle metabolism [15]. This study using a 4×4 Latin square design showed that log CK activity was significantly higher with higher starch diets (21.2%, 14.8%, 8.4% DE) compared to the lowest starch diet (3.9% DE), while postprandial insulin and glucose responses were correspondingly elevated on high-starch feeding.
Low starch and high fat diets are recommended for PSSM1 to prevent episodes of rhabdomyolysis, representing a classic example of nutrigenomics in equine practice [18]. Both PSSM1 and PSSM2-ER respond well to a low nonstructural carbohydrate, high fat diet combined with regular exercise [1]. The physiological basis involves reducing glycogen synthesis stimulation (by lowering insulin response) while providing alternative fuel substrates.
4.2 Fat Supplementation and Exercise Integration
Fat supplementation at greater than 12% of DE, combined with regular exercise, forms the therapeutic foundation for PSSM management. The response to dietary intervention in PSSM2 Warmbloods demonstrated that 80% of owners reported overall improvement with decreased rhabdomyolysis episodes following implementation of low-starch/fat-supplemented diet and exercise recommendations [13]. However, the incomplete response—with 53% still not advancing as expected and approximately one-third showing persistent reluctance to go forward and collect—highlights the limitations of extrapolating PSSM1 recommendations to PSSM2.
Individual variation occurs in PSSM horse responses to diets differing in starch and fat content [15]. This variability underscores the need for monitoring and adjustment. Muscle oxidative capacity and lipid stores were low in PSSM horses, while muscle glycogen and abnormal polysaccharide content remained high even on very low starch diets, indicating that dietary management ameliorates clinical signs without necessarily correcting the underlying metabolic defect.
4.3 Vitamin E Supplementation Strategies
Vitamin E supplementation serves different purposes across these conditions. In PSSM, vitamin E provides antioxidant support but is not the primary therapeutic intervention. In contrast, vitamin E is central to the pathogenesis and management of EMND and eNAD/EDM. Vitamin E dietary deficiencies can lead to neuromuscular diseases including eNAD/EDM, EMND, and vitamin E deficient myopathy [19]. Supplementation is effective at slowing or halting clinical signs of some of these diseases, but the neuromuscular damage is usually irreversible except in vitamin E deficient myopathy.
Individual variation in vitamin E absorption and metabolism is substantial. Research on nutrigenomics of vitamin E demonstrates that some horses obtain high serum concentrations with minimal intake while others require high doses to remain in normal range [18]. A study comparing natural (d-alpha-tocopherol acetate) versus synthetic (dl-alpha-tocopherol acetate) supplementation at 2,000 IU daily found that both forms increased serum alpha-tocopherol to normal ranges (2.0-4.0 μg/mL) within 4 weeks in horses with marginal/deficient baseline levels [16].
For eNAD/EDM prevention, current recommendations emphasize providing pregnant mares and foals access to pasture, or supplementing with high doses of water-soluble RRR-α-tocopherol during the last trimester and continuing through the first two years of life [9]. Horses with eNAD/EDM have demonstrated increased α-tocopherol metabolism, with upregulated expression of the CYP4F2 enzyme, highlighting the need for high-dose supplementation in genetically susceptible individuals [20].

Figure 2. Comparison of nutritional management priorities across PSSM, EPM supportive care, and concurrent presentation scenarios. Values above bars indicate recommended ranges for each parameter.
---
5. EPM Treatment and Supportive Care Considerations
5.1 Anti-Protozoal Therapy and Treatment Duration
Treatment of confirmed EPM cases requires anti-protozoal medication, typically administered until CSF antibody tests return negative. In an epizootic of EPM on a Kentucky farm, treatment duration ranged from 45 to 211 days, with one horse persistently maintaining CSF antibodies to S. neurona [14]. Adverse effects from pyrimethamine and trimethoprim-sulfamethoxazole administration included transient fever, anorexia, and depression in 2 horses; acute worsening of ataxia in 2 horses; mild anemia in 4 horses; and abortions in 3 horses. These treatment complications highlight the importance of careful monitoring during therapy.
5.2 Nutritional Support During EPM Recovery
Nutritional support for EPM recovery focuses on different priorities than PSSM management. Protein quality becomes paramount for horses experiencing neurogenic muscle atrophy, as muscle wasting secondary to denervation requires adequate amino acid substrate for any recovery potential. Vitamin E supplementation is typically recommended at higher doses (5,000-10,000 IU daily) to support nervous system recovery and provide antioxidant protection during the inflammatory and healing phases.
Unlike PSSM, NSC restriction is not therapeutically indicated for EPM per se; however, maintaining overall metabolic health through balanced nutrition supports immune function and recovery. Exercise during EPM recovery should be dictated by neurologic status and gait safety rather than a structured rehabilitation program. Horses should exercise as tolerated without forcing activity that could result in falls or injury due to ataxia.
5.3 Relapse Prevention and Long-Term Management
Relapse of EPM is a recognized concern, and long-term management may require periodic reassessment of neurologic status and CSF testing in horses with incomplete resolution. Subtle clinical signs that progress to obvious neurologic signs emphasize the importance of early recognition and treatment [14]. Horses that have recovered from EPM may remain at risk for neurologic deterioration with concurrent stressors or immunosuppression.
---
6. Integrated Management Framework for Concurrent Presentations
6.1 Diagnostic Algorithm for Overlapping Cases
When horses present with features suggestive of both myopathic and neurogenic disease, a systematic diagnostic approach is essential to avoid delayed treatment of infectious disease while managing metabolic concerns. The recommended sequence involves:
- Neurologic examination to characterize symmetry and severity of deficits
- Serum CK and vitamin E levels as baseline metabolic assessment
- CSF collection and analysis for EPM serologic and molecular testing when neurologic signs are present
- GYS1 genetic testing for PSSM1 in appropriate breeds
- Muscle biopsy if PSSM2 or other myopathy is suspected and genetic testing is negative

Figure 3. Clinical decision framework for differential diagnosis and management of horses presenting with features of both PSSM and EPM.
6.2 Differential-Diagnosis-Aware Feeding Framework
The following table presents a feeding framework that accounts for diagnostic uncertainty and the possibility of concurrent disease:
| Parameter | PSSM Primary | EPM Primary | Uncertain/Concurrent |
|---|---|---|---|
| NSC (% DE) | <5% | 10-15% (standard) | <5-8% (conservative) |
| Fat (% DE) | >12% | 5-8% (moderate) | >10% (supportive) |
| Vitamin E (IU/day) | 2,000-4,000 | 5,000-10,000 | 5,000-8,000 |
| Protein Quality | Standard | High (amino acid focus) | High quality |
| Exercise | Regular, structured | As neurologically tolerated | Gradual increase, gait-dependent |
| Forage Base | Grass hay, low NSC | Mixed, adequate pasture | Grass hay, vitamin E supplemented |
6.3 Critical Clinical Decision Points
A key principle is that neurologic signs inconsistent with metabolic myopathy should never be attributed solely to PSSM. If a horse with confirmed PSSM develops asymmetric ataxia, focal deficits, or progressive neurologic deterioration despite optimal dietary management, EPM or other neurologic disease must be ruled out. Conversely, horses with confirmed EPM may have concurrent metabolic myopathy contributing to muscle dysfunction beyond what would be expected from neurogenic atrophy alone.
Diet alone cannot resolve infectious neurologic disease. This critical point prevents inappropriate prolongation of dietary intervention when anti-protozoal therapy is indicated. The response to therapy provides diagnostic information: horses that fail to improve with appropriate PSSM dietary management warrant reevaluation, while horses that partially improve with anti-protozoal therapy but retain muscle dysfunction may have concurrent myopathic disease.

Figure 4. Role of vitamin E across equine neuromuscular disorders, illustrating varying importance in pathogenesis, reversibility with supplementation, and prevention potential.
6.4 Outcome Monitoring and Response Assessment
Monitoring parameters differ by primary diagnosis:
| Condition | Primary Outcomes | Monitoring Frequency | Relapse Indicators |
|---|---|---|---|
| PSSM | CK post-exercise, tying-up episodes, exercise tolerance | CK: every 2-4 weeks initially; Episodes: ongoing log | Elevated CK, stiffness, reluctance to work |
| EPM | Neurologic grade, gait quality, CSF testing | Neuro exam: every 2-4 weeks; CSF: end of treatment | Worsening ataxia, new deficits, behavioral changes |
| EMND/eNAD | Serum vitamin E, muscle mass, fasciculations | Vitamin E: every 4-8 weeks; Clinical: monthly | Declining vitamin E levels, progressive weakness |
| Concurrent | All above parameters | Combined schedule | Any deterioration in either system |
---
Summary and Conclusions
Key Synthesis Points
This integrated review highlights several critical principles for managing horses with suspected concurrent or overlapping PSSM and EPM:
- Diagnostic primacy of infectious disease: EPM should be ruled out in any horse presenting with neurologic signs, even when PSSM is suspected or confirmed, as diet cannot treat infectious neurologic disease.
- Complementary rather than competing approaches: Low-NSC/high-fat diets beneficial for PSSM do not contradict EPM supportive care; combined approaches are feasible when concurrent disease is present.
- Vitamin E serves multiple roles: As antioxidant support in PSSM, as neuroprotective agent during EPM recovery, and as preventive intervention for eNAD/EDM, vitamin E supplementation (5,000-10,000 IU daily) is appropriate across overlapping presentations [19].
- Response to therapy informs diagnosis: Incomplete response to single-condition management should prompt reevaluation for concurrent disease or alternative diagnosis.
- Individual variation demands monitoring: Both dietary response in PSSM [15] and vitamin E metabolism [18] show substantial individual variation, requiring outcome-based adjustment rather than protocol adherence alone.
Limitations and Research Gaps
Direct evidence of PSSM-EPM comorbidity remains absent from the current literature, necessitating the synthesis approach employed here. Future research should characterize the prevalence of concurrent diagnoses, optimize combined management protocols, and develop biomarkers that distinguish myopathic from neurogenic muscle dysfunction more precisely. The genetic basis of PSSM2 remains undefined [5], limiting diagnostic precision in horses without the GYS1 mutation. Additionally, antemortem diagnosis of eNAD/EDM is not currently available in any breed [9], complicating the differential in horses with vitamin E deficiency and neurologic signs.
Clinical Implications
For equine practitioners managing horses with overlapping muscle and neurologic signs, this framework emphasizes: (1) systematic diagnostic workup before committing to dietary management; (2) recognition that partial therapeutic response warrants expanded differential consideration; (3) vitamin E supplementation as a common supportive measure across conditions; and (4) outcome monitoring tailored to suspected primary diagnosis while remaining alert to concurrent disease. The goal is avoiding the diagnostic error of over-attributing neurologic signs to diet when infectious disease requires specific therapy, while still optimizing metabolic support for horses with confirmed or suspected myopathy.
References
References
Numbered in order of first citation, IEEE style. Each entry links to the original source via DOI or publisher URL.
- A. Firshman and S. Valberg, “Polysaccharide Storage Myopathy.,” The Veterinary clinics of North America. Equine practice, Jan. 2025. doi: 10.1016/j.cveq.2024.11.004. View source
- A. N. Hay, B. Wagner, C. M. Leeth, T. LeRoith, T. Cecere, K. Lahmers, F. Andrews, S. Werre, A. L. Johnson, C. Clark, N. Pusterla, S. Reed, D. Lindsay, S. D. Taylor, K. Estell, M. Furr, R. MacKay, F. Del Piero, and S. Witonsky, “Horses affected by EPM have increased sCD14 compared to healthy horses.,” Veterinary Immunology and Immunopathology, Oct. 2021. doi: 10.1016/j.vetimm.2021.110338. View source
- R. Stanley, M. E. McCue, S. Valberg, J. R. Mickelson, I. G. Mayhew, C. M. McGowan, C. Hahn, J. C. Patterson-Kane, and R. J. Piercy, “A glycogen synthase 1 mutation associated with equine polysaccharide storage myopathy and exertional rhabdomyolysis occurs in a variety of UK breeds.,” Equine Veterinary Journal, Jul. 2009. doi: 10.2746/042516409X407611. View source
- N. Blohina, S. I. Sorokin, L. Khrabrova, A. Zelenchenkova, and B. Bazaron, “Biodiversity and functional variability of the allele pool of horse breed populations,” IOP Conference Series: Earth and Environment, Sep. 2021. doi: 10.1088/1755-1315/848/1/012229. View source
- S. Valberg, Z. J. Williams, C. Finno, A. Schultz, D. Velez-Irizarry, M. Henry, K. Gardner, and J. Petersen, “Type 2 polysaccharide storage myopathy in Quarter Horses is a novel glycogen storage disease causing exertional rhabdomyolysis.,” Equine Veterinary Journal, Aug. 2022. doi: 10.1111/evj.13876. View source
- S. Valberg, M. Henry, K. L. Herrick, D. Velez-Irizarry, C. Finno, and J. Petersen, “Absence of myofibrillar myopathy in Quarter Horses with a histopathological diagnosis of type 2 polysaccharide storage myopathy and lack of association with commercial genetic tests,” Equine Veterinary Journal, Mar. 2022. doi: 10.1111/evj.13574. View source
- P. Bernardino, W. Smith, P. Conrad, A. Packham, E. Tamez-Trevino, S. Barnum, and N. Pusterla, “Molecular detection of Sarcocystis neurona in cerebrospinal fluid from 210 horses with suspected neurologic disease.,” Veterinary parasitology, Feb. 2021. doi: 10.1016/j.vetpar.2021.109372. View source
- P. Bernardino, N. Pusterla, P. Conrad, A. Packham, E. Tamez-Trevino, M. Aleman, K. James, and W. Smith, “Evidence of intrathecally-derived antibodies against Toxoplasma gondii in horses suspected of neurological disease consistent with equine protozoal myeloencephalitis.,” Veterinary parasitology, Mar. 2023. doi: 10.1016/j.vetpar.2023.109919. View source
- A. E. Young and C. J. Finno, “Current insights into equine neuroaxonal dystrophy/equine degenerative myeloencephalopathy.,” The Veterinary Journal, May 2024. doi: 10.1016/j.tvjl.2024.106129. View source
- R. D. L. Rúa-Doménech, H. Mohammed, J. F. Cummings, T. Divers, A. Lahunta, and B. Summers, “Association between plasma vitamin E concentration and the risk of equine motor neuron disease.,” The Veterinary Journal, Nov. 1997. doi: 10.1016/S1090-0233(97)80021-4. View source
- E. Diez de Castro, R. Zafra, L. Acevedo, J. Pérez, I. Acosta, J. Rivero, and E. Aguilera-Tejero, “Eosinophilic Enteritis in Horses with Motor Neuron Disease,” Journal of Veterinary Internal Medicine, Mar. 2016. doi: 10.1111/jvim.13944. View source
- H. Mohammed, T. Divers, B. Summers, and A. de Lahunta, “Vitamin E deficiency and risk of equine motor neuron disease,” Acta Veterinaria Scandinavica, Jul. 2007. doi: 10.1186/1751-0147-49-17. View source
- Z. J. Williams, M. Bertels, and S. Valberg, “Muscle glycogen concentrations and response to diet and exercise regimes in Warmblood horses with type 2 Polysaccharide Storage Myopathy,” PLoS ONE, Sep. 2018. doi: 10.1371/journal.pone.0203467. View source
- C. Fenger, D. Granstrom, J. Langemeier, and S. Stamper, “Epizootic of equine protozoal myeloencephalitis on a farm.,” Journal of the American Veterinary Medical Association, Apr. 1997. doi: 10.2460/javma.1997.210.07.923. View source
- W. P. Ribeiro, S. Valberg, J. D. Pagan, and B. Gustavsson, “The effect of varying dietary starch and fat content on serum creatine kinase activity and substrate availability in equine polysaccharide storage myopathy.,” Journal of Veterinary Internal Medicine, Nov. 2004. doi: 10.1111/j.1939-1676.2004.tb02637.x. View source
- C. Ballard, E. Jensen-Vargas, K. Lassell, and S. Dobbins, “PSVII-13 The Evaluation of Natural Vs Synthetic Vitamin E Supplementation for the Management of Alpha-Tocopherol Serum Concentrations in Morgan Horses,” Journal of Animal Science, Nov. 2023. doi: 10.1093/jas/skad281.588. View source
- R. D. L. Rúa-Domènech, H. O. Mohammed, J. F. Cummings, T. Divers, A. de Lahunta, and B. A. Summers, “Intrinsic, management, and nutritional factors associated with equine motor neuron disease.,” Journal of the American Veterinary Medical Association, Nov. 1997. doi: 10.2460/javma.1997.211.10.1261. View source
- C. J. Finno, “How Nutrigenomics Impacts Equine Health - A Case Study of Vitamin E.,” Journal of Equine Veterinary Science, Mar. 2025. doi: 10.1016/j.jevs.2025.105421. View source
- A. E. Young and C. J. Finno, “The role of vitamin E deficiency in equine neuromuscular disorders,” UK-Vet Equine, Jan. 2025. doi: 10.12968/ukve.2023.0049. View source
- E. Hales, H. Habib, G. Favro, S. Katzman, R. Sakai, S. A. Marquardt, M. H. Bordbari, B. Ming-Whitfield, J. A. M. Peterson, A. R. Dahlgren, V. Rivas, C. Ramirez, S. Peng, C. Donnelly, B. Dizmang, A. Kallenberg, R. Grahn, A. D. Miller, K. Woolard, B. Moeller, B. Puschner, and C. Finno, “Increased α‐tocopherol metabolism in horses with equine neuroaxonal dystrophy,” Journal of Veterinary Internal Medicine, Jul. 2021. doi: 10.1111/jvim.16233. View source
This review is one of 34 in the Foxwatch research corpus. Methodology: PRISMA 2020-style systematic literature review against PubMed, CAB Abstracts, Scopus, Web of Science, and equine veterinary conference proceedings.