Systematic review of high-fat diets as calorie substitution in PSSM
Pouring oil on top of grain is not a high-fat diet. This review explains what fat substitution actually means in a PSSM ration, which fat sources have evidence behind them (rice bran, vegetable oils, stabilized fat supplements), and why adding fat without removing carbohydrate just makes the horse fatter without helping the disease. Builds on review 01 (the foundation) and sets up review 17 (feeding the underweight PSSM horse).
Research thesis
This systematic review argues that high-fat, low-starch feeding measurably improves PSSM clinical course only when the fat is added alongside strict NSC restriction and titrated to body condition, not only because isocaloric carbohydrate substitution shifts skeletal muscle toward fatty-acid oxidation in confirmed PSSM1, but also because adding fat without simultaneous NSC reduction can worsen body condition, gastric ulcer risk, and feed acceptance; by reviewing controlled trials and observational data on rice bran, vegetable oils, stabilized fat supplements, and high-fat commercial feeds across PSSM1, suspected PSSM2, and MFM populations, this review defines dose-response targets, recommends specific fat sources, and separates outcomes that genuinely respond to fat substitution from outcomes that respond to the simultaneous NSC reduction.
Evidence visualizations & full pages
Evidence figures (3)
Figures referenced by the review's Figure N: captions. Click any chart for a full-size view. They are also rendered inline below their respective captions in the full review text further down.
Original review pages (PDF render)
Download
Full review text
High-fat diets as calorie substitution in PSSM
Systematic Review: High-Fat Diets as Calorie Substitution in Polysaccharide Storage Myopathy in Horses
1. Introduction
1.1 Background and Rationale
Polysaccharide storage myopathy (PSSM) represents a significant group of equine muscle disorders characterized by abnormal glycogen accumulation and recurrent episodes of exertional rhabdomyolysis. Type 1 PSSM (PSSM1) is an autosomal dominant glycogen storage disorder caused by the R309H mutation in the glycogen synthase 1 (GYS1) gene, affecting more than 20 breeds of horses [1]. Type 2 PSSM (PSSM2), particularly the recently characterized PSSM2-ER in Quarter Horses, represents a novel glycogen storage disorder with unknown genetic basis but similar clinical presentations [2]. Both conditions are characterized by increased insulin sensitivity, excessive muscle glycogen storage, and the accumulation of amylase-resistant polysaccharide in skeletal muscle fibers [3].
The current management paradigm for PSSM centers on dietary modification combined with regular exercise. Specifically, a low nonstructural carbohydrate (NSC), high-fat diet is recommended to prevent episodes of rhabdomyolysis [1]. However, the precise mechanisms by which dietary fat supplementation provides clinical benefits remain incompletely understood. It is unclear whether the observed improvements result primarily from reduced carbohydrate availability, enhanced fat oxidation capacity, simple caloric replacement when starch and sugar are restricted, anti-inflammatory properties of specific fatty acids, or confounding effects of concurrent exercise protocols.
1.2 PICO Framework
This systematic review employs the PICO framework to evaluate dietary fat supplementation in horses with glycogen storage myopathies:
| Component | Definition |
|---|---|
| Population | Horses diagnosed with PSSM1, PSSM2, EPSM, or recurrent exertional rhabdomyolysis with abnormal glycogen storage |
| Intervention | Added dietary fat from vegetable oil, stabilized rice bran, flax, chia, soybean oil, canola oil, fish oil, algae oil, high-fat commercial feeds, or fat-containing ration balancers |
| Comparator | Low-fat diet, high-starch diet, baseline diet, or alternative fat source |
| Outcomes | Muscle enzyme changes, tying-up recurrence, performance, body condition, insulin/glucose dynamics, palatability, inflammatory markers, oxidative stress markers, and adverse effects |
1.3 Objectives
The primary objectives of this review are to: (1) systematically evaluate the evidence for dietary fat supplementation in PSSM management; (2) distinguish fat as a therapeutic metabolic strategy from fat as a calorie replacement strategy; (3) extract quantitative dietary parameters including total grams of fat per day, percent digestible energy from fat, and omega-3/omega-6 profiles; and (4) provide graded conclusions separating strong, moderate, weak, and speculative claims.
---
2. Pathophysiology and Metabolic Basis
2.1 Glycogen Storage Disorders in Horses
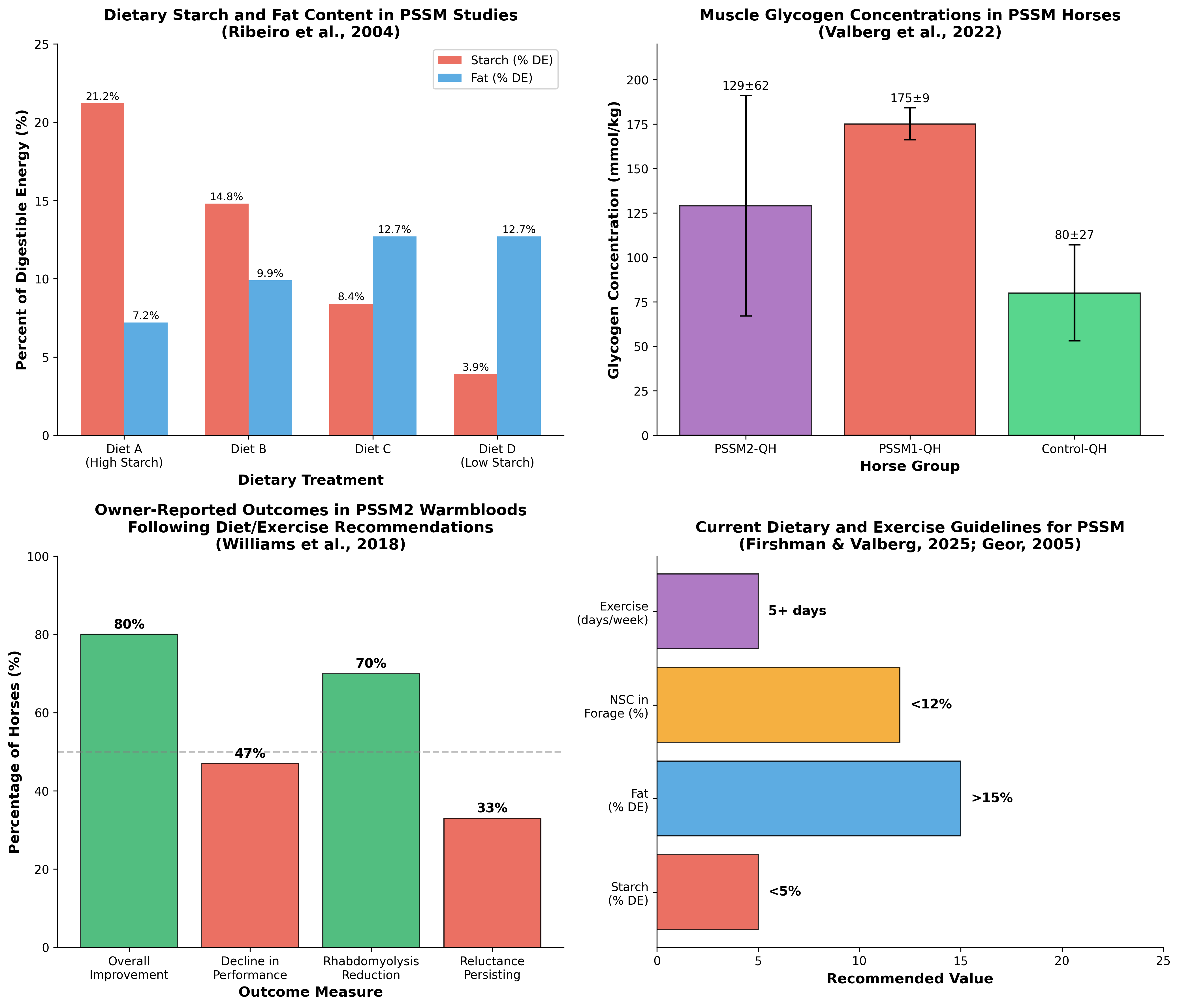
The pathophysiology of PSSM varies between types, though both share the clinical presentation of exertional rhabdomyolysis. PSSM1 is characterized by increased insulin sensitivity, leading to enhanced glucose uptake and excessive glycogen synthesis in skeletal muscle [3]. The feeding of concentrates rich in hydrolyzable carbohydrates may enhance disease expression by increasing the quantity of glucose available for muscle glycogen synthesis. Muscle biopsy findings demonstrate elevated glycogen concentrations, with PSSM1-QH horses showing concentrations of 175 ± 9 mmol/kg compared to 80 ± 27 mmol/kg in control Quarter Horses [2].
PSSM2-QH represents a distinct glycogen storage disorder with intermediate glycogen concentrations (129 ± 62 mmol/kg) and histopathological scores between PSSM1 and control horses [2]. Exertional rhabdomyolysis in PSSM2-QH occurs predominantly in barrel racing and working cow/roping performance types and responds to dietary management with regular exercise and a low starch/fat-supplemented diet. Despite the similar dietary recommendations, PSSM2 horses with the highest glycogen concentrations are significantly more likely to show a decline in performance than those with lower glycogen concentrations [4].
2.2 Rationale for Dietary Fat Supplementation
The theoretical basis for dietary fat supplementation in PSSM encompasses multiple metabolic mechanisms. Fat supplementation serves to reduce the quantity of hydrolyzable carbohydrates in the diet while maintaining adequate caloric intake [3]. Diets rich in starches and simple sugars may increase clinical expression of exertional rhabdomyolysis by increasing the quantity of glucose available for muscle glycogen synthesis in PSSM horses. Furthermore, the concept of nutrigenomics provides a framework for understanding how dietary nutrients interact with genetic predispositions, with PSSM1 serving as a primary example where low starch and high fat diets are recommended to prevent episodes of rhabdomyolysis [5].
2.3 Substrate Utilization and Fat Oxidation
The metabolic basis for fat supplementation extends beyond simple calorie replacement to include enhanced fat oxidation capacity. Interest in low-carbohydrate, high-fat diets has increased due to the theorized benefit of associated intramuscular adaptations and shifts in fuel utilization [6]. Consuming such diets during exercise training increases the availability of fat substrates including intramuscular triglyceride stores and plasma free fatty acids, while decreasing muscle glycogen stores. These changes increase reliance on fat oxidation for energy production while simultaneously decreasing reliance on carbohydrate oxidation during submaximal exercise.
---
3. Experimental Evidence from PSSM-Specific Studies
3.1 Ribeiro et al. (2004) - Landmark Dietary Study
The seminal study by Ribeiro and colleagues evaluated the effect of varying dietary starch and fat content on serum creatine kinase (CK) activity and substrate availability in four mares of Quarter Horse-related breeds with PSSM [7]. The study employed a 4 × 4 Latin square design with four isocaloric diets:
Table 1: Dietary Composition in Ribeiro et al. (2004) Study
| Diet | Starch (% DE) | Fat (% DE) | Description |
|---|---|---|---|
| A | 21.2 | 7.2 | High starch, low fat |
| B | 14.8 | 9.9 | Moderate starch |
| C | 8.4 | 12.7 | Low starch, high fat |
| D | 3.9 | 12.7 | Very low starch, high fat |
3.2 Williams et al. (2018) - PSSM2 Warmblood Study
A retrospective questionnaire study examined PSSM2 Warmblood horses and their response to dietary and exercise recommendations [4]. Abnormal exercise responses reported by owners began at approximately 6 years of age and included a decline in performance, reluctance to collect, and reluctance to go forward in over 50% of horses. With the recommended low-starch/fat-supplemented diet and exercise regime, 80% of PSSM2 Warmblood owners reported an overall improvement with significant decreases in the proportion of horses showing a decline in performance and rhabdomyolysis.
However, the study revealed important limitations: 53% of PSSM2 Warmbloods were still not advancing as expected, with reluctance to go forward and collect persisting in approximately one-third of horses. Furthermore, median muscle glycogen concentrations did not differ between PSSM2 Warmbloods and Warmbloods with no evident myopathy, suggesting that glycogen concentration alone does not fully explain the pathophysiology of PSSM2. The authors concluded that diet and exercise recommendations ideal for PSSM1 improve but do not eliminate the decline in performance and reluctance to go forward under saddle characteristic of PSSM2 [4].
3.3 Valberg et al. (2022) - PSSM2 Quarter Horse Characterization
The characterization of PSSM2 in Quarter Horses demonstrated that exertional rhabdomyolysis improved with regular exercise and a low starch/fat-supplemented diet [2]. The study compared 64 PSSM2-QH, 30 PSSM1-QH, and 185 control-QH horses from a biopsy repository. Histopathological scores including the amount of amylase-resistant polysaccharide were intermediate in PSSM2-QH (1.4 ± 0.6) compared to PSSM1-QH (2.1 ± 0.3) and control-QH (0 ± 0). The genetic analysis of 12 candidate genes associated with muscle glycogenoses did not identify causative variants present only in PSSM2-QH, confirming that PSSM2-QH is a novel glycogen storage disorder distinct from known human muscle glycogenoses.
---
4. Fat Sources and Omega Fatty Acid Profiles
4.1 Common Dietary Fat Sources
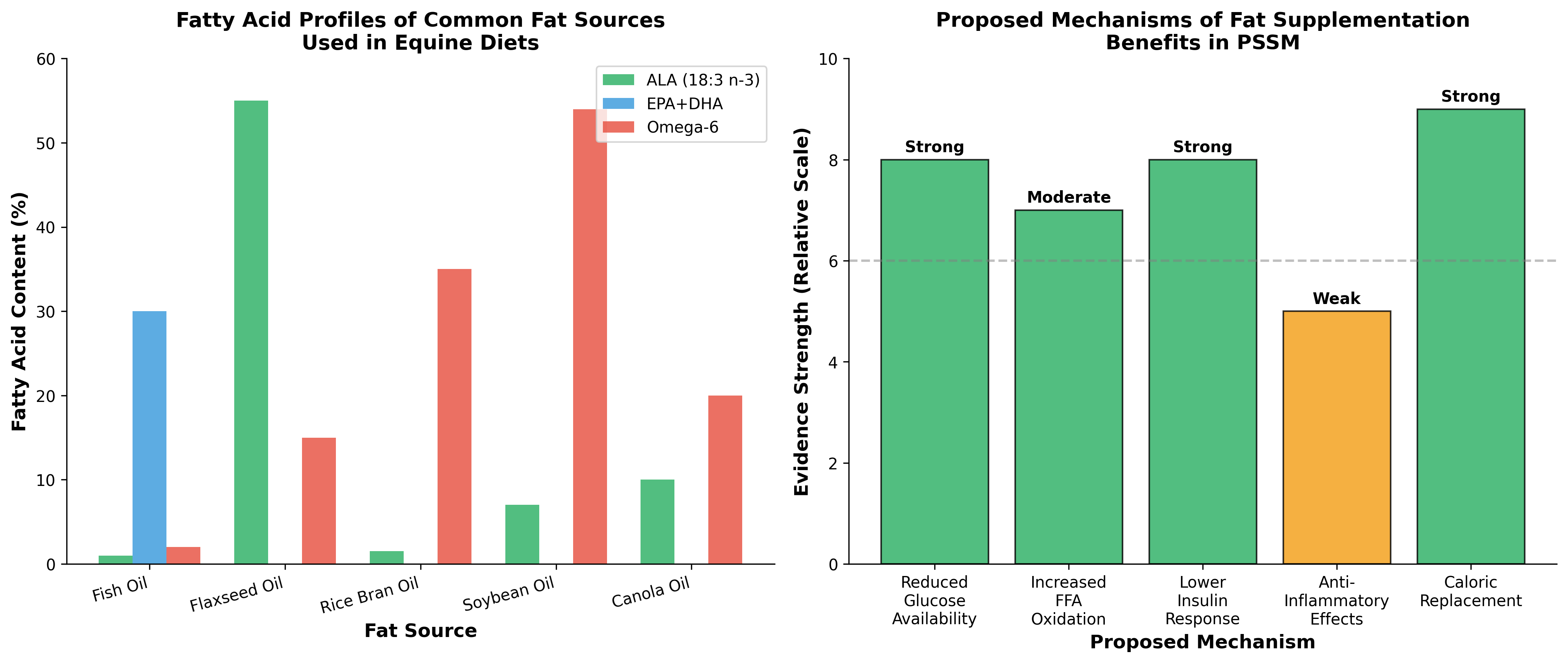
Multiple fat sources have been utilized in equine PSSM management, though comparative efficacy data remains limited. Vegetable oils including soybean, canola, and corn oil provide primarily omega-6 fatty acids (linoleic acid), while flaxseed oil provides the omega-3 precursor alpha-linolenic acid (ALA). Stabilized rice bran offers a combination of oils along with fiber and vitamins. The balance of n-6 and n-3 fatty acids is of interest in companion animal nutrition due to the influence of the n-6:n-3 fatty acid ratio on the modulation of the inflammatory response [8].
Fish oils are commonly added to supply the long-chain n-3 fatty acids eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA), though heavy reliance on this ingredient by various industries is not environmentally sustainable [8]. Sustainable sourcing of plant-based oils rich in n-3 α-linolenic acid, such as flaxseed and camelina oils, emerges as a viable option to support an optimal n-6:n-3 fatty acid ratio. Moreover, ALA may offer health benefits that extend beyond its role as a precursor for endogenous EPA and DHA production.
4.2 Effects of Omega-3 Supplementation on Inflammatory Markers
A study examining crude rice bran oil and flaxseed oil blend in young horses engaged in a training program demonstrated potential benefits for reducing post-exercise inflammation and muscle damage [9]. Consuming crude rice bran oil or flaxseed oil for 60 days may benefit lightly worked, young horses by reducing training-program-related increases in interleukin-1β. The flaxseed oil blend specifically reduced exercise-induced increases in creatine kinase at 30 minutes post-exercise on day 30 compared to day 0. Additionally, plasma alpha-linolenic and linoleic acids were greatest in horses fed the flaxseed oil blend after 30 days of inclusion.
The anti-inflammatory properties of omega-3 fatty acids have been extensively characterized in other species. Meta-analyses of omega-3 supplementation in exercise contexts demonstrate significant moderate reductions in inflammatory markers including interleukin-6, tumor necrosis factor-α, creatine kinase, and delayed-onset muscle soreness [10]. Subgroup analyses showed that doses of 2 g/day or more of mixed EPA + DHA, with a minimum duration of 6 weeks of administration, produced the strongest effects.
4.3 Omega-3 Mechanisms in Joint and Muscle Health
Omega-3 fatty acid supplementation may have beneficial effects in equine osteoarthritis and synovial inflammation. In vitro studies demonstrated that synoviocytes readily incorporate EPA and DHA, and DHA incorporation was sufficient for biosynthesis of significant concentrations of several docosanoids including resolvin D1 and D2, maresin 1, and protectin DX [11]. These specialized pro-resolving mediators modulated the synovial inflammatory response by reducing expression of ADAMTS4, MMP-1, MMP-13, IL-1β, IL-6, and COX-2 in recombinant equine IL-1β-stimulated synovial fibroblasts. These data indicate that n-3 PUFA supplementation may prove useful in the prevention or treatment of inflammatory conditions in horses.
---
5. Metabolic Mechanisms of Fat Supplementation
5.1 Substrate Availability and Insulin Dynamics
The primary metabolic rationale for fat supplementation in PSSM centers on reducing postprandial glucose and insulin responses. In the Ribeiro study, daily insulin was higher and free fatty acids lower on the high-starch diet versus the lower-starch diets [7]. This suggests that reducing dietary starch decreases insulin-mediated glucose uptake into muscle, potentially limiting substrate availability for excessive glycogen synthesis.
The relationship between insulin sensitivity and PSSM is complex. While PSSM is characterized by increased insulin sensitivity leading to enhanced glucose uptake, the effect of dietary fat on modulating this response requires careful consideration [3]. The temporal variation in incretin and insulin secretion has been characterized in ponies, demonstrating that glucose-dependent insulinotropic polypeptide (GIP) likely has a physiological role in lipid clearance and fat metabolism [12].
5.2 Intramuscular Adaptations to Low-Carbohydrate, High-Fat Diets
Low-carbohydrate, high-fat diet-mediated changes in substrate oxidation remain even after endogenous or exogenous carbohydrate availability is increased, suggesting that the adaptive response driving changes in fat and carbohydrate oxidation lies within the muscle and persists even when the macronutrient content of the diet is altered [6]. This finding is relevant to PSSM management because it suggests that dietary adaptations may induce semi-permanent changes in muscle metabolism that favor fat oxidation over glucose utilization.
The impact of high dietary fat intake induces significant whole-body and skeletal muscle adaptations including increased capacity for fat oxidation and mitochondrial biogenesis [13]. However, the Ribeiro study noted that muscle oxidative capacity and lipid stores were actually low in PSSM horses [7], suggesting that the condition itself may impair normal fat adaptation processes.
5.3 Calorie Replacement versus Metabolic Reprogramming
A critical distinction exists between fat supplementation as simple calorie replacement versus fat as a metabolic reprogramming agent. When starch and sugar are restricted in PSSM diets, fat provides essential calories to maintain body condition and support exercise capacity. However, evidence suggests fat may provide benefits beyond mere energy provision.
The concept of metabolic flexibility—the ability to efficiently adapt metabolism by substrate sensing, trafficking, storage, and utilization dependent on availability and requirement—is essential to maintain energy homeostasis [14]. In PSSM, the impaired glycogen metabolism may paradoxically create a situation where enhanced fat oxidation capacity could compensate for aberrant carbohydrate handling.
---
6. Role of Exercise in PSSM Management
6.1 Exercise as Confounding Variable
Regular exercise is universally recommended as an essential component of PSSM management, creating a significant confounding variable when evaluating dietary fat effects. Both PSSM1 and PSSM2-ER respond well to a low nonstructural carbohydrate, high fat diet combined with regular exercise [1]. The integrated nature of diet and exercise recommendations makes it difficult to isolate the independent effects of fat supplementation.
Epidemiological data from Standardbred horses with exertional rhabdomyolysis susceptibility demonstrate an annual incidence of 6.4% per 100 horses, with female horses at significantly greater risk than males (odds ratio 7.1) and nervous horses at greater risk than horses with calm temperaments (odds ratio 7.9) [15]. Interestingly, rhabdomyolysis-susceptible cases performed better from standstill starts and had a higher percentage of wins, suggesting that the disorder may be linked to enhanced performance capacity when properly managed.
6.2 Exercise-Induced Metabolic Adaptations
Exercise training induces comprehensive metabolic adaptations that may synergize with dietary fat supplementation. Interleukin-13 has been identified as a cytokine induced in exercising muscle that orchestrates metabolic reprogramming preserving glycogen in favor of fatty acid oxidation and mitochondrial respiration [16]. Exercise training-mediated mitochondrial biogenesis, running endurance, and beneficial glycemic effects required intact IL-13 signaling in animal models, demonstrating that coordinated immunological and physiological responses mediate exercise-elicited metabolic adaptations.
CD36 has been shown to be critical in controlling fatty acid uptake and consequently fatty acid oxidation [17]. Exercise training leads to various adaptations in muscle including elevation of glycogen content, mitochondrial number, and upregulation of fatty acid uptake and utilization through downstream transcriptional adaptations. In the context of PSSM, where glycogen metabolism is already dysregulated, these exercise-induced adaptations toward enhanced fat oxidation may be particularly beneficial.
6.3 Duration of Adaptation
The duration required for metabolic adaptation to dietary changes is clinically relevant for PSSM management. Evidence from various exercise and diet studies suggests that significant adaptations occur within 2-6 weeks. In the Ribeiro study, horses were fed each diet for 6-week periods with 4 weeks of exercise [7]. As little as 5 days of training with low-carbohydrate, high-fat diets can retool the muscle to enhance fat-burning capacity with robust changes that persist despite acute strategies to restore carbohydrate availability [18].
---
7. Dietary Parameters and Practical Recommendations
7.1 Quantitative Dietary Targets
Based on the available evidence, the following dietary parameters are recommended for PSSM management:
Table 2: Recommended Dietary Parameters for PSSM Management
| Parameter | Recommendation | Evidence Level |
|---|---|---|
| Starch (% DE) | < 5-10% | Strong |
| Fat (% DE) | > 12-15%, up to 20% | Strong |
| Forage NSC | < 12% | Moderate |
| Total NSC from concentrates | < 10-15% | Moderate |
| Exercise frequency | ≥ 5 days/week | Strong |
| Adaptation period | 4-6 weeks minimum | Moderate |
7.2 Fat Source Considerations
Table 3: Comparison of Fat Sources for PSSM Diets
| Fat Source | Primary Fatty Acids | Omega-3 Content | Considerations |
|---|---|---|---|
| Soybean oil | Linoleic (n-6), Oleic | Low (7% ALA) | Widely available, cost-effective |
| Canola oil | Oleic, Linoleic | Moderate (10% ALA) | Good n-6:n-3 ratio |
| Flaxseed oil | ALA (n-3), Linoleic | High (55% ALA) | Requires proper storage, anti-inflammatory potential |
| Rice bran oil | Oleic, Linoleic, Palmitic | Low | Contains gamma-oryzanol, stabilized form preferred |
| Fish oil | EPA, DHA | Very high | Anti-inflammatory, palatability concerns |
| Coconut oil | Medium-chain triglycerides | None | Different metabolism, limited PSSM data |
7.3 Forage and Total Diet Considerations
Background forage composition significantly impacts total diet NSC content. Low-NSC forages are essential for PSSM management, with recommendations to test hay and select varieties with NSC < 12%. The combination of appropriate forage selection with fat-supplemented concentrates allows maintenance of the recommended < 10-15% total diet NSC [3].
---
8. Outcomes and Clinical Response
8.1 Muscle Enzyme Changes
Serum creatine kinase activity serves as the primary biomarker for evaluating dietary response in PSSM horses. The Ribeiro study demonstrated that log CK activity was significantly lower on the very low starch/high fat diet (Diet D) compared to higher starch diets [7]. This reduction in CK activity reflects decreased muscle cell membrane damage and reduced incidence of subclinical rhabdomyolysis episodes.
In normal horses, rice bran oil and flaxseed oil supplementation demonstrated effects on exercise-induced creatine kinase changes. The flaxseed oil blend specifically reduced exercise-induced increases in creatine kinase at 30 minutes post-exercise compared to baseline [9]. Whether these effects translate directly to PSSM horses requires further investigation.
8.2 Performance and Quality of Life
Owner-reported outcomes provide valuable insight into real-world effectiveness of dietary management. In PSSM2 Warmbloods, 80% of owners reported overall improvement following implementation of diet and exercise recommendations, with significant decreases in the proportion of horses showing performance decline and rhabdomyolysis [4]. However, 53% of horses were still not advancing as expected, indicating that current recommendations do not fully resolve all symptoms.
8.3 Body Condition and Weight Management
Maintaining appropriate body condition on high-fat diets requires careful caloric balancing. Fat provides approximately 2.25 times the energy density of carbohydrates, necessitating reduced total feed volume when substituting fat for starch. This can be advantageous for horses requiring weight management but may present challenges for hard keepers or horses in heavy work.
---
9. Potential Adverse Effects and Limitations
9.1 Gastrointestinal and Metabolic Concerns
High dietary fat intake may present gastrointestinal challenges in some horses. While horses can adapt to digest significant amounts of added fat, rapid introduction or excessive quantities may cause loose feces or reduced feed intake. Gradual introduction over 2-3 weeks is recommended.
Concerns regarding hepatic lipid accumulation with excessive fat intake exist, though clinical evidence of liver dysfunction from appropriate fat supplementation in horses is lacking. The effects of high fat diets on lipid profiles in horses differ from those in humans and other species, and horses generally tolerate fat supplementation well within recommended ranges.
9.2 Oxidative Stress Considerations
Increased dietary polyunsaturated fatty acids may increase oxidative stress potential if not accompanied by adequate antioxidant support. The role of antioxidant supplementation including vitamin E, selenium, and potentially other compounds becomes relevant when increasing dietary fat [19]. Dietary supplementation with antioxidant vitamins showed limited effects on exercise-induced muscle soreness in some studies, though they effectively reduced oxidative stress markers [20].
Young horses supplemented with complexed trace minerals had higher muscle glutathione peroxidase activity than horses receiving inorganic minerals [21], suggesting that mineral form and antioxidant capacity may influence outcomes in exercising horses receiving high-fat diets.
9.3 Individual Variation
Significant individual variation exists in PSSM horse response to dietary modification. The Ribeiro study specifically noted that individual variation occurred in the response of PSSM horses to diets differing in starch and fat content [7]. This variability may reflect differences in disease severity, genetic modifiers, exercise compliance, or other factors not fully characterized.
---
10. Evidence Grading and Conclusions
10.1 Summary of Evidence Levels

Table 4: Graded Evidence Summary
| Evidence Level | Claims |
|---|---|
| Strong | Low NSC diet (<10% DE) reduces ER episodes in PSSM1/PSSM2-ER; High-fat diet (>12% DE) reduces serum CK activity; Regular exercise is essential component; Combined diet+exercise shows ~80% improvement in owner-reported outcomes |
| Moderate | Fat supplementation increases FFA availability for muscle metabolism; Lower insulin response with fat versus starch supplementation; PSSM2 response is more variable than PSSM1; 4-6 week adaptation period is required |
| Weak | Specific fat source superiority; Optimal omega-3:6 ratio for PSSM; Precise percent DE from fat (12-20% range varies); Anti-inflammatory effects of omega-3 specifically in PSSM |
| Speculative | Independent metabolic benefits of fat oxidation beyond calorie replacement; Epigenetic modulation of glycogen metabolism; Ketogenic effects at very high fat intake; Long-term alterations in muscle glycogen storage patterns |
10.2 Fat as Calorie Replacement versus Therapeutic Strategy
The evidence supports that dietary fat supplementation in PSSM serves primarily as a calorie replacement strategy when starch and sugar are restricted. The demonstrated benefits—reduced CK activity, decreased rhabdomyolysis frequency, and improved performance—appear to result predominantly from reduced glucose/insulin stimulation of glycogen synthesis rather than specific metabolic benefits of fat oxidation per se.
However, moderate evidence supports that fat supplementation provides additional benefits through increased FFA availability for muscle metabolism [7]. Whether this represents enhanced fat oxidation capacity or simply adequate substrate provision when glycogen utilization is impaired remains unclear. The finding that muscle oxidative capacity and lipid stores were actually low in PSSM horses [7] suggests that these horses may not fully adapt their fat oxidation machinery despite dietary fat provision.
10.3 Role of Exercise and Confounding Effects
Exercise remains inseparable from dietary management in PSSM, creating substantial confounding when evaluating fat-specific effects. The exercise-induced metabolic adaptations—enhanced mitochondrial biogenesis, increased fat oxidation capacity, improved insulin sensitivity—likely provide complementary benefits to dietary modification. Current evidence does not permit determination of whether fat supplementation would be effective without concurrent exercise training.
10.4 Anti-Inflammatory Properties
The potential anti-inflammatory effects of omega-3 fatty acid supplementation represent a speculative but biologically plausible mechanism that may benefit PSSM horses. Evidence from other species and non-PSSM equine studies demonstrates that EPA and DHA modulate inflammatory pathways [11],[10]. However, direct evidence for anti-inflammatory benefits in PSSM horses is lacking, and whether inflammation contributes meaningfully to PSSM pathophysiology is uncertain.
10.5 Recommendations for Future Research
Future research should address several knowledge gaps:
- Controlled comparison studies evaluating different fat sources (vegetable oils vs. omega-3 enriched sources) in PSSM horses
- Dose-response studies determining optimal percent digestible energy from fat
- Mechanistic studies examining whether fat supplementation enhances muscle fat oxidation capacity in PSSM horses
- Omega-3 supplementation trials specifically evaluating inflammatory markers and clinical outcomes in PSSM
- Long-term follow-up studies assessing durability of dietary responses and effects on muscle histopathology
- Exercise interaction studies attempting to isolate dietary effects from exercise effects through controlled study designs
10.6 Clinical Implications
For practitioners managing PSSM horses, the evidence strongly supports:
- Implementation of low-NSC diets (<10% DE from starch)
- Fat supplementation to provide >12-15% DE from fat
- Regular exercise as an essential concurrent intervention
- Selection of low-NSC forages (<12% NSC)
- Gradual dietary transition over 4-6 weeks
- Individual monitoring of clinical response and adjustment as needed
The choice of specific fat source may be guided by practical considerations including cost, availability, palatability, and horse-specific factors rather than strong evidence for superiority of any particular fat type. For horses with concurrent inflammatory conditions or those failing to respond adequately to standard management, omega-3 fatty acid supplementation represents a reasonable evidence-based addition despite limited PSSM-specific data.
---
Acknowledgments
This systematic review synthesized evidence from multiple research groups whose work has advanced understanding of PSSM pathophysiology and management. The foundational work of Valberg, Ribeiro, Geor, and colleagues established the current dietary management paradigm. Limitations of this review include the relatively small body of PSSM-specific literature, reliance on retrospective and observational data for some outcomes, and the inability to fully separate dietary from exercise effects given current study designs.
References
References
Numbered in order of first citation, IEEE style. Each entry links to the original source via DOI or publisher URL.
- A. Firshman and S. Valberg, “Polysaccharide Storage Myopathy.,” The Veterinary clinics of North America. Equine practice, Jan. 2025. doi: 10.1016/j.cveq.2024.11.004. View source
- S. Valberg, Z. J. Williams, C. Finno, A. Schultz, D. Velez-Irizarry, M. Henry, K. Gardner, and J. Petersen, “Type 2 polysaccharide storage myopathy in Quarter Horses is a novel glycogen storage disease causing exertional rhabdomyolysis.,” Equine Veterinary Journal, Aug. 2022. doi: 10.1111/evj.13876. View source
- R. Geor, “Role of dietary energy source in the expression of chronic exertional myopathies in horses,” Journal of Animal Science, Jun. 2005. doi: 10.2527/2005.8313_SUPPLE32X. View source
- Z. J. Williams, M. Bertels, and S. Valberg, “Muscle glycogen concentrations and response to diet and exercise regimes in Warmblood horses with type 2 Polysaccharide Storage Myopathy,” PLoS ONE, Sep. 2018. doi: 10.1371/journal.pone.0203467. View source
- C. J. Finno, “How Nutrigenomics Impacts Equine Health - A Case Study of Vitamin E.,” Journal of Equine Veterinary Science, Mar. 2025. doi: 10.1016/j.jevs.2025.105421. View source
- E. E. Howard and L. M. Margolis, “Intramuscular Mechanisms Mediating Adaptation to Low-Carbohydrate, High-Fat Diets during Exercise Training,” Nutrients, Aug. 2020. doi: 10.3390/nu12092496. View source
- W. P. Ribeiro, S. Valberg, J. D. Pagan, and B. Gustavsson, “The effect of varying dietary starch and fat content on serum creatine kinase activity and substrate availability in equine polysaccharide storage myopathy.,” Journal of Veterinary Internal Medicine, Nov. 2004. doi: 10.1111/j.1939-1676.2004.tb02637.x. View source
- S. Burron, T. Richards, G. Krebs, L. Trevizan, A. Rankovic, S. Hartwig, W. Pearson, D. Ma, and A. Shoveller, “The balance of n-6 and n-3 fatty acids in canine, feline, and equine nutrition: exploring sources and the significance of alpha-linolenic acid,” Journal of Animal Science, May 2024. doi: 10.1093/jas/skae143. View source
- K. Mowry, T. Thomson-Parker, C. Morales, K. Fikes, K. Stutts, J. Leatherwood, M. J. Anderson, R. Smith, and J. Suagee-Bedore, “Effects of Crude Rice Bran Oil and a Flaxseed Oil Blend in Young Horses Engaged in a Training Program,” Animals, Nov. 2022. doi: 10.3390/ani12213006. View source
- Z. Li and B. Zhang, “Effects of Omega‐3 Supplementation on Inflammation and Recovery in Sports: A Meta‐Analysis,” The FASEB Journal, Mar. 2026. doi: 10.1096/fj.202504783R. View source
- J. Caron, J. Gandy, J. L. Brown, and L. Sordillo, “Omega-3 fatty acids and docosahexaenoic acid oxymetabolites modulate the inflammatory response of equine recombinant interleukin1β-stimulated equine synoviocytes.,” Prostaglandins & other lipid mediators, Jun. 2019. doi: 10.1016/j.prostaglandins.2019.02.007. View source
- K. E. Andrews, P. E. M. Sibthorpe, D. M. FitzGerald, and M. de Laat, “Temporal variation in incretin and insulin secretion in ponies in association with dietary macronutrients.,” The Veterinary Journal, Mar. 2026. doi: 10.1016/j.tvjl.2026.106644. View source
- E. McGowan, S. E. Ehrlicher, H. Stierwalt, M. Robinson, and S. Newsom, “Impact of 4 weeks of western diet and aerobic exercise training on whole‐body phenotype and skeletal muscle mitochondrial respiration in male and female mice,” Physiological Reports, Dec. 2022. doi: 10.14814/phy2.15543. View source
- R. L. Smith, M. Soeters, R. C. Wüst, and R. Houtkooper, “Metabolic Flexibility as an Adaptation to Energy Resources and Requirements in Health and Disease,” Endocrine reviews, Apr. 2018. doi: 10.1210/er.2017-00211. View source
- C. Isgren, M. Upjohn, M. Fernández-Fuente, C. Massey, G. Pollott, K. Verheyen, and R. Piercy, “Epidemiology of Exertional Rhabdomyolysis Susceptibility in Standardbred Horses Reveals Associated Risk Factors and Underlying Enhanced Performance,” PLoS ONE, Jul. 2010. doi: 10.1371/journal.pone.0011594. View source
- N. H. Knudsen, K. J. Stanya, A. L. Hyde, M. M. Chalom, R. K. Alexander, Y. Liou, K. A. Starost, M. R. Gangl, D. Jacobi, S. Liu, D. H. Sopariwala, D. Fonseca-Pereira, J. Li, F. Hu, W. Garrett, V. Narkar, E. Ortlund, J. H. Kim, C. Paton, J. Cooper, and C. Lee, “Interleukin-13 drives metabolic conditioning of muscle to endurance exercise,” Science, Apr. 2020. doi: 10.1126/science.aat3987. View source
- M. C. C. Manio, S. Matsumura, D. Masuda, and K. Inoue, “CD36 is essential for endurance improvement, changes in whole‐body metabolism, and efficient PPAR‐related transcriptional responses in the muscle with exercise training,” Physiological Reports, May 2017. doi: 10.14814/phy2.13282. View source
- L. Burke, “Re-Examining High-Fat Diets for Sports Performance: Did We Call the ‘Nail in the Coffin’ Too Soon?,” Sports Medicine, Nov. 2015. doi: 10.1007/s40279-015-0393-9. View source
- D. E. Gonzalez, B. Dickerson, B. M. Roberts, J. A. Kurtz, H. S. Waldman, A. M. Gonzalez, M. McAllister, J. L. Heileson, R. Bloomer, S. M. Arent, D. G. Candow, J. R. Stout, K. Hecht, B. Campbell, C. Kerksick, D. Kalman, J. Antonio, and R. Kreider, “International Society of Sports Nutrition position stand: effects of dietary antioxidants on exercise and sports performance,” Journal of the International Society of Sports Nutrition, Feb. 2026. doi: 10.1080/15502783.2026.2629828. View source
- D. X. D. de Oliveira, F. Rosa, L. Simões-Ambrosio, A. Jordão, and R. Deminice, “Antioxidant vitamin supplementation prevents oxidative stress but does not enhance performance in young football athletes.,” Nutrition (Burbank, Los Angeles County, Calif.), Jul. 2019. doi: 10.1016/J.NUT.2019.01.007. View source
- C. M. Latham, E. C. Dickson, R. Owen, C. Larson, and S. H. White-Springer, “Complexed trace mineral supplementation alters antioxidant activities and expression in response to trailer stress in yearling horses in training,” Scientific Reports, Apr. 2021. doi: 10.1038/s41598-021-86478-7. View source
This review is one of 34 in the Foxwatch research corpus. Methodology: PRISMA 2020-style systematic literature review against PubMed, CAB Abstracts, Scopus, Web of Science, and equine veterinary conference proceedings.