Genetic Testing Access, Carrier Screening, Pre-Breeding
The cheapest and most informative diagnostic step in the PSSM workflow, and the one most often skipped. This review walks through the testing-laboratory landscape (UC Davis, Animal Genetics, Etalon, EquiSeq), breed-specific carrier frequencies, the pre-breeding decision, and the live scientific controversy over the marketed "PSSM2 polygenic panels." Read with reviews 01 (PSSM foundation) and 04 (PSSM2 evidence map).
Research thesis
This systematic review argues that genetic testing for the GYS1 R309H mutation is the single most informative diagnostic step for suspected PSSM1 and is widely underused in clinical practice, while the polygenic test panels marketed for PSSM Type 2 remain scientifically controversial and are routinely misapplied, not only because GYS1 testing is inexpensive, breed-validated across Quarter Horse, Belgian, Percheron, and Warmblood populations, and definitive in confirmed cases, but also because the so-called PSSM2 polygenic markers (P2, P3, P4, K1, Px) lack peer-reviewed validation in independent populations; by reviewing the testing-laboratory landscape (UC Davis, Animal Genetics, Etalon, EquiSeq), the breed-specific carrier-frequency literature, and the pre-breeding decision evidence, this review constructs a defensible genetic-testing decision framework and clearly separates validated from controversial test offerings.
Evidence visualizations & full pages
Evidence figures (2)
Figures referenced by the review's Figure N: captions. Click any chart for a full-size view. They are also rendered inline below their respective captions in the full review text further down.
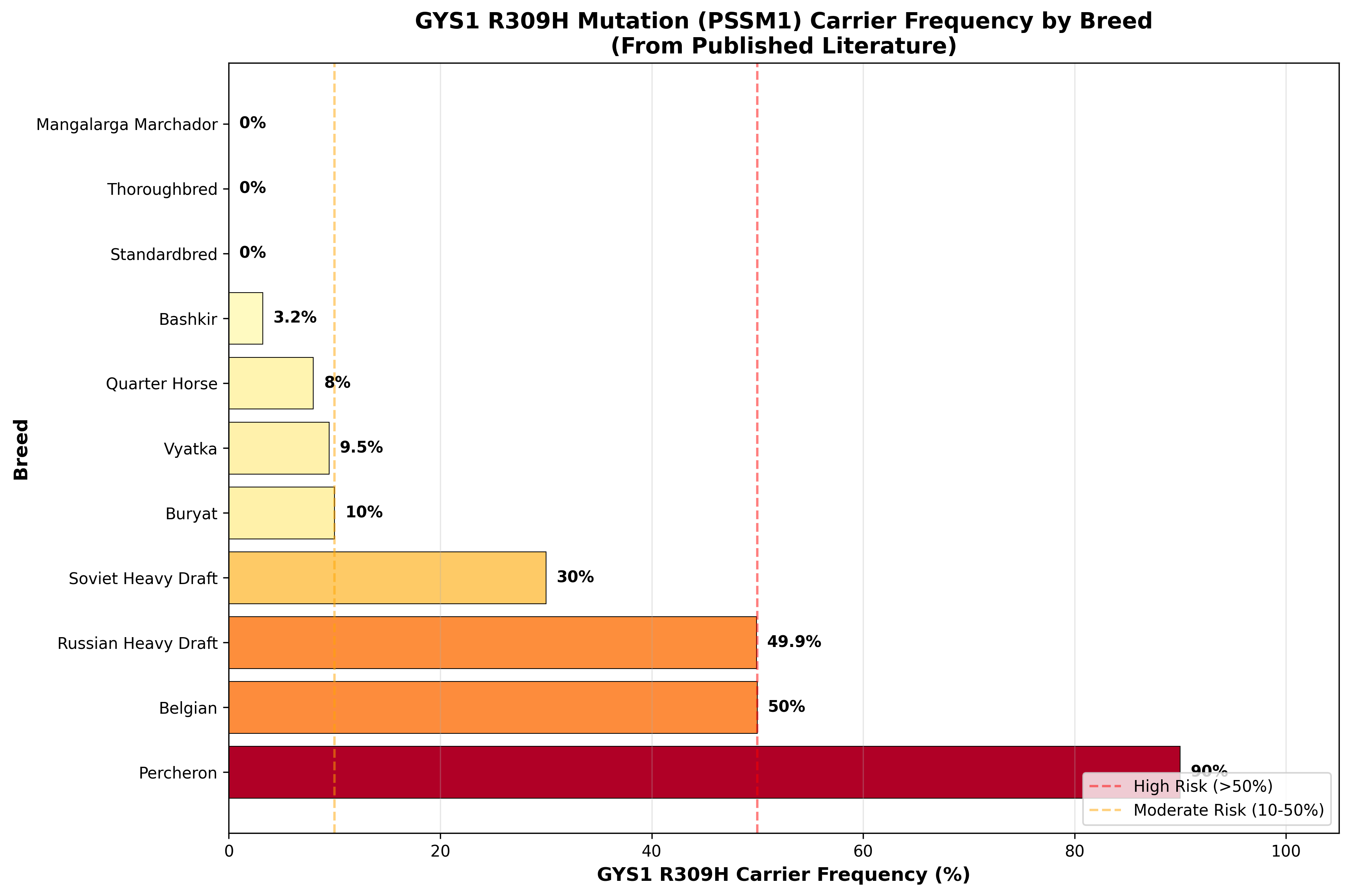 Figure 1: GYS1 R309H mutation carrier frequency by breed as documented in published literature. Draft breeds, particularly Percheron and Belgian, show the highest carrier frequencies, warranting mandatory gene
Figure 1: GYS1 R309H mutation carrier frequency by breed as documented in published literature. Draft breeds, particularly Percheron and Belgian, show the highest carrier frequencies, warranting mandatory gene
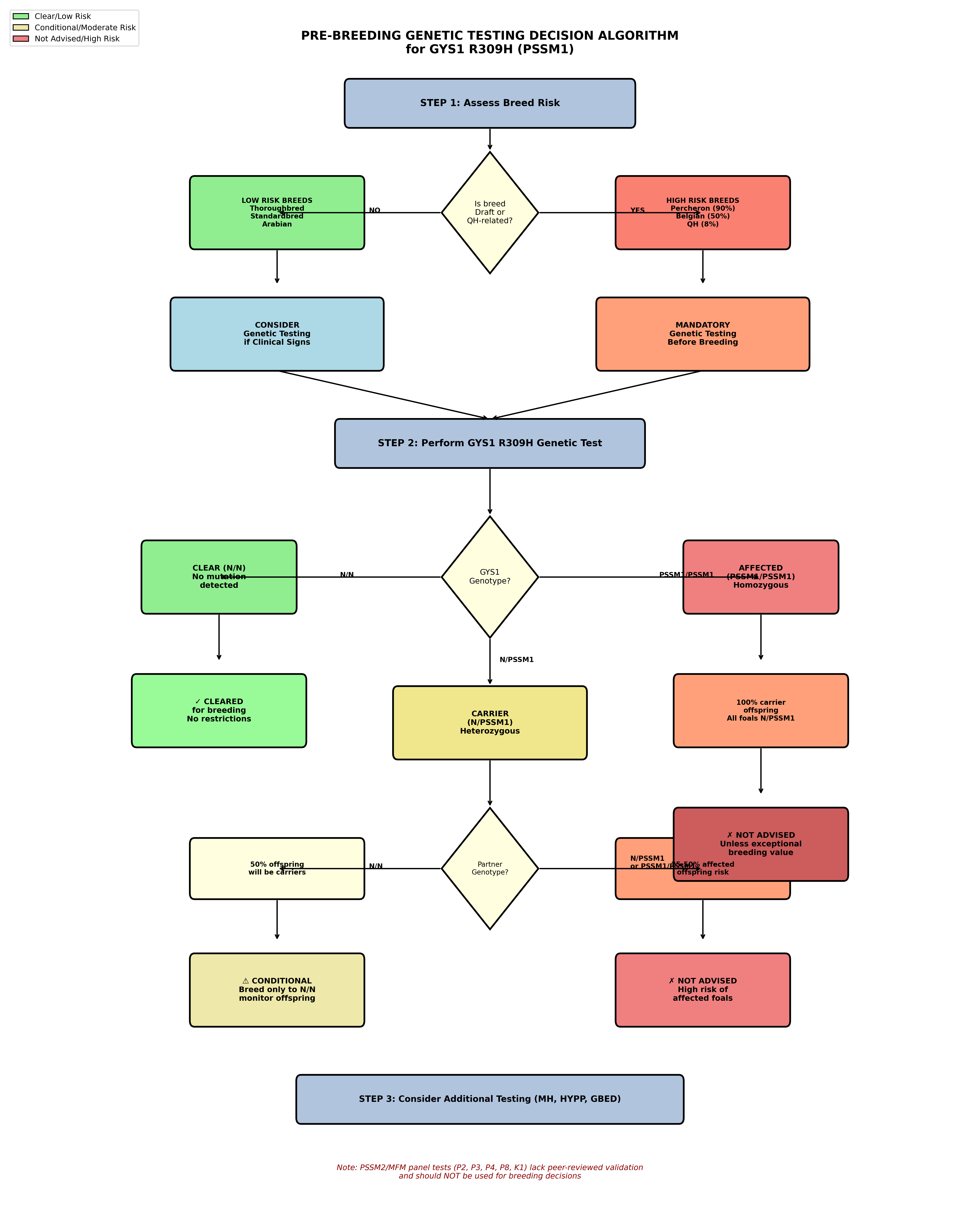 Figure 1: GYS1 R309H mutation carrier frequency by breed as documented in published literature. Draft breeds, particularly Percheron and Belgian, show the highest carrier frequencies, warranting mandatory gene
Figure 1: GYS1 R309H mutation carrier frequency by breed as documented in published literature. Draft breeds, particularly Percheron and Belgian, show the highest carrier frequencies, warranting mandatory gene
Original review pages (PDF render)
Download
Full review text
Genetic Testing Access, Carrier Screening, Pre-Breeding
Systematic Review: GYS1 R309H Mutation Testing Access, Carrier Screening, and Pre-Breeding Considerations in Equine Polysaccharide Storage Myopathy
1. Introduction and Background
1.1 Overview of GYS1 R309H Mutation and PSSM1
Type 1 Polysaccharide Storage Myopathy (PSSM1) is a common glycogenosis in horses caused by an R309H founder mutation in the glycogen synthase 1 gene (GYS1) [1]. This gain-of-function mutation leads to excessive accumulation of glycogen and amylase-resistant alpha-crystalline polysaccharide inclusions within skeletal muscle, resulting in a glycogen storage disease that causes exertional rhabdomyolysis [2]. The mutation causes a disruption in the amino acid sequence, which in turn disrupts an energy-producing process that allows muscles to operate normally. At least 20 breeds have been identified with Type 1 PSSM, with the prevalence being particularly high in draft horse breeds [3].
The clinical presentation of PSSM1 is characterized by muscle pain, weakness, reluctance to move, skin twitching, and sweating. Both type 1 (PSSM1) and type 2 polysaccharide storage myopathy (PSSM2) are characterized by aggregates of abnormal polysaccharide in skeletal muscle; however, whereas the genetic basis for PSSM1 is the known R309H GYS1 mutation, the cause of PSSM2 remains undefined [4]. This distinction is critical for accurate diagnosis and appropriate breeding management. In the field of equine muscle disorders, many conditions have a genetic basis, making genetic testing an important part of diagnostic evaluation [5].
1.2 Clinical Significance and Importance of Genetic Testing
The GYS1 mutation is an important cause of exertional rhabdomyolysis across multiple horse breeds, though it does not account for all forms of PSSM [6]. The clinical signs observed include rigid gait (particularly affecting pelvic limbs), muscle weakness, and recumbency, with horses presenting episodes of muscle disorder frequently after work [7]. Timely diagnostics and monitoring of horse breeds for the presence of genetic abnormalities that reduce their health and performance allow avoiding the accumulation of genetic burden in populations and contribute to increasing the efficiency of breeding programs [8].
Validated genetic tests are currently available for five equine muscle disorders: hyperkalemic periodic paralysis (HYPP), malignant hyperthermia (MH), glycogen branching enzyme deficiency (GBED), type 1 polysaccharide storage myopathy (PSSM1), and myosin heavy chain myopathy (MYHM) [5]. These diseases should be tested for in appropriate breeds with clinical signs of disease or as part of breeding management. Importantly, genetic testing in veterinary medicine is not regulated, and therefore any new genetic test offered in horses should be carefully evaluated and confirmed to be a valid test before use.
---
2. Genetic Testing Laboratories and Services
2.1 UC Davis Veterinary Genetics Laboratory (VGL)
The Veterinary Genetics Laboratory at UC Davis is a non-profit, self-supporting unit of the School of Veterinary Medicine that provides scientifically validated genetic testing services. The VGL offers PSSM1 testing as both a standalone test and as part of breed-specific disease panels [9]. The laboratory obtained a license to offer type 1 polysaccharide storage disease (PSSM1) testing, which is critical for informing horse owners and veterinarians on clinical, management, and breeding decisions across multiple breeds [10].
For the PSSM1 test at UC Davis VGL, the cost is $45 for a single test per animal, with an additional $15 for each subsequent test from a predefined list including GBED, HERDA, HYPP, MYHM, and MH [11]. Alternatively, the complete Quarter Horse Disease Panel (including all six tests) is available for $100 per animal. The turnaround time is at least 10 business days, though delays may occur if the sample requires additional testing or a new sample is requested [12]. Testing is performed under a license agreement with Equine LLC and the American Quarter Horse Association [11].
2.2 Animal Genetics Inc.
Animal Genetics Inc. is described as the largest private provider of genetic testing services for horses, dogs, and pigeons, offering a variety of reliable, state-of-the-art DNA tests [13]. The PSSM1 test is available for $40 per sample, with a turnaround time of 3-5 business days domestically [3]. The laboratory offers comprehensive breed-specific panels including the Quarter Horse Panel (HYPP, HERDA, GBED, PSSM, MH, IMM) for $75-95 [14].
Animal Genetics also provides combination testing packages with discounted pricing. The company tests for Polysaccharide Storage Myopathy Type 1 (PSSM1) and verifies the presence of the dominant PSSM mutation [15]. Results are reported as P1/P1 (homozygous affected), n/P1 (heterozygous affected), or n/n (clear). Notably, the laboratory is actively researching to identify genetic links in other breeds of horses that may be suffering from muscle disorders [15].
2.3 Etalon Diagnostics
Etalon Equine Genetics offers comprehensive genetic testing with a user-friendly online platform for accessing horse DNA test results [16]. Their DNA tests include screenings for all equine genetic diseases included on standard panels required by breed registries, plus more than 70 additional tests. Sample collection requires a simple, non-invasive pulled mane or tail hair sample, with all genetic data found in the root of the hair [16]. Diagnostic results are typically ready within 7-10 business days after sample receipt by the lab.
2.4 EquiSeq Inc.
EquiSeq is a biotechnology company founded in 2015 and headquartered in Albuquerque, New Mexico, offering genetic testing for horses with a focus on exercise intolerance conditions [17]. The company offers a MIM Panel for $299 to identify horses at risk for exercise intolerance before symptoms appear [18]. EquiSeq's researchers have identified genetic variants associated with Muscle Integrity Myopathy (MIM), a form of exercise intolerance found in many horse breeds that has also been called PSSM2 and MFM. Their DNA Panel tests for variants of MYOT, FLNC, MYOZ3, PYROXD1, COL6A3, and CACNA2D3 [18].
---
3. Comparative Laboratory Testing Summary
Table 1: Comparison of GYS1 R309H (PSSM1) Testing Services
| Laboratory | Test Cost | Panel Cost | Turnaround | Sample Type |
|---|---|---|---|---|
| UC Davis VGL | $45 (single) | $100 (6-panel) | 10+ business days | Hair (20-30 hairs with roots) |
| Animal Genetics Inc. | $40 (single) | $75-95 (breed panels) | 3-5 business days | Hair (30-40 hairs with roots) |
| Etalon Diagnostics | Variable | Comprehensive panels | 7-10 business days | Hair (mane or tail) |
| EquiSeq | N/A (PSSM2 focus) | $299 (MIM panel) | Variable | Hair or blood |
Table 2: Test Result Interpretation
| Genotype | Result | Clinical Interpretation | Breeding Recommendation |
|---|---|---|---|
| N/N | Clear | No mutation, no disease | Cleared for breeding |
| N/PSSM1 | Carrier (Heterozygous) | May show signs of disease, 50% transmission risk | Breed only to N/N partners |
| PSSM1/PSSM1 | Affected (Homozygous) | More severely affected, 100% transmission to offspring | Not recommended for breeding |
---
4. Heterozygous vs. Homozygous Phenotype Severity
4.1 Incomplete Dominance Pattern
Research demonstrates that PSSM1 exhibits incomplete dominance, with homozygous affected horses showing more severe pathology than heterozygotes [1]. A comprehensive study comparing matched cohorts of PSSM1 homozygotes, heterozygotes, and control horses revealed significant differences in resting plasma enzyme activities between groups. Median resting creatine kinase (CK) activities were 364 U/L for homozygotes, 301 U/L for heterozygotes, and 260 U/L for controls, with statistically significant differences between groups (P=0.04). Similarly, mean AST activity for homozygotes was 502 U/L, for heterozygotes 357 U/L, and for controls 311 U/L (P=0.01).
The study further demonstrated that resting plasma AST activity was significantly associated with the severity of subsarcolemmal vacuolation (rho=0.816; P=0.01) and cytoplasmic inclusions (rho=0.766; P=0.01) [1]. This association between plasma muscle enzyme activity and severity of underlying pathology suggests that physical disruption of myofibers contributes to the myopathic phenotype. There were also fewer type 2x and more type 2a muscle fibers in PSSM1-affected horses, indicating altered muscle fiber composition related to disease status.
4.2 Histopathological Differences
Histopathological scores, including the amount of amylase-resistant polysaccharide, differ significantly between PSSM1 and PSSM2 horses [4]. In PSSM1 Quarter Horses, scores averaged 2.1±0.3 compared to 1.4±0.6 for PSSM2 horses and 0±0 for controls (p<0.001). Glycogen concentrations were highest in PSSM1 horses at 175±9 mmol/kg, intermediate in PSSM2 at 129±62 mmol/kg, and lowest in controls at 80±27 mmol/kg (p<0.0001). Additionally, the abnormal polysaccharide in PSSM2 horses had a less filamentous ultrastructure than in PSSM1 horses.
Laboratory studies have also identified that the My allele, associated with susceptibility for myosin heavy chain myopathy (MYHM), follows a similar pattern where homozygotes (16/27) present a more severe phenotype compared to heterozygotes (6/33) [19]. This demonstrates that allele copy number effects are a common feature across equine muscle disease genetics.
---
5. Breed-Specific Prevalence of GYS1 R309H Mutation
5.1 High-Prevalence Draft Breeds
Population genetic studies have documented striking differences in GYS1 mutation frequency across breeds. In a study of 181 horses across ten breeds in Russia, the Percheron breed demonstrated the highest carrier frequency at 90% [8]. Russian heavy draft horses showed a frequency of 49.9%, Soviet heavy draft horses 30%, and the Buryat breed 10%. The Vyatka breed showed a prevalence of 9.5%, and the Bashkir breed 3.2%. Notably, Donskaya, Thoroughbred, and Orlov trotter horse breeds showed no PSSM1 mutation carriers.
Research confirms the prevalence of the PSSM1 mutation in Belgian horses is approximately 50% [3]. About 8% of Quarter Horse-related breeds carry this mutation. Case reports from Percheron horses confirm the clinical relevance of this mutation, with horses presenting two distinct clinical evolution patterns—one case showing severe multifocal rhabdomyonecrosis with moderate multifocal myoglobin nephrosis and severe diffuse transmural fibrinonecrotic gastritis, while another heterozygous case improved with management after two episodes of muscle disorder [7].
5.2 Light Horse and Pony Breeds
In the UK, the GYS1 mutation has been found across multiple breeds including Quarter Horse, Appaloosa, Warmblood, Connemara-cross, Cob, Polo Pony, and Thoroughbred cross [6]. Among 47 horses with exertional rhabdomyolysis, 10 (21%) carried the GYS1 mutation. The mutation was found in 24/37 (65%) of horses with grade 2 PSSM (amylase-resistant histopathology). Importantly, the mutation was only found in horses with grade 2 PSSM and not in horses with normal histology, idiopathic myopathy, or grade 1 PSSM biopsy samples.
The Mangalarga Marchador breed of Brazil, despite being the most numerous horse breed in the country and frequently exhibiting clinical signs of myopathy, showed no GYS1 mutation in breeding program animals evaluated [20]. This suggests that while myopathy is common in this breed, the underlying cause differs from PSSM1, warranting further histological analysis and molecular studies.
5.3 Carrier Frequency Map by Breed

Figure 1: GYS1 R309H mutation carrier frequency by breed as documented in published literature. Draft breeds, particularly Percheron and Belgian, show the highest carrier frequencies, warranting mandatory genetic testing before breeding [8],[3].
---
6. PSSM2/MFM Polygenic Test Panels: Scientific Status and Controversy
6.1 Commercial Tests and Gene Variants
Genetic tests for variants in MYOT (P2; rs1138656462), FLNC (P3a; rs1139799323 or P3b; rs1142918816), and MYOZ3 (P4; rs1142544043) genes are offered commercially to diagnose myofibrillar myopathy (MFM) and type 2 polysaccharide storage myopathy (PSSM2) in Quarter Horses [21]. The EquiSeq MIM Panel tests for variants of MYOT, FLNC, MYOZ3, PYROXD1, COL6A3, and CACNA2D3 [18]. These tests are marketed to identify horses at risk for developing exercise intolerance and to inform breeding decisions.
6.2 Scientific Validation Concerns
A rigorous peer-reviewed study investigated whether PSSM2 in Quarter Horses has histopathological features consistent with myofibrillar myopathy and evaluated the association between commercial genetic test variants and histopathologically diagnosed PSSM2 [21]. The study included 229 healthy control Quarter Horses and 163 PSSM2 Quarter Horses (GYS1 mutation negative). The results were striking:
Histopathological features of MFM were absent in all Quarter Horses examined. A P variant allele at any locus was not associated (P > 0.05) with a histopathological diagnosis of PSSM2. One or more P variants were common in both control Quarter Horses (57%) and PSSM2 Quarter Horses (61%). Allele frequencies between control and PSSM2 horses were: P2 (0.24/0.21), P3a (0.07/0.12), P3b (0.07/0.11), and P4 (0.06/0.08).
6.3 Diagnostic Accuracy Assessment
A receiver operator curve analysis did not accurately predict the PSSM2 phenotype (AUC = 0.67, 95% CI 0.62-0.72), and there was no difference in the total number of variant loci or total variant allele count between control and PSSM2 horses [21]. The researchers concluded that P2, P3, and P4 variants in genes associated with human MFM were not associated with PSSM2 in 392 Quarter Horses examined.
Critical finding: The use of these commercial tests would improperly diagnose PSSM2/MFM in 57% of healthy Quarter Horses and fail to diagnose PSSM2 in 40% of Quarter Horses with histopathological evidence of PSSM2 [21]. This represents an unacceptably high rate of both false positives and false negatives.
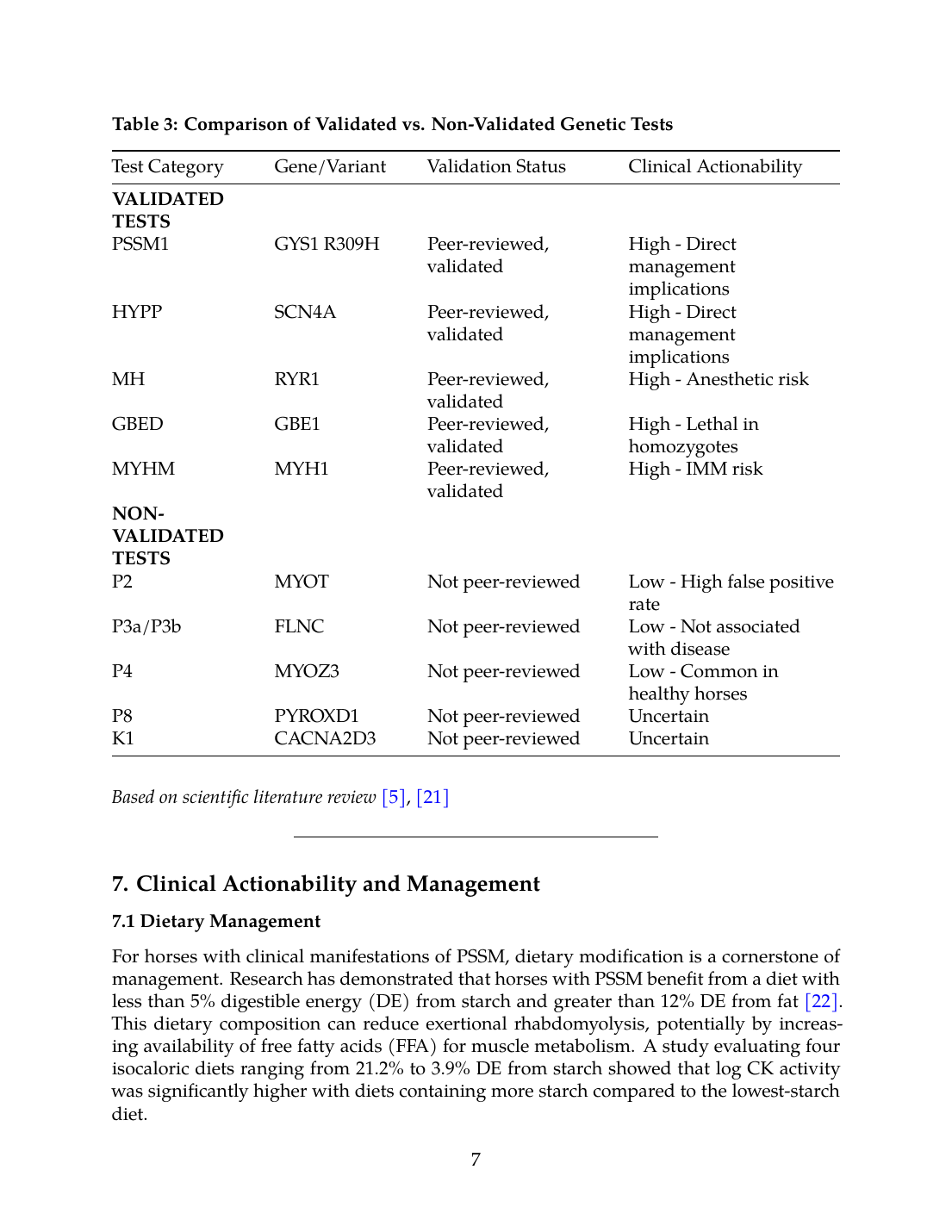
Table 3: Comparison of Validated vs. Non-Validated Genetic Tests
| Test Category | Gene/Variant | Validation Status | Clinical Actionability |
|---|---|---|---|
| VALIDATED TESTS | |||
| PSSM1 | GYS1 R309H | Peer-reviewed, validated | High - Direct management implications |
| HYPP | SCN4A | Peer-reviewed, validated | High - Direct management implications |
| MH | RYR1 | Peer-reviewed, validated | High - Anesthetic risk |
| GBED | GBE1 | Peer-reviewed, validated | High - Lethal in homozygotes |
| MYHM | MYH1 | Peer-reviewed, validated | High - IMM risk |
| NON-VALIDATED TESTS | |||
| P2 | MYOT | Not peer-reviewed | Low - High false positive rate |
| P3a/P3b | FLNC | Not peer-reviewed | Low - Not associated with disease |
| P4 | MYOZ3 | Not peer-reviewed | Low - Common in healthy horses |
| P8 | PYROXD1 | Not peer-reviewed | Uncertain |
| K1 | CACNA2D3 | Not peer-reviewed | Uncertain |
---
7. Clinical Actionability and Management
7.1 Dietary Management
For horses with clinical manifestations of PSSM, dietary modification is a cornerstone of management. Research has demonstrated that horses with PSSM benefit from a diet with less than 5% digestible energy (DE) from starch and greater than 12% DE from fat [22]. This dietary composition can reduce exertional rhabdomyolysis, potentially by increasing availability of free fatty acids (FFA) for muscle metabolism. A study evaluating four isocaloric diets ranging from 21.2% to 3.9% DE from starch showed that log CK activity was significantly higher with diets containing more starch compared to the lowest-starch diet.
Horses with PSSM should be maintained with low-starch diets, and individual variation occurs in the response of PSSM horses to diets differing in starch and fat content [22]. Muscle oxidative capacity and lipid stores were found to be low in PSSM horses, while muscle glycogen and abnormal polysaccharide content remained high regardless of diet, emphasizing the importance of consistent dietary management.
7.2 Exercise Management
Exertional rhabdomyolysis in PSSM2 Quarter Horses occurred predominantly in barrel racing and working cow/roping performance types and improved with regular exercise and a low starch/fat-supplemented diet [4]. The clinical response to management confirms that both dietary and exercise modifications are essential components of PSSM management. For clinical signs including stiffness, muscle weakness, and recumbency, therapeutic protocols include rest, gradual recovery, and subsequent limitation to light work, with no return of the clinical condition when properly managed [7].
7.3 Genotype-Phenotype Correlations and Clinical Decision-Making
A significant association exists between the number of rhabdomyolysis episodes and GYS1 mutation status (P = 0.03) and positive amylase-resistant PAS staining (P = 0.01) [23]. Nevertheless, the correlation between GYS1 mutation and PAS staining was weak (P = 0.06), confirming the versatile and complex nature of muscle disease. This emphasizes that while genetic testing provides valuable information, clinical assessment remains essential.
All cases with the malignant hyperthermia (MH) allele were fatal upon triggering by anesthesia, stress, or concurrent myopathy [19]. The RYR1 gene mutation that causes malignant hyperthermia in horses also influences PSSM severity, and can increase the severity of symptoms of PSSM1 in Quarter Horses and related breeds [3]. The combination of PSSM and MH genetic tests is therefore recommended for comprehensive assessment.
---
8. Pre-Breeding Decision Algorithm
8.1 Breeding Recommendations Based on Genotype
For PSSM1 specifically, breeding recommendations follow clear patterns based on genotype. Homozygous PSSM horses (P1/P1) are genetically bound to pass the gene to 100% of their progeny when bred, meaning all foals will have at least one copy of the dominant PSSM gene mutation [15]. Heterozygous carriers (n/P1) have a 50% chance of passing a PSSM allele to offspring. Clear horses (N/N) will not pass on the defective mutations to offspring [15].
The breeding program implications are significant: testing can be used to purchase horses with confidence, understand the genetic makeup of current horses, and aid in breeding better horses [17]. For young horses, hair samples should be taken from the tail rather than the mane to ensure adequate root material for testing [24].
8.2 Visual Decision Algorithm

Figure 2: Pre-breeding genetic testing decision algorithm for GYS1 R309H (PSSM1). This flowchart guides breeding decisions based on breed risk assessment and genetic test results. High-risk draft breeds require mandatory testing, while carrier horses should only be bred to clear partners [11],[15],[8].
---
9. Ethical Considerations for Breeding
9.1 Regulatory Landscape
Genetic testing in veterinary medicine is not regulated, and therefore any new genetic test offered in horses should be carefully evaluated and confirmed to be a valid test before use [5]. This lack of regulation creates significant ethical concerns, particularly regarding non-validated tests that may inappropriately influence breeding decisions. The American Quarter Horse Association (AQHA) requires a Five-Panel Genetic Disease Test for registration, with testing recommended through the registry to avoid additional recording fees [12].
9.2 Balancing Genetic Diversity and Disease Prevention
The high carrier frequencies in certain breeds, particularly draft breeds, present challenging ethical considerations. With Percheron horses showing 90% carrier frequency and Belgian horses at 50% [8], eliminating all carriers from breeding programs would dramatically reduce genetic diversity. A more nuanced approach involves breeding carriers only to clear horses while maintaining the broader genetic base. The 17 of 30 PSSM2 Quarter Horses with available pedigrees that descended from one of three stallions within four generations highlights the importance of monitoring founder effects in breeding populations [4].
9.3 Responsibility to Future Generations
Both muscle histological and genetic analyses are essential in the investigation of muscle disease, since 10% of horses with muscle disease and normal histology had a muscle disease-causing genetic variant, and 63% of histologically confirmed muscle with alterations had no known genetic variants [19]. This emphasizes that current genetic tests do not explain all muscle disease, and responsible breeding should incorporate multiple assessment modalities. The presence of reduced or normal enzyme levels does not preclude the presence of genetic mutations, making comprehensive genetic testing important even in clinically normal horses intended for breeding.
---
10. Conclusions and Recommendations
10.1 Key Findings
This systematic review confirms that GYS1 R309H mutation testing for PSSM1 is scientifically validated, clinically actionable, and essential for breeding management in high-risk breeds. Multiple accredited laboratories offer reliable testing at accessible price points ($40-45 for single tests) with reasonable turnaround times (3-10 business days). The mutation shows incomplete dominance with homozygotes exhibiting more severe pathology than heterozygotes. Carrier frequencies vary dramatically by breed, from 90% in Percheron horses to 0% in Thoroughbreds and Standardbreds.
In stark contrast, PSSM2/MFM polygenic test panels (P2, P3, P4, P8, K1) lack peer-reviewed validation. The P2, P3, and P4 variants were not associated with PSSM2 in a large study of 392 Quarter Horses, and their use would improperly diagnose PSSM2/MFM in 57% of healthy horses while failing to diagnose 40% of truly affected horses [21]. These tests should not be used for breeding decisions until proper validation studies are published.
10.2 Clinical Recommendations
- For all high-risk breeds (Draft horses, Quarter Horses, related breeds): Mandatory GYS1 R309H testing before breeding
- Heterozygous carriers: Breed only to N/N partners to reduce affected offspring
- Homozygous affected horses: Generally not recommended for breeding unless exceptional breeding value
- PSSM2/MFM panels: Not recommended for breeding decisions due to lack of validation
- Management: Low-starch (<5% DE), high-fat (>12% DE) diet with regular exercise for affected horses
10.3 Future Directions
Continued research is needed to identify the true genetic basis of PSSM2, which remains unknown despite characterization as a novel glycogen storage disorder distinct from PSSM1 [4]. The lack of regulation in veterinary genetic testing necessitates ongoing vigilance by veterinarians and breeders to distinguish validated from unvalidated tests. Standardization of testing protocols and results reporting across laboratories would benefit the equine community and improve breeding program outcomes.
References
References
Numbered in order of first citation, IEEE style. Each entry links to the original source via DOI or publisher URL.
- R. Naylor, L. Livesey, J. Schumacher, N. Henke, C. Massey, K. Brock, M. Fernández-Fuente, and R. Piercy, “Allele Copy Number and Underlying Pathology Are Associated with Subclinical Severity in Equine Type 1 Polysaccharide Storage Myopathy (PSSM1),” PLoS ONE, Jul. 2012. doi: 10.1371/journal.pone.0042317. View source
- R. Naylor, V. Luis-Fuentes, L. Livesey, C. Mobley, N. Henke, K. Brock, M. Fernández-Fuente, and R. Piercy, “Evaluation of cardiac phenotype in horses with type 1 polysaccharide storage myopathy.,” Journal of Veterinary Internal Medicine, Nov. 2012. doi: 10.1111/j.1939-1676.2012.00988.x. View source
- “Polysaccharide Storage Myopathy – Type 1 (PSSM1) - Animal Genetics,” internet, Nov. 2022. Available online
- S. Valberg, Z. J. Williams, C. Finno, A. Schultz, D. Velez-Irizarry, M. Henry, K. Gardner, and J. Petersen, “Type 2 polysaccharide storage myopathy in Quarter Horses is a novel glycogen storage disease causing exertional rhabdomyolysis.,” Equine Veterinary Journal, Aug. 2022. doi: 10.1111/evj.13876. View source
- C. J. Finno, “Genetics of Muscle Disease.,” The Veterinary clinics of North America. Equine practice, Jan. 2025. doi: 10.1016/j.cveq.2024.10.002. View source
- R. Stanley, M. E. McCue, S. Valberg, J. R. Mickelson, I. G. Mayhew, C. M. McGowan, C. Hahn, J. C. Patterson-Kane, and R. J. Piercy, “A glycogen synthase 1 mutation associated with equine polysaccharide storage myopathy and exertional rhabdomyolysis occurs in a variety of UK breeds.,” Equine Veterinary Journal, Jul. 2009. doi: 10.2746/042516409X407611. View source
- M. B. S. Teixeira, G. R. Bonatto, T. G. Cristo, D. Hemckmeier, N. L. Pisetta, and R. Casagrande, “Polysaccharide storage myopathy in two Percheron horses,” Revista de Ciências Agroveterinárias, Mar. 2023. doi: 10.5965/223811712212023078. View source
- N. Blohina, S. I. Sorokin, L. Khrabrova, A. Zelenchenkova, and B. Bazaron, “Biodiversity and functional variability of the allele pool of horse breed populations,” IOP Conference Series: Earth and Environment, Sep. 2021. doi: 10.1088/1755-1315/848/1/012229. View source
- J. 11 and 2020, “PSSM1 | Veterinary Genetics Laboratory,” internet, Jun. 2020. Available online
- R. W. J. 15 and 2020, “UC Davis Veterinary Genetics Laboratory Launches New Website, DNA Tests,” internet, Jun. 2020. Available online
- “Polysaccharide Storage Myopathy (PSSM1),” internet, Unknown Year. Available online
- “Quarter Horse & Related Breeds Disease Panel (5-panel plus),” internet, Jan. 2025. Available online
- “Equine Services - Animal Genetics,” internet, Sep. 2023. Available online
- “Quarter Horse Disorder Panel - Animal Genetics,” internet, Aug. 2023. Available online
- “Polysaccharide Storage Myopathy (PSSM) - Animal Genetics,” internet, Unknown Year. Available online
- “Etalon Equine Genetics,” internet, Unknown Year. Available online
- Exa, “EquiSeq,” internet, Feb. 2026. Available online
- P. shlyakhovskii, “Product – EquiSeq,” internet, Unknown Year. Available online
- M. Aleman, R. Scalco, J. Malvick, R. Grahn, A. True, and R. R. Bellone, “Prevalence of genetic mutations in horses with muscle disease from a neuromuscular disease laboratory.,” Journal of Equine Veterinary Science, Sep. 2022. doi: 10.2139/ssrn.4100989. View source
- N. V. D. Silva, A. L. H. Albuquerque, J. P. O. Oliveira Filho, A. S. Borges, and D. Delfiol, “Study of the mutation causing type 1 polysaccharide storage myopathy in a Mangalarga Marchador population used in breeding programs,” Acta Veterinaria Brasilica, Mar. 2023. doi: 10.21708/avb.2023.17.1.11422. View source
- S. Valberg, M. Henry, K. L. Herrick, D. Velez-Irizarry, C. Finno, and J. Petersen, “Absence of myofibrillar myopathy in Quarter Horses with a histopathological diagnosis of type 2 polysaccharide storage myopathy and lack of association with commercial genetic tests,” Equine Veterinary Journal, Mar. 2022. doi: 10.1111/evj.13574. View source
- W. P. Ribeiro, S. Valberg, J. D. Pagan, and B. Gustavsson, “The effect of varying dietary starch and fat content on serum creatine kinase activity and substrate availability in equine polysaccharide storage myopathy.,” Journal of Veterinary Internal Medicine, Nov. 2004. doi: 10.1111/j.1939-1676.2004.tb02637.x. View source
- C. A. Kósa, O. Szenci, L. Lénárt, I. Biksi, R. Szép, Á. Keresztesi, M. Mircean, M. Tăulescu, and O. Kutasi, “Genetic background of the Transylvanian endemic equine recurrent rhabdomyolysis.,” Acta Veterinaria Hungarica, Feb. 2026. doi: 10.1556/004.2026.01199. View source
- “Equine Sample Collection - Animal Genetics,” internet, Nov. 2022. Available online
This review is one of 34 in the Foxwatch research corpus. Methodology: PRISMA 2020-style systematic literature review against PubMed, CAB Abstracts, Scopus, Web of Science, and equine veterinary conference proceedings.