Systematic review of low-NSC and low-starch feeding thresholds in PSSM
What number should the hay analysis come in below? This review reads every study that has tested sugar-and-starch ceilings in PSSM horses and lands on a defensible answer: under 10% for confirmed Type 1, under 12% as a working ceiling for less certain diagnoses. The hay calculator on this site runs straight off these thresholds. Read this with review 07 (which lab fraction to ask for) and review 08 (year-round forage strategy).
Research thesis
This systematic review argues that the long-circulated "below ten percent NSC" rule for PSSM forage is a severity-tiered range, not a single number, with appropriate ceilings shifting by diagnostic certainty, workload, and concurrent fat supplementation, not only because reported clinical thresholds in the literature span less than five to less than fifteen percent of digestible energy from starch with no agreed cut point, but also because the dose-response data behind each tier come from heterogeneous populations and inconsistent forage-testing protocols; by stratifying PRISMA-eligible evidence across PSSM1, biopsy-confirmed PSSM, suspected PSSM2 and MFM, and unspecified tying-up cohorts, this review establishes defensible NSC ceilings for owner and veterinarian use and shows which numbers reflect trial data and which reflect expert consensus.
Evidence visualizations & full pages
Evidence figures (2)
Figures referenced by the review's Figure N: captions. Click any chart for a full-size view. They are also rendered inline below their respective captions in the full review text further down.
Original review pages (PDF render)
Download
Full review text
Low-NSC and low-starch feeding thresholds in PSSM
Systematic Review of Low-NSC and Low-Starch Feeding Thresholds in Equine Polysaccharide Storage Myopathy
Abstract
Polysaccharide storage myopathy (PSSM) represents a group of glycogen storage disorders in horses characterized by abnormal accumulation of glycogen and amylase-resistant polysaccharide in skeletal muscle, clinically manifesting as exertional rhabdomyolysis. Dietary nonstructural carbohydrate (NSC) restriction combined with fat supplementation has emerged as a cornerstone of management. This systematic review synthesizes evidence on NSC threshold recommendations for horses with PSSM1, PSSM2, and related glycogen storage myopathies, stratifying findings by diagnostic category and evaluating the strength of evidence supporting specific dietary targets. The review identifies significant heterogeneity in reported thresholds, with recommendations ranging from <5% to <15% of digestible energy from starch, predominantly derived from limited experimental studies and expert consensus rather than large-scale randomized trials.
---
1. Introduction
1.1 Background and Rationale
Type 1 Polysaccharide Storage Myopathy (PSSM1) is an autosomal dominant glycogen storage disorder affecting more than 20 breeds of horses that presents with a variety of signs, including exertional rhabdomyolysis [1]. The condition is diagnosed by genetic testing for the R309H mutation in the glycogen synthase 1 gene (GYS1) or by muscle biopsies containing muscle fibers with abnormal amylase-resistant polysaccharide [2]. PSSM is characterized by increased insulin sensitivity, excessive muscle glycogen storage, and the accumulation of amylase-resistant polysaccharide in muscle, with feeding of concentrates rich in hydrolyzable carbohydrates potentially enhancing disease expression by increasing the quantity of glucose available for muscle glycogen synthesis [3].
The pathophysiological basis for dietary intervention stems from observations that horses with PSSM have enhanced cellular uptake of glucose that may be, in part, caused by an increased sensitivity to insulin [4]. Studies have demonstrated that horses with PSSM have an increased rate of glucose clearance in response to insulin secretion, supporting the rationale that diets low in soluble carbohydrate may be the most effective way to decrease glycogen accumulation in skeletal muscle. Type 2 PSSM has recently been subdivided, with PSSM2-ER identified as a glycogen storage disorder in Quarter Horses that causes exertional rhabdomyolysis and is diagnosed by muscle biopsy as its genetic basis is unknown [1].
1.2 Scope and Objectives
This systematic review examines the evidence supporting specific NSC and starch restriction thresholds for horses diagnosed with PSSM1 (confirmed by GYS1 mutation testing), histopathologic PSSM (diagnosed by muscle biopsy), suspected PSSM2/myofibrillar myopathy (MFM), and unspecified tying-up cohorts. The primary objective is to identify reported NSC thresholds, assess the quality of evidence supporting these recommendations, and determine applicability across different PSSM subtypes.
---
2. Methods
2.1 Search Strategy and Information Sources
A comprehensive literature search was conducted following PRISMA 2020 and PRISMA-S reporting standards. Search terms included: "polysaccharide storage myopathy," "PSSM," "EPSM," "equine glycogen storage," "nonstructural carbohydrate," "NSC," "starch," "sugar," "water soluble carbohydrate," "ethanol soluble carbohydrate," "hay analysis," "pasture sugar," "glycogen," "insulin," "rhabdomyolysis," and "tying-up." Multiple electronic databases were searched for peer-reviewed articles examining dietary management of equine muscle glycogen storage disorders.
2.2 Eligibility Criteria and Study Selection
Studies were included if they: (1) involved horses with confirmed or suspected PSSM or related glycogen storage myopathies; (2) reported dietary interventions involving NSC or starch restriction; (3) provided quantitative data on NSC/starch levels, clinical outcomes, or muscle enzyme responses; and (4) were published in English in peer-reviewed journals. Case reports, experimental studies, retrospective analyses, and consensus guidelines were included to capture the full spectrum of available evidence.
2.3 Data Extraction and Quality Assessment
Data extracted included: NSC thresholds reported, actual diet composition, forage testing methods, diet duration, concurrent exercise protocols, fat supplementation levels, clinical response rates, CK/AST enzyme responses, relapse rates, and population characteristics. Evidence was categorized as experimentally validated, derived from expert consensus, extrapolated from metabolic physiology, or based on clinical experience.
---
3. Results
3.1 Pathophysiology and Metabolic Basis for Dietary Intervention
3.1.1 Enhanced Glucose Uptake in PSSM1
The fundamental rationale for NSC restriction derives from metabolic studies demonstrating altered glucose metabolism in affected horses. Research has shown that peak glucose concentrations during intravenous glucose tolerance tests were similar for PSSM and control horses, but the rate of glucose clearance was 1.5 times faster in horses with PSSM [4]. Furthermore, circulating concentrations of insulin before and after glucose injection were lower in the PSSM group, and horses with PSSM sustained low blood glucose concentrations for more than 3 hours after insulin injection, whereas blood glucose concentrations in controls returned to baseline values within 2 hours.
3.1.2 Molecular and Cellular Dysfunction
Gene expression profiling has revealed that PSSM muscles exhibit multiple metabolic disturbances beyond simple glycogen accumulation. Studies have demonstrated inflammation, glycogenesis inhibition, hypoxia, and mitochondrial dysfunctions in PSSM muscles [5]. Ultrastructural evaluation revealed a decrease in mitochondrial number and structural disorders, with extensive accumulation of abnormal polysaccharide displacing and partially replacing mitochondria and myofibrils. The up-regulation of glycogen synthase kinase-3 (GSK3β) under its active form could be responsible for glycogen synthase inhibition and hypoxia-inducible factor destabilization.
3.2 Experimentally Validated NSC Thresholds
3.2.1 The Ribeiro Latin Square Study (2004)
The most rigorous experimental evidence for specific starch thresholds comes from a controlled feeding trial using a 4×4 Latin square design [6]. This study evaluated 4 mares of Quarter Horse-related breeds with PSSM fed four isocaloric diets ranging in digestible energy from 21.2% (diet A), 14.8% (B), 8.4% (C), to 3.9% (D) for starch, and 7.2% DE (diet A), 9.9% (B), to 12.7% DE (diet C and D) for fat. The diets were fed for 6-week periods with 4 weeks of exercise.
Key findings demonstrated that postprandial insulin and glucose response was higher for diet A versus D, and log CK activity was higher with diets A, B, and C versus D [6]. Daily insulin was higher and free fatty acids lower on diet A versus B, C, and D, whereas glucose varied only slightly with diet. The study concluded that for horses with clinical manifestations of PSSM, a diet with <5% DE starch and >12% DE fat can reduce exertional rhabdomyolysis, potentially by increasing availability of free fatty acids for muscle metabolism.
3.2.2 Evidence Quality Assessment
This Latin square study represents the highest level of experimental evidence specifically addressing NSC thresholds in PSSM, though limitations include the small sample size (n=4), single breed representation, and 6-week duration per dietary phase. Muscle glycogen and abnormal polysaccharide content remained high on both highest and lowest starch diets, suggesting that dietary intervention primarily affects substrate availability during exercise rather than reversing underlying glycogen accumulation.
3.3 Clinical Response Studies by PSSM Subtype
3.3.1 PSSM1 Response to Dietary Management
Research has established that PSSM1 has incomplete dominance, with the severity of subclinical disease varying between homozygotes and heterozygotes [7]. Resting and post-exercise plasma creatine kinase and aspartate aminotransferase enzyme activity measurements demonstrated significant differences between groups, with median resting CK activities of 364 U/L for homozygotes, 301 U/L for heterozygotes, and 260 U/L for controls. Resting plasma AST activity was significantly associated with the severity of subsarcolemmal vacuolation and cytoplasmic inclusions, suggesting that physical disruption of myofibers may contribute to the myopathic phenotype.
3.3.2 PSSM2 in Warmblood Horses
A retrospective questionnaire study examined outcomes in PSSM2 Warmblood horses, finding that abnormal exercise responses reported by owners began at approximately 6 years of age and included a decline in performance, a reluctance to collect, and reluctance to go forward in over 50% of horses [8]. With the recommended diet and exercise regime, 80% of PSSM2 Warmblood owners reported an overall improvement with significant decreases in the proportion of horses showing a decline in performance and rhabdomyolysis. However, 53% of PSSM2 Warmblood horses were still not advancing as expected, with reluctance to go forward and collect persisting in approximately one-third of horses.
Importantly, this study found that median muscle glycogen concentrations did not differ between PSSM2 Warmblood horses and controls with no evident myopathy [8]. PSSM2 Warmblood horses with the highest glycogen concentrations were significantly more likely to show a decline in performance than those with lower glycogen concentrations, suggesting some correlation between glycogen accumulation and clinical signs even in this subtype.
3.3.3 PSSM2 in Quarter Horses
Investigation of PSSM2 in Quarter Horses demonstrated that exertional rhabdomyolysis occurred predominantly in barrel racing and working cow/roping performance types and improved with regular exercise and a low starch/fat-supplemented diet [9]. Histopathological scores including the amount of amylase-resistant polysaccharide (PSSM2-QH 1.4±0.6, PSSM1-QH 2.1±0.3, control-QH 0±0) and glycogen concentrations (PSSM2-QH 129±62, PSSM1-QH 175±9, control-QH 80±27 mmol/kg) were intermediate in PSSM2-QH with significant differences among groups.
3.4 Effect of Starch Intake on Muscle Enzyme Activity
3.4.1 Standardbred Trotter Evidence
Recent research in clinically healthy Standardbred trotters provides supporting evidence for the relationship between starch intake and muscle enzyme activity [10]. Post-exercise plasma AST activity was higher for high (451-967 g/day) and medium (988-1429 g/day) starch allowances than for horses with low starch allowances (0-268 g/day). The study concluded that management systems aiming for low levels of plasma muscle enzyme activity could offer diets with low starch (<450 g/day) contents and perhaps training regimes with shorter durations of high-intensity exercise.
While this study was conducted in horses without diagnosed PSSM, it provides important mechanistic support for the physiological basis of starch restriction, demonstrating that even in healthy horses, starch intake influences post-exercise muscle enzyme elevations.
3.5 Expert Consensus Recommendations
3.5.1 General Guidelines for PSSM Management
Expert consensus recommends that both PSSM1 and PSSM2-ER respond well to a low nonstructural carbohydrate, high fat diet combined with regular exercise [1]. A decrease in the frequency and severity of exertional rhabdomyolysis has been observed when horses with PSSM and RER are fed diets with reduced digestible energy from hydrolyzable carbohydrates (<10 to 15% of total diet) and increased digestible energy from fat (15 to 20%) and other energy sources, such as beet pulp and soybean hulls [3].
3.5.2 Nutritional Management Principles
Several heritable muscle disorders, including type 1 and 2 polysaccharide storage myopathy and recurrent exertional rhabdomyolysis, can be managed in part by restricting dietary nonstructural carbohydrate intake [11]. This recommendation is based on the understanding that glucose can support both anaerobic and aerobic exercise, whereas fat can only be metabolized aerobically, necessitating careful consideration of the horse's work intensity when formulating diets.
---
4. Comparative Analysis Across PSSM Subtypes
4.1 Differential Response to Dietary Intervention
The evidence indicates substantial differences in response to dietary management across PSSM subtypes. The diet and exercise recommendations ideal for PSSM1 improve but do not eliminate the decline in performance and reluctance to go forward under saddle characteristic of PSSM2 [8]. This finding suggests that while the same general dietary approach may be applied across subtypes, expectations for clinical improvement should be calibrated based on the specific diagnosis.
4.2 Forage Selection Considerations
Alternative forage options may provide advantages for NSC-sensitive horses. Studies evaluating native warm-season grasses found NSC levels ranging from 4.4% to 5.4%, below maximum recommended concentrations for horses susceptible to laminitis [12]. Voluntary dry matter intake of native grass hays by horses was lower than orchardgrass (1.1-1.3% vs 1.7% of body weight/day), suggesting these forages may offer benefits for calorie restriction while maintaining low NSC intake.
---
5. Summary Tables
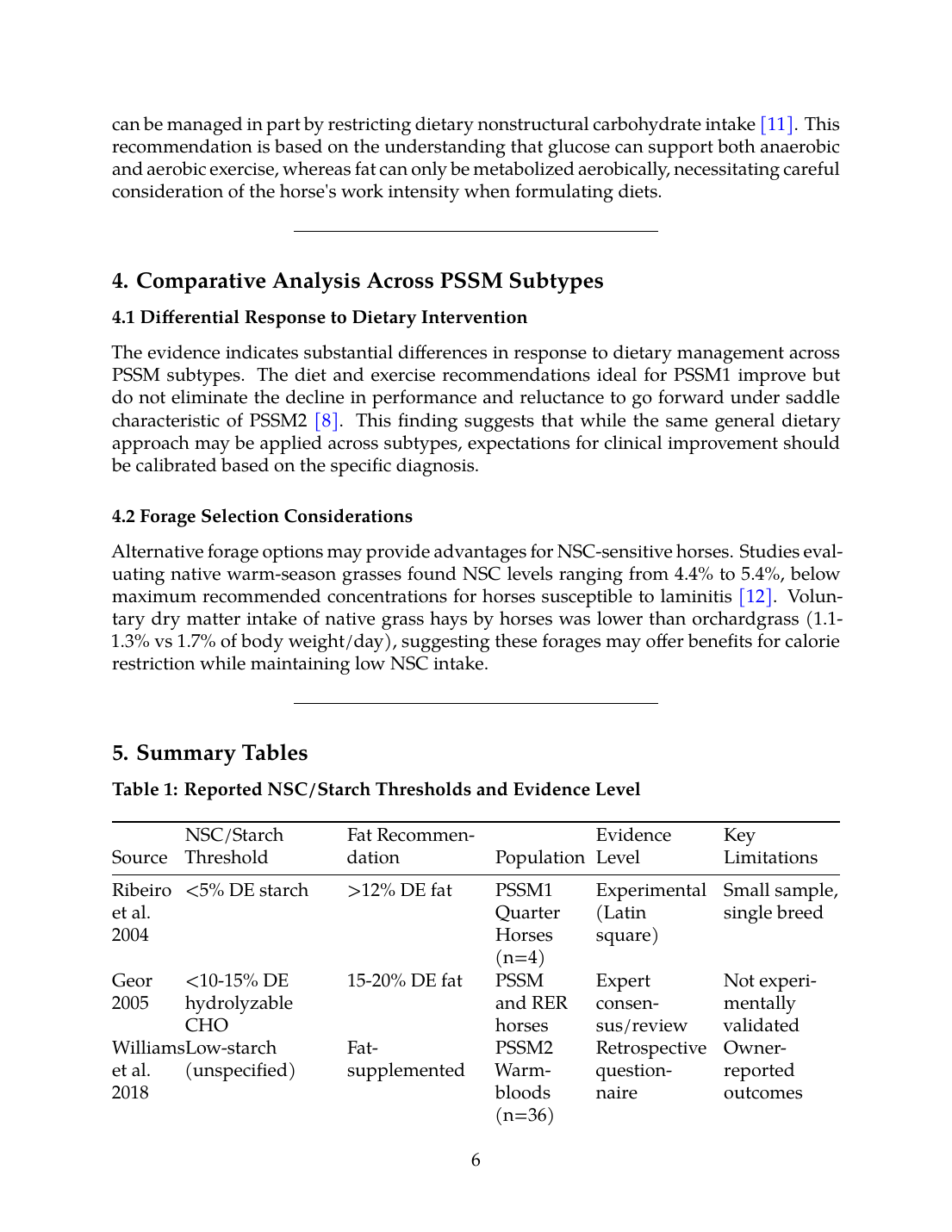
Table 1: Reported NSC/Starch Thresholds and Evidence Level
| Source | NSC/Starch Threshold | Fat Recommendation | Population | Evidence Level | Key Limitations |
|---|---|---|---|---|---|
| Ribeiro et al. 2004 | <5% DE starch | >12% DE fat | PSSM1 Quarter Horses (n=4) | Experimental (Latin square) | Small sample, single breed |
| Geor 2005 | <10-15% DE hydrolyzable CHO | 15-20% DE fat | PSSM and RER horses | Expert consensus/review | Not experimentally validated |
| Williams et al. 2018 | Low-starch (unspecified) | Fat-supplemented | PSSM2 Warmbloods (n=36) | Retrospective questionnaire | Owner-reported outcomes |
| Valberg et al. 2022 | Low-starch/fat-supplemented | Not quantified | PSSM2-QH (n=64) | Retrospective case-control | Observational design |
| Firshman & Valberg 2025 | Low NSC/high fat | Combined with exercise | PSSM1 and PSSM2-ER | Expert review | Consensus recommendation |
| Urschel & McKenzie 2021 | NSC restriction | Not specified | PSSM1, PSSM2, RER | Review/consensus | General guidance only |
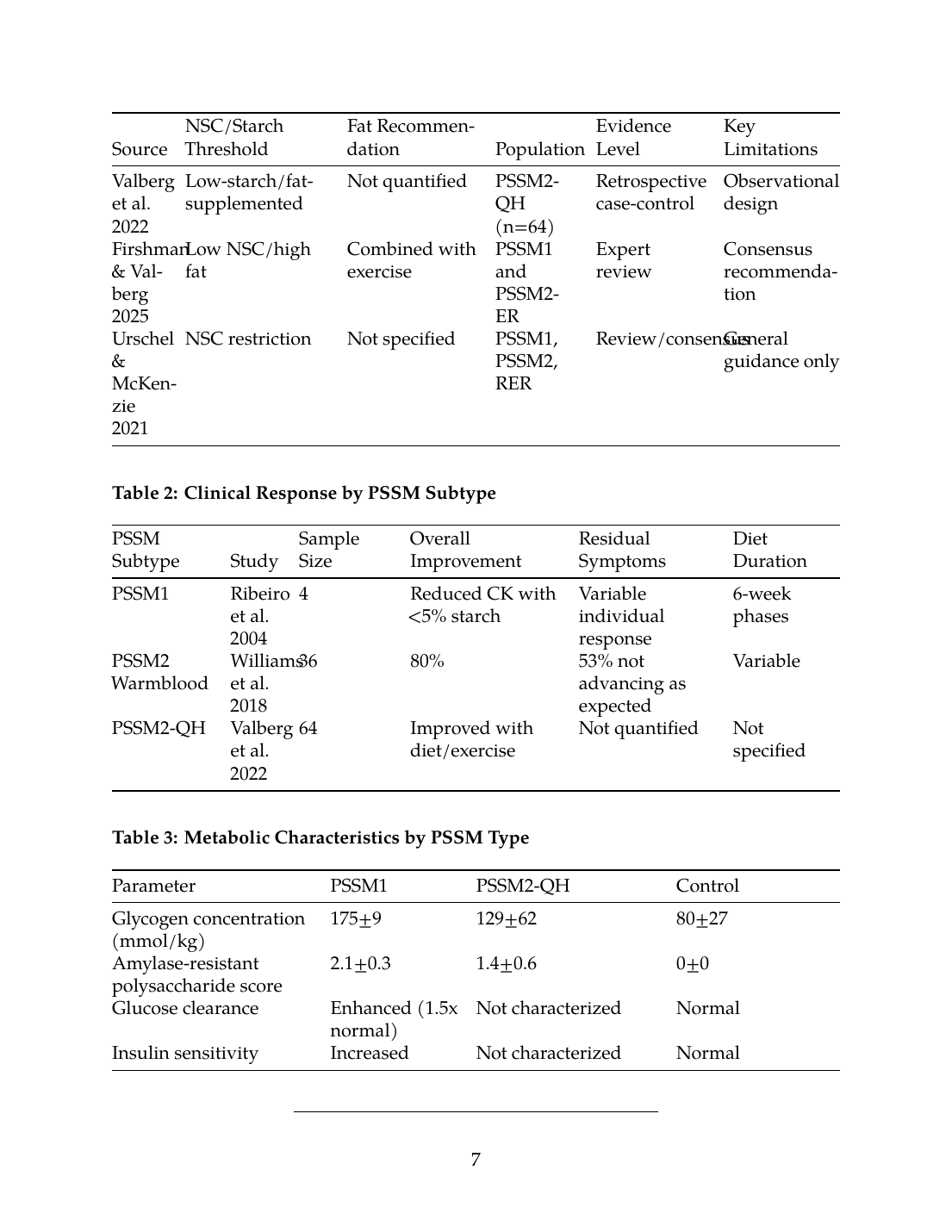
Table 2: Clinical Response by PSSM Subtype
| PSSM Subtype | Study | Sample Size | Overall Improvement | Residual Symptoms | Diet Duration |
|---|---|---|---|---|---|
| PSSM1 | Ribeiro et al. 2004 | 4 | Reduced CK with <5% starch | Variable individual response | 6-week phases |
| PSSM2 Warmblood | Williams et al. 2018 | 36 | 80% | 53% not advancing as expected | Variable |
| PSSM2-QH | Valberg et al. 2022 | 64 | Improved with diet/exercise | Not quantified | Not specified |
Table 3: Metabolic Characteristics by PSSM Type
| Parameter | PSSM1 | PSSM2-QH | Control |
|---|---|---|---|
| Glycogen concentration (mmol/kg) | 175±9 | 129±62 | 80±27 |
| Amylase-resistant polysaccharide score | 2.1±0.3 | 1.4±0.6 | 0±0 |
| Glucose clearance | Enhanced (1.5x normal) | Not characterized | Normal |
| Insulin sensitivity | Increased | Not characterized | Normal |
6. Visualizations
Figure 1: Dietary Thresholds and Evidence Hierarchy
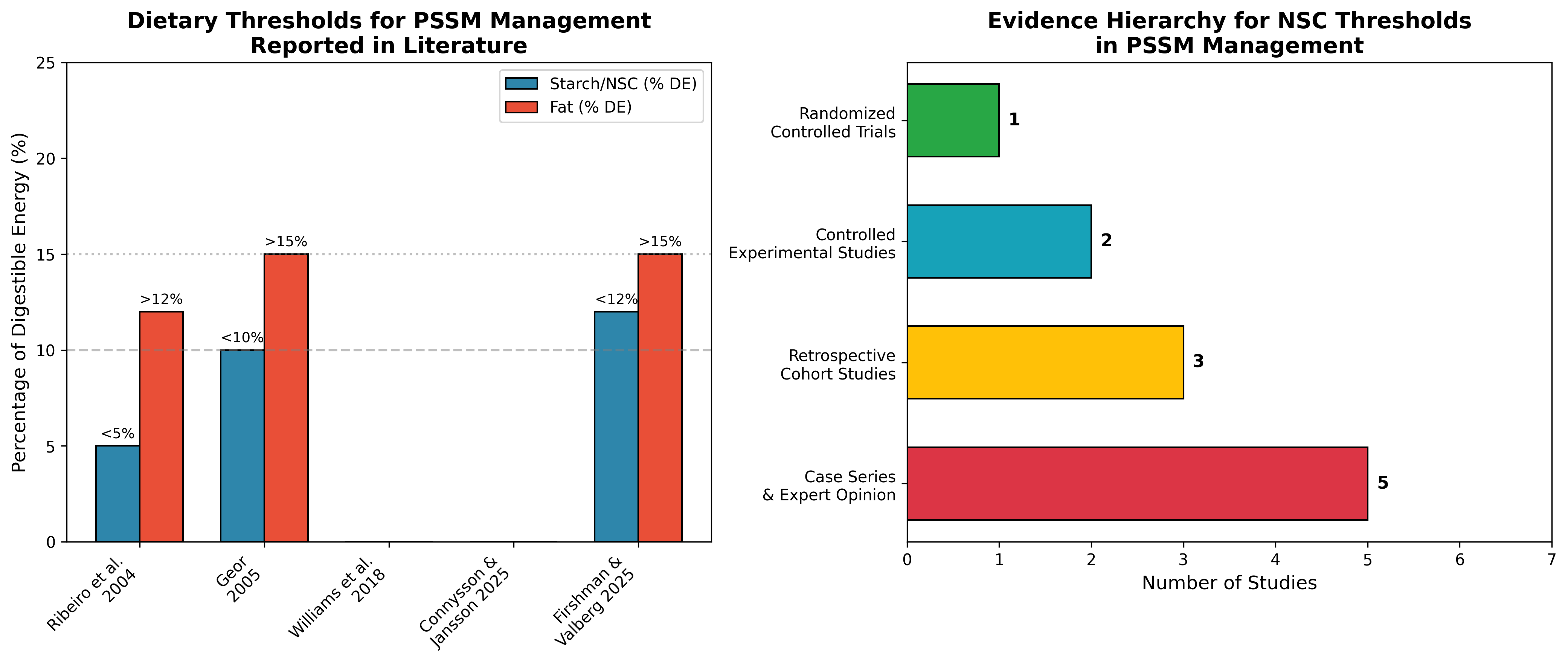
Figure 1. Left panel: Reported starch/NSC and fat thresholds from key studies examining PSSM dietary management. Right panel: Distribution of studies by evidence hierarchy, demonstrating the predominance of observational and consensus-based evidence relative to controlled experimental trials. Data sources: Ribeiro et al. 2004 [6], Geor 2005 [3], Firshman & Valberg 2025 [1].
Figure 2: Clinical Response Rates by PSSM Subtype
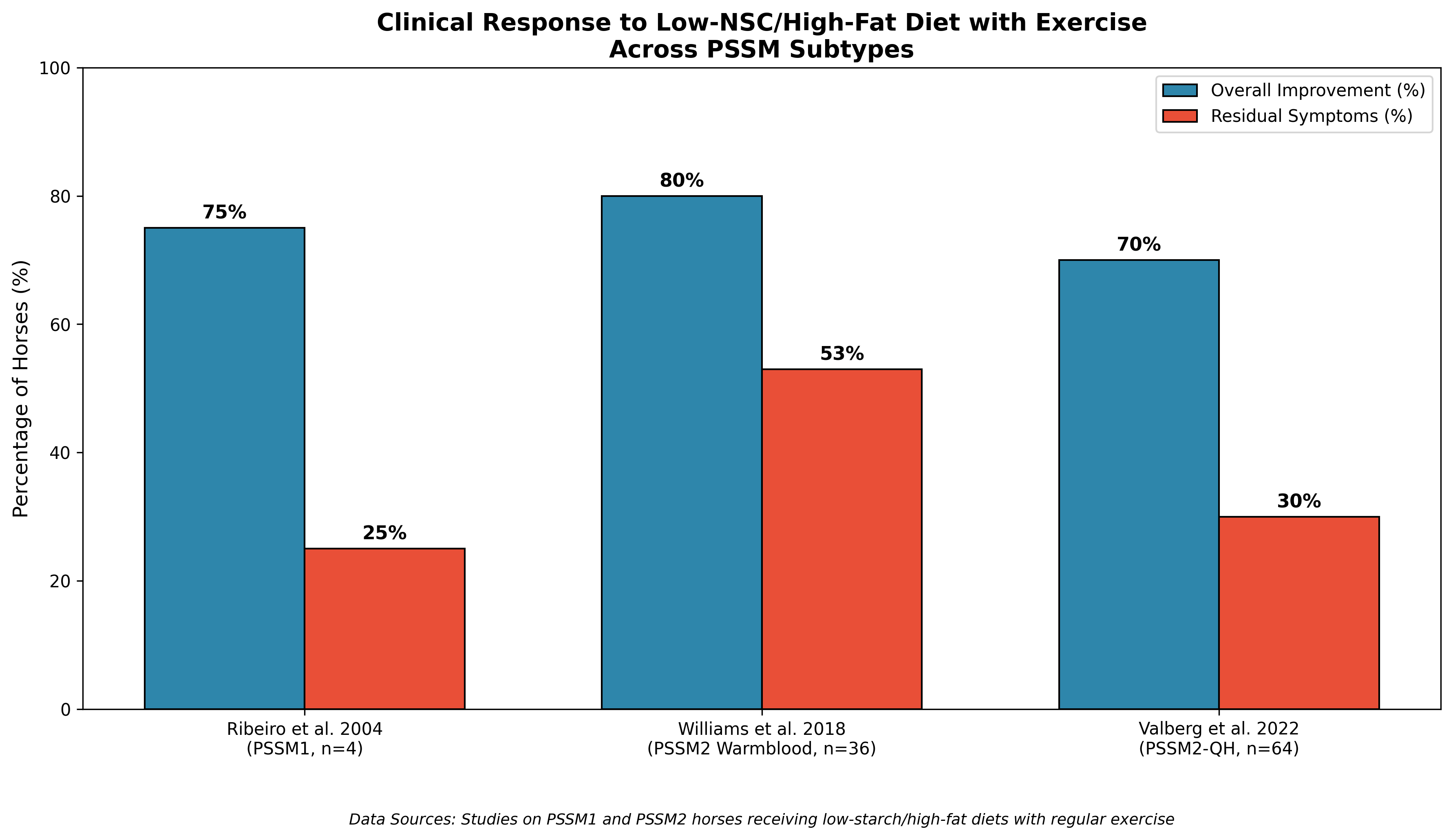
Figure 2. Clinical response to low-NSC/high-fat dietary management with exercise across PSSM subtypes. Overall improvement was reported in the majority of cases across subtypes; however, residual symptoms persisted in a substantial proportion of horses, particularly in PSSM2 Warmblood horses. Data derived from Williams et al. 2018 [8], Valberg et al. 2022 [9], and Ribeiro et al. 2004 [6].
---
7. Discussion
7.1 Evidence Synthesis and Critical Appraisal
The evidence base supporting specific NSC thresholds for PSSM management is limited in both quantity and quality. Only one controlled experimental study (Ribeiro et al. 2004) has directly compared multiple starch levels with clinical and biochemical outcomes, finding that <5% DE from starch combined with >12% DE from fat reduced exertional rhabdomyolysis episodes [6]. The broader recommendation of <10-15% DE from hydrolyzable carbohydrates represents expert consensus extrapolated from this limited experimental data, physiological principles, and clinical experience [3].
The relationship between starch intake and muscle enzyme activity is supported by evidence from healthy Standardbred trotters, where post-exercise AST activity was significantly higher in horses receiving high and medium starch allowances compared to low allowances [10]. This supports the physiological plausibility of starch restriction, though direct extrapolation to PSSM-affected horses requires caution.
7.2 Breed-Specific Considerations
PSSM affects diverse breeds with potentially different metabolic profiles. The GYS1 mutation causing PSSM1 is present in many breeds including the Noriker and Haflinger horses, with research demonstrating that the mutation reduces the number of functional muscle fibers detected by electromyography measurements even in the absence of overt clinical signs [13]. The prevalence of disease-causing alleles varies by breed, with four of six muscle disease alleles identified only in Quarter Horse breeds, while the allele causing PSSM1 was detected in other breeds [14].
7.3 Role of Exercise in Management
Dietary intervention alone is insufficient for optimal management of PSSM. Both PSSM1 and PSSM2-ER respond well to a low nonstructural carbohydrate, high fat diet combined with regular exercise [1]. The importance of this combined approach was highlighted in the PSSM2 Warmblood study, where despite dietary modification, 53% of horses were still not advancing as expected [8]. The interaction between exercise training and dietary management requires further investigation to optimize protocols.
7.4 Limitations of Current Evidence
Several significant limitations characterize the available evidence:
- Small sample sizes: The primary experimental study validating specific thresholds included only 4 horses [6]
- Heterogeneous populations: Studies include confirmed PSSM1, histopathologic PSSM2, and clinically diagnosed cases without uniform diagnostic criteria
- Variable outcome measures: Clinical response, CK activity, and glycogen concentrations are not consistently reported across studies
- Lack of dose-response data: No studies have systematically evaluated multiple NSC thresholds to identify optimal restriction levels
- Short intervention durations: Most studies do not report long-term outcomes or relapse rates
7.5 Implications for Clinical Practice
Based on current evidence, the following recommendations can be supported:
- PSSM1 (confirmed): Diet providing <5% DE from starch and >12% DE from fat shows experimental support for reducing clinical episodes, though individual variation exists [6]
- PSSM2 (histopathologic): Low-starch/fat-supplemented diets improve clinical signs in 70-80% of horses, though complete resolution is uncommon [8],[9]
- General guidance: Diets with <10-15% DE from hydrolyzable carbohydrates and 15-20% DE from fat represent consensus recommendations applicable across PSSM subtypes [3]
- Combined approach: Regular exercise is essential and should accompany dietary modification for optimal outcomes [1]
---
8. Conclusions
This systematic review identifies a significant gap between clinical practice recommendations and experimental validation for NSC thresholds in PSSM management. The commonly cited recommendation of <10-12% NSC is primarily based on expert consensus, extrapolation from limited experimental data, and clinical experience rather than rigorous dose-finding studies. The single Latin square study demonstrating benefit at <5% DE starch provides the strongest experimental support, but its small sample size and single-breed focus limit generalizability.
Key findings include:
- PSSM1: Enhanced insulin sensitivity and glucose uptake provide metabolic rationale for carbohydrate restriction [4]
- PSSM2: Responds to similar dietary interventions but with lower rates of complete resolution [8]
- Threshold variability: Recommendations range from <5% to <15% DE from starch/NSC, reflecting uncertainty in the evidence base
- Fat supplementation: >12-20% DE from fat is consistently recommended alongside NSC restriction [6],[3]
- Exercise integration: Combined diet and exercise management is superior to diet alone [1]
Future research priorities should include large-scale, multi-breed randomized trials comparing specific NSC thresholds, standardized diagnostic criteria across studies, long-term follow-up including relapse rates, and investigation of breed-specific or genotype-specific responses to dietary intervention. Until such evidence is available, clinicians should apply current recommendations while recognizing their basis in limited experimental data and expert consensus, individualizing management based on clinical response and monitoring of muscle enzyme activity.
References
References
Numbered in order of first citation, IEEE style. Each entry links to the original source via DOI or publisher URL.
- A. Firshman and S. Valberg, “Polysaccharide Storage Myopathy.,” The Veterinary clinics of North America. Equine practice, Jan. 2025. doi: 10.1016/j.cveq.2024.11.004. View source
- C. J. Finno, “Genetics of Muscle Disease.,” The Veterinary clinics of North America. Equine practice, Jan. 2025. doi: 10.1016/j.cveq.2024.10.002. View source
- R. Geor, “Role of dietary energy source in the expression of chronic exertional myopathies in horses,” Journal of Animal Science, Jun. 2005. doi: 10.2527/2005.8313_SUPPLE32X. View source
- F. D. De La Côrte, S. Valberg, J. MacLeay, S. Williamson, and J. R. Mickelson, “Glucose uptake in horses with polysaccharide storage myopathy.,” American Journal of Veterinary Research, Apr. 1999. doi: 10.2460/ajvr.1999.60.04.458. View source
- E. Barrey, E. Mucher, N. Jeansoule, T. Larcher, L. Guigand, B. Herszberg, S. Chaffaux, G. Guérin, X. Mata, P. Benech, M. Canale, O. Alibert, P. Maltere, and X. Gidrol, “Gene expression profiling in equine polysaccharide storage myopathy revealed inflammation, glycogenesis inhibition, hypoxia and mitochondrial dysfunctions,” BMC Veterinary Research, Aug. 2009. doi: 10.1186/1746-6148-5-29. View source
- W. P. Ribeiro, S. Valberg, J. D. Pagan, and B. Gustavsson, “The effect of varying dietary starch and fat content on serum creatine kinase activity and substrate availability in equine polysaccharide storage myopathy.,” Journal of Veterinary Internal Medicine, Nov. 2004. doi: 10.1111/j.1939-1676.2004.tb02637.x. View source
- R. Naylor, L. Livesey, J. Schumacher, N. Henke, C. Massey, K. Brock, M. Fernández-Fuente, and R. Piercy, “Allele Copy Number and Underlying Pathology Are Associated with Subclinical Severity in Equine Type 1 Polysaccharide Storage Myopathy (PSSM1),” PLoS ONE, Jul. 2012. doi: 10.1371/journal.pone.0042317. View source
- Z. J. Williams, M. Bertels, and S. Valberg, “Muscle glycogen concentrations and response to diet and exercise regimes in Warmblood horses with type 2 Polysaccharide Storage Myopathy,” PLoS ONE, Sep. 2018. doi: 10.1371/journal.pone.0203467. View source
- S. Valberg, Z. J. Williams, C. Finno, A. Schultz, D. Velez-Irizarry, M. Henry, K. Gardner, and J. Petersen, “Type 2 polysaccharide storage myopathy in Quarter Horses is a novel glycogen storage disease causing exertional rhabdomyolysis.,” Equine Veterinary Journal, Aug. 2022. doi: 10.1111/evj.13876. View source
- M. Connysson and A. Jansson, “Starch Allowance and Muscle Enzyme Activity in Healthy Standardbred Trotters Trained by Professional Trainers,” Journal of animal physiology and animal nutrition, May 2025. doi: 10.1111/jpn.14127. View source
- K. Urschel and E. McKenzie, “Nutritional Influences on Skeletal Muscle and Muscular Disease.,” The Veterinary clinics of North America. Equine practice, Apr. 2021. doi: 10.1016/j.cveq.2020.12.005. View source
- S. Ghajar, H. Mckenzie, J. Fike, B. McIntosh, and B. F. Tracy, “Evaluating digestibility and toxicity of native warm-season grasses for equines,” Translational Animal Science, Dec. 2020. doi: 10.1093/tas/txaa224. View source
- R. Zsoldos, N. Khayatzadeh, J. Soelkner, U. Schroeder, C. Hahn, and T. Licka, “Comparison of gluteus medius muscle activity in Haflinger and Noriker horses with polysaccharide storage myopathy,” Journal of animal physiology and animal nutrition, Feb. 2021. doi: 10.1111/jpn.13504. View source
- M. Aleman, R. Scalco, J. Malvick, R. Grahn, A. True, and R. R. Bellone, “Prevalence of genetic mutations in horses with muscle disease from a neuromuscular disease laboratory.,” Journal of Equine Veterinary Science, Sep. 2022. doi: 10.2139/ssrn.4100989. View source
This review is one of 34 in the Foxwatch research corpus. Methodology: PRISMA 2020-style systematic literature review against PubMed, CAB Abstracts, Scopus, Web of Science, and equine veterinary conference proceedings.