Evidence map of PSSM2, MFM, and nutrition
If your horse keeps tying up but the GYS1 test came back negative, the answer is probably here. PSSM Type 2 and myofibrillar myopathy are different diseases from Type 1, with different causes, different muscle biology, and different feeding needs. A low-sugar diet alone often does not solve them. This review also walks through which "PSSM2 polygenic panels" actually have peer-reviewed validation, and which are still scientifically controversial. Read with reviews 05 (amino acids), 23 (trace minerals), and 28 (genetic testing).
Research thesis
This scoping review and evidence map argues that PSSM Type 2 and myofibrillar myopathy are clinically and metabolically separate conditions from PSSM Type 1, and that feeding recommendations carried over from PSSM1 are often wrong for these horses, not only because the genetic basis of PSSM2 and MFM remains uncharacterized despite commercial polygenic test panels, but also because amino acid demand, oxidative stress response, and fat tolerance differ measurably between Type 1 and non-GYS1 muscle disease; by applying the JBI Population, Concept, and Context framework across biopsy-confirmed MFM, clinically suspected PSSM2, genetic-panel-only diagnoses, and owner-reported cases, this review maps the evidence base for nutrition and supplementation in non-GYS1 equine muscle disorders and separates direct trial evidence from extrapolation and anecdote.
Evidence visualizations & full pages
Evidence figures (2)
Figures referenced by the review's Figure N: captions. Click any chart for a full-size view. They are also rendered inline below their respective captions in the full review text further down.
Original review pages (PDF render)
Download
Full review text
PSSM2, MFM, and nutrition
Scoping Review and Evidence Map: Nutritional Interventions for PSSM2, Myofibrillar Myopathy, and Non-GYS1 Equine Muscle Disorders
Executive Summary
This scoping review applies the JBI Population–Concept–Context (PCC) framework to map nutritional interventions proposed for type 2 polysaccharide storage myopathy (PSSM2), myofibrillar myopathy (MFM), and related non-GYS1 equine muscle disorders. The evidence base for these conditions remains substantially limited compared to PSSM1 (GYS1-associated), with most nutritional recommendations extrapolated from studies on PSSM1 or general equine rhabdomyolysis rather than directly evaluated in biopsy-confirmed PSSM2 or MFM populations.
---
1. Background and Rationale
1.1 Definition and Pathophysiology of PSSM2 and MFM
PSSM2 and MFM represent distinct but overlapping categories of equine muscle disease characterized by histopathological abnormalities without the GYS1 R309H mutation that defines PSSM1. Both PSSM1 and PSSM2 are characterized by aggregates of abnormal polysaccharide in skeletal muscle; however, the cause of PSSM2 in Quarter Horses remains unknown, and glycogen concentrations are intermediate between PSSM1 and control horses [1]. Exertional rhabdomyolysis in PSSM2 Quarter Horses occurs predominantly in barrel racing and working cow/roping performance types and has been reported to improve with regular exercise and a low starch/fat-supplemented diet, though controlled trials specific to PSSM2 are limited [1].
Myofibrillar myopathy in horses is a late-onset disease of unknown origin characterized by poor performance, atrophy, myofibrillar disarray, and desmin aggregation in skeletal muscle [2]. Integrated proteomic and transcriptomic profiling has identified aberrant gene and protein expression in the sarcomere, mitochondrial complex I, and the extracellular matrix in Warmblood horses with MFM, suggesting that aberrant Z-disc mechano-signaling, impaired Z-disc stability, decreased mitochondrial complex I expression, and a pro-oxidative cellular environment contribute to the development of MFM [2]. The molecular signatures identified include significantly elevated expression of CSRP3, PDLIM3, SYNPO2, and the glutathione-degrading gene CHAC1, which is associated with oxidative stress and apoptosis.
1.2 Diagnostic Categories and Challenges
A critical issue in evaluating nutritional interventions is the diagnostic certainty of PSSM2 and MFM. Muscle conditions affecting sport horses include polysaccharide storage myopathies, recurrent exertional rhabdomyolysis, malignant hyperthermia, and myofibrillar myopathy, all requiring different diagnostic approaches [3]. Horses are particularly susceptible to developing exertional rhabdomyolysis characterized by muscle stiffness, pain, and reluctance to move, with diagnosis requiring establishing abnormal increases in serum creatine kinase activity when horses exhibit clinical signs [4]. The two main categories include sporadic ER arising from extrinsic causes and chronic ER arising from intrinsic continuous or episodic abnormalities in muscle function.
Commercial genetic tests for PSSM2 and MFM have not been validated by peer-review, and formal regulation of veterinary genetic testing is lacking [5]. A critical study comparing genotype and allele frequencies of commercial test variants (P2, P3, P4) in MYOT, FLNC, and MYOZ3 genes between horses diagnosed with PSSM2/MFM by muscle histopathology and breed-matched controls found no significant association between any P locus and a histopathological diagnosis. For all tests, sensitivity was less than 0.33, and all P variants were present in early domestic horses (400- to 5500-year-old) with P2 present even in the Przewalski [5]. This lack of association means that these variant genotypes cannot be recommended for selection and breeding, prepurchase examination, or diagnosis of a myopathy.
1.3 Search Strategy and JBI Framework Application
This review follows the JBI PCC framework with the following parameters:
- Population: Horses diagnosed or suspected with PSSM2, MFM, or non-GYS1 myopathy
- Concept: Nutrition, supplementation, and feeding management
- Context: Clinical, performance, breeding, rehabilitation, and owner-managed settings
---
2. Low Starch and High Fat Dietary Interventions
2.1 Evidence from PSSM Studies
The most robust evidence for dietary intervention comes from studies on polysaccharide storage myopathy, though predominantly in PSSM1 populations with extrapolation to PSSM2. A controlled study evaluating the effect of dietary starch and fat content on serum creatine kinase (CK) activity and substrate availability in four mares with PSSM found that four isocaloric diets ranging in digestible energy from 21.2% to 3.9% for starch, and 7.2% to 12.7% for fat demonstrated significant effects [6]. Log CK activity was higher with diets containing higher starch (>5% DE) versus the lowest starch diet (<5% DE). For horses with clinical manifestations of PSSM, a diet with <5% DE starch and >12% DE fat can reduce exertional rhabdomyolysis, potentially by increasing availability of free fatty acids for muscle metabolism.
In horses, nutrigenomics examples include Hyperkalemic Periodic Paralysis (HYPP), where clinical signs are managed through maintaining a diet low in potassium, and Polysaccharide Storage Myopathy Type 1 (PSSM1), where low starch and high fat diets are recommended to prevent episodes of rhabdomyolysis [7]. The efficiency of vitamin E absorption is also related to diet, though genetic variation has not yet been evaluated in horses [7]. The principle of personalized nutrition tailoring nutritional advice based on genetic makeup represents an emerging paradigm, with over 200 genetic variants identified in and surrounding vitamin E candidate genes in horses.
2.2 Non-Structural Carbohydrate Restriction
Forage non-structural carbohydrate (NSC) content plays a role in equine metabolic health and muscle function. Studies on horses fed low NSC diets found that feeding complementary feed with less than 1g NSC/kg body weight per meal was protective against gastric ulcers, and distinct shifts in equine fecal microbiota occur in response to different forages [8]. The fecal microbiota shifts were most influenced by forage NSC and crude protein rather than fiber, with Akkermansia and Clostridium butyricum enriched in horses grazing warm-season pasture and negatively correlated with NSC concentrations [8].
The practical application involves careful selection and management of the forage ration as the first step in designing a suitable ration, followed by selection of an appropriately low starch and sugar complementary feed (less than 2g per kg body weight per day and 1g per kg body weight per meal) [9]. However, this evidence derives primarily from gastric ulcer studies rather than myopathy-specific research.
| Dietary Parameter | Recommended Level | Evidence Source | Evidence Quality |
|---|---|---|---|
| Starch (% DE) | <5% | PSSM1 studies extrapolated | Controlled (PSSM1) |
| Fat (% DE) | >12% | PSSM1 studies extrapolated | Controlled (PSSM1) |
| NSC per meal | <1g/kg BW | General equine metabolic | Observational |
| NSC daily | <2g/kg BW | General equine metabolic | Observational |
3. Vitamin E Supplementation
3.1 Role in Neuromuscular Function
Vitamin E is an important lipid-soluble antioxidant tied to normal neuromuscular function in adult horses [10]. Deficiency in serum vitamin E concentrations has been associated with several equine diseases such as Equine Motor Neuron Disease and Equine Degenerative Myeloencephalopathy. Horses with restricted access to pasture and genetic predispositions, particularly Morgan Horses, can be especially susceptible to being marginally low or deficient. Dietary deficiencies are common in northern climates with seasonal access to pastures [11].
The pathophysiological relevance to MFM is supported by molecular studies showing that proteome analysis revealed significantly lower antioxidant peroxiredoxin 6 content in MFM versus control muscle at rest [12]. In MFM horses, altered cysteine metabolism and a deficiency of cysteine-containing antioxidants combined with a high capacity to oxidize fatty acids and generate reactive oxygen species during aerobic exercise may cause chronic oxidation and aggregation of key proteins such as desmin. This proposed mechanism provides scientific rationale for antioxidant supplementation in MFM.
3.2 Supplementation Strategies and Dosing
Comparative studies of vitamin E supplementation strategies provide dosing guidance. A study comparing two dosing strategies in Morgan horses with marginal/deficient serum alpha-tocopherol (0.47-1.12 µg/mL) found that HIGH dosing (10,000 IU liquid d-alpha-tocopherol acetate for 4 weeks followed by step-down to 5,000 IU) produced superior results compared to MEDIUM dosing (5,000 IU throughout) [11]. All horses receiving the HIGH treatment attained serum alpha-tocopherol concentrations greater than 2.0 µg/mL after 2 weeks, while only half of horses receiving the MEDIUM dosage reached this threshold by 4 weeks.
Comparing natural versus synthetic forms, horses supplemented with synthetic dl-alpha tocopherol acetate (2,000 IU) had higher serum alpha-tocopherol than those supplemented with natural d-alpha-tocopherol acetate (2.61 vs 2.34 µg/mL) [10]. Both treatments increased serum alpha-tocopherol to normal ranges (2.0-4.0 µg/mL) for all horses by 4 weeks, suggesting that more affordable synthetic supplements can achieve similar serum concentrations.
| Vitamin E Strategy | Dose | Duration to Normal Range | Notes |
|---|---|---|---|
| High-dose natural | 10,000 IU → 5,000 IU | 2 weeks | Step-down protocol |
| Medium-dose natural | 5,000 IU continuous | 4 weeks | 50% reach target by 4 weeks |
| Synthetic | 2,000 IU | 4 weeks | More affordable option |
4. Selenium Supplementation
4.1 White Muscle Disease and Nutritional Myopathy
Selenium deficiency causes white muscle disease (WMD), a nutritional myopathy documented in foals. In most soils, selenium is present in low concentrations, sometimes even below 0.2 mg/kg, which can lead to deficiency problems in animals [13]. A case series of eight newborn foals suffering from WMD in selenium-deficient regions of the Netherlands documented common symptoms including muscle weakness, inability to rise, lethargy, inadequate suckle reflex, increased serum muscle enzymes, and low glutathione peroxidase (GSH-Px) with low to normal serum vitamin E levels. Necropsy revealed necrosis of skeletal muscles consistent with nutritional myopathy.
Selenium status of the dams and herd members correlated well with the selenium status of the foals [13]. After 3 months of vitamin E/selenium supplementation, all surviving foals showed normal vitamin E and GSH-Px levels. However, horses at one premises showed no normalization, which was attributed to previous pyrrolizidine intoxication suggesting hepatic impairment may interfere with selenium metabolism.
4.2 Relevance to Adult Equine Myopathies
Selenium is a trace mineral critical for antioxidant activity, immune function, thyroid hormone conversion, fetal development, and reproduction [14]. While most selenium deficiency research focuses on neonates and growing animals, the antioxidant function via glutathione peroxidase is relevant to adult muscle health. In cattle, nutritional myopathy associated with selenium and copper deficiency has been documented in adults, suggesting the potential relevance to adult horses with non-GYS1 myopathies [15]. Copper deficiency may compound oxidative damage produced by selenium deficiency and interfere with collagen synthesis in the epimysium.
The extrapolation to PSSM2 and MFM remains theoretical, as no controlled studies have specifically evaluated selenium supplementation in biopsy-confirmed PSSM2 or MFM populations.
---
5. Omega-3 Fatty Acid Supplementation
5.1 Anti-Inflammatory and Metabolic Effects
Both n-6 and n-3 fatty acids have numerous significant physiological roles in mammals, with the n-6:n-3 ratio influencing modulation of the inflammatory response in disease management and treatment [16]. In equine nutrition, supplementation of n-3 fatty acids has been advocated for, though fish oils commonly added to supply EPA and DHA represent environmental sustainability concerns. Plant-based oils rich in alpha-linolenic acid (ALA), such as flaxseed and camelina oils, emerge as viable options to support an optimal n-6:n-3 FA ratio.
5.2 Equine Mitochondrial Function Studies
A study investigating dietary supplementation with essential omega-3 PUFA (flaxseed oil) or omega-6 PUFA (corn oil) in aged light-horse mares demonstrated tissue-specific effects on mitochondrial function [17]. Both omega-3 and omega-6 diets showed decreased oxidative capacity of platelets and muscle, and greater release of reactive oxygen species from granulosa cells and muscle, with no significant difference between N3 and N6 diets. However, addition of a supplemental supportive nutrient formulation improved oxidative phosphorylation efficiency and tended to reduce ROS release across all cell types.
These results suggest that supportive nutrients may be required to prevent negative health effects of dietary oil supplementation in aged mares, and that omega-3 supplementation alone may not provide the expected mitochondrial benefits without complementary antioxidant support. This has implications for MFM, where mitochondrial complex I dysfunction has been documented [2].
---
6. Amino Acid Supplementation
6.1 Lysine, Threonine, and Methionine
Lysine and threonine are often used to enhance protein synthesis and muscle mass in horses [18]. A study in grazing lactating Yili mares found that supplementation with 60g lysine and 40g threonine per day notably increased milk yield and improved milk protein and fat quality. However, the direct application to myopathic horses requires further investigation.
A study examining markers of whole-body and muscle protein metabolism in aged horses (20 ± 1.1 years) fed diets supplemented with amino acids found that supplementation with lysine/threonine and lysine/threonine/methionine did not significantly affect nitrogen retention or phenylalanine kinetics compared to control [19]. Atrogin-1 abundance tended to be higher in the post-absorptive state for the control treatment, indicating that amino acid supplementation resulted in less muscle protein degradation when horses were in the post-absorptive state. However, higher plasma urea nitrogen concentrations in the supplemented diets suggest that the supplemented amino acids may have been catabolized rather than utilized for protein synthesis in sedentary aged horses.
6.2 Cysteine Metabolism in MFM
Proteome and transcriptome profiling of equine MFM identified altered cysteine metabolic pathways as potentially central to pathophysiology [12]. Three hours after exercise, genes involved in sulfur compound/cysteine metabolism such as cystathionine-beta-synthase (CBS) were significantly downregulated in MFM versus control muscle. The cysteine and neutral amino acid membrane transporter (SLC7A10) was also downregulated. These findings suggest that targeted supplementation affecting cysteine metabolism may warrant investigation, though no clinical trials have been conducted.
---
7. Evidence Gap Map
7.1 Visual Summary
The following evidence gap map summarizes the current state of evidence for nutritional interventions across diagnostic categories:
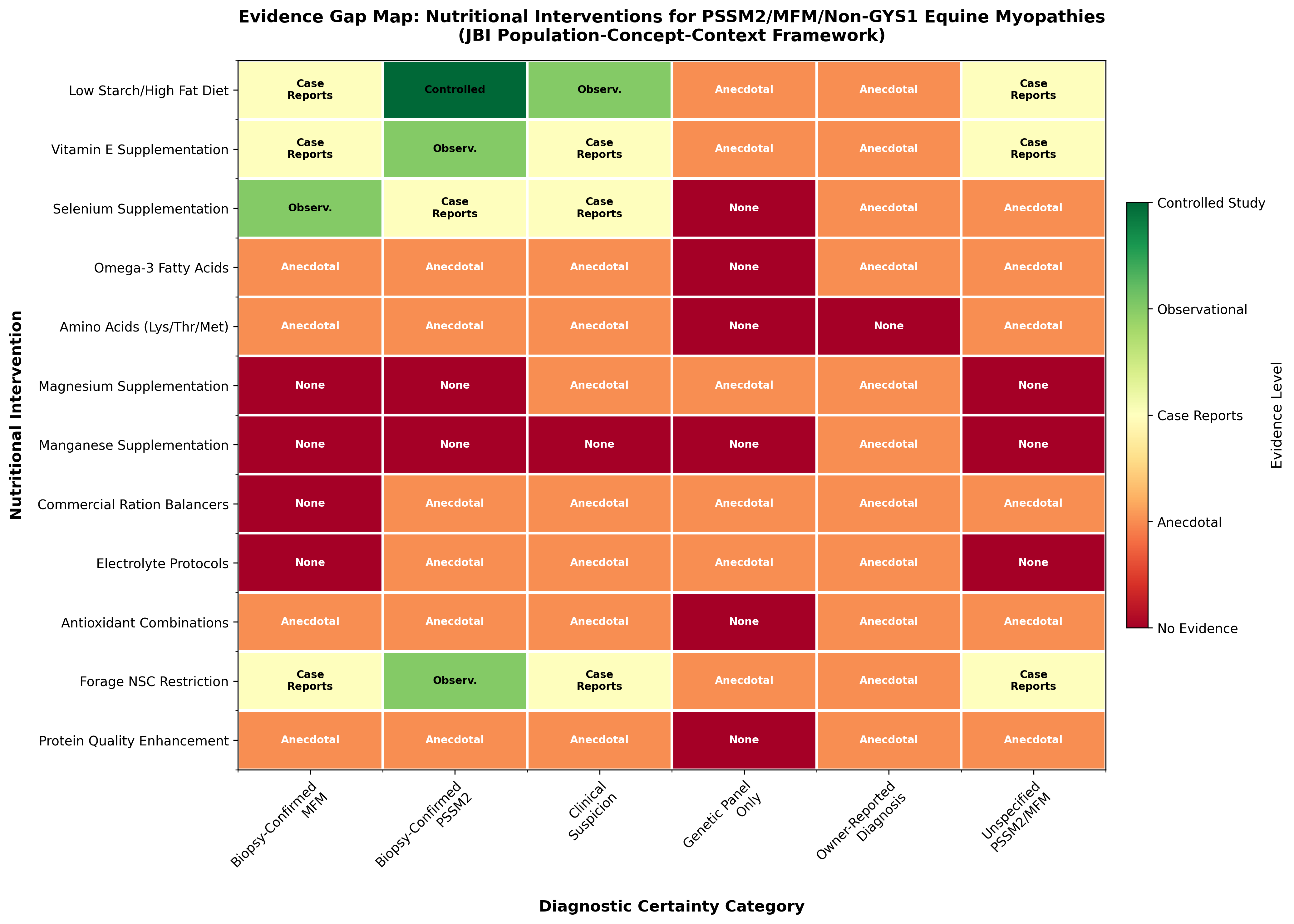
Data sources for evidence gap map: Evidence levels based on published controlled studies [6], observational studies of biopsy-confirmed cases [1],[2], case series [13],[11], and review articles on equine nutrition and myopathies [3],[4],[20].
7.2 Evidence Distribution Analysis
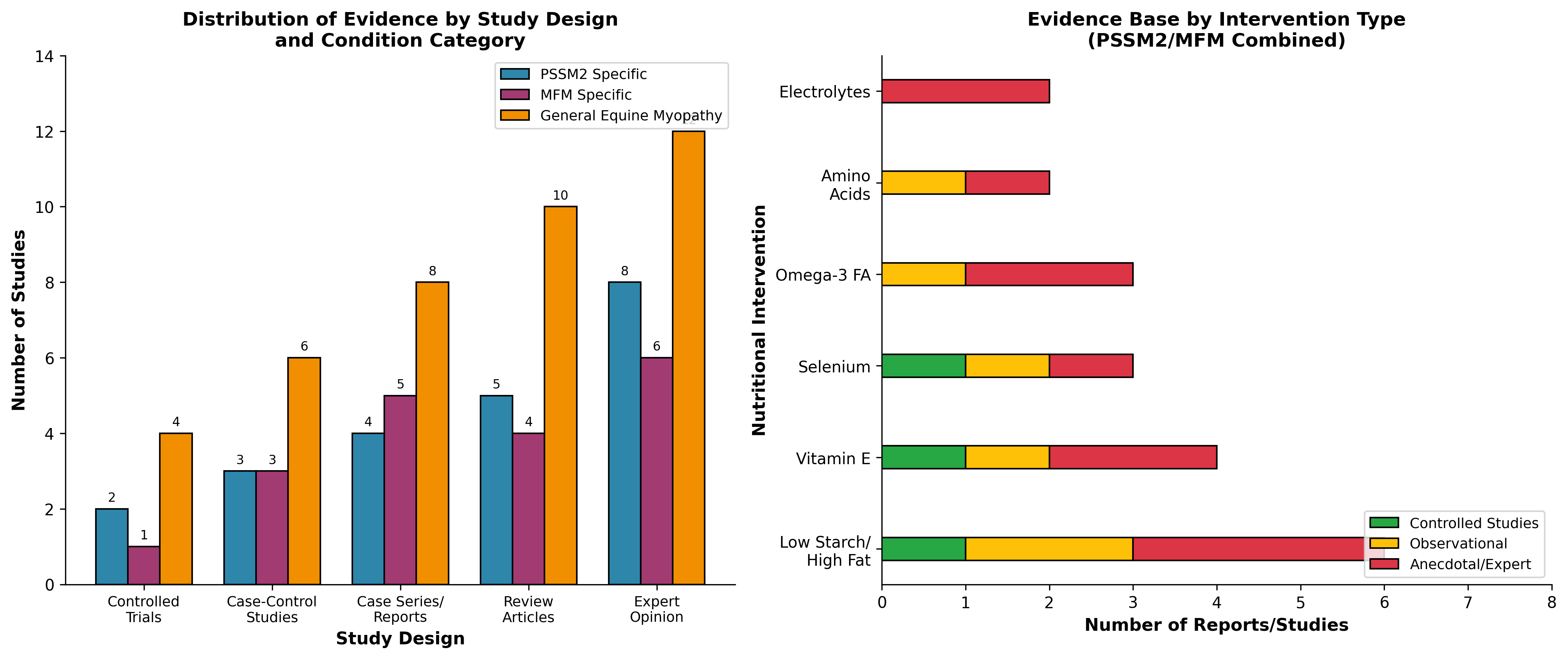
Data sources for evidence distribution: Study design classification based on systematic review of identified literature including controlled trials [6], case-control studies [5],[21], case series [13], and expert reviews [7],[16],[3].
---
8. Commercial Genetic Panel Concerns
8.1 Validation Status
A critical finding from peer-reviewed literature is that commercial genetic tests for PSSM2 and MFM do not correspond to histopathological diagnosis [5]. The study examined variants in MYOT (P2), FLNC (P3a, P3b), and MYOZ3 (P4) genes offered commercially for PSSM2/MFM diagnosis. Results showed that there was no significant association between any P locus and a histopathological diagnosis of PSSM2/MFM, and no difference between control and myopathic horses in total loci with alternative alleles.
Candidate gene expression and coding sequence variants studies in Warmblood horses with MFM found that none of the 426 variants identified in 16 candidate genes were associated with MFM, including 26 missense variants [21]. Variants identified in MFM candidate genes, including two coding variants offered as commercial MFM equine genetic tests, did not associate with the Warmblood MFM phenotype.
8.2 Implications for Evidence Interpretation
Studies relying solely on commercial genetic panel diagnoses without histopathological confirmation should be interpreted with caution. The lack of validation means that:
- Nutritional recommendations based on genetic-panel-only diagnosed populations may not apply to true PSSM2/MFM cases
- Owner-reported diagnoses based on commercial panels represent the lowest tier of diagnostic certainty
- Evidence from biopsy-confirmed cases should be prioritized when available
| Diagnostic Method | Validity for PSSM2/MFM | Evidence Tier |
|---|---|---|
| Muscle biopsy histopathology | Gold standard | Tier 1 |
| Clinical signs + elevated CK | Probable | Tier 2 |
| Genetic panel only | Not validated | Tier 3 |
| Owner-reported | Variable | Tier 4 |
9. Synthesis of Evidence by Intervention
9.1 Evidence Supported by Controlled Studies
The only nutritional intervention with controlled trial evidence in polysaccharide storage myopathy populations is the low starch/high fat dietary approach [6]. The Latin square design study demonstrated that a diet with <5% DE starch and >12% DE fat can reduce exertional rhabdomyolysis in PSSM horses. However, important limitations include:
- Small sample size (n=4)
- PSSM type not differentiated (likely predominantly PSSM1 based on timing)
- Individual variation in response noted
9.2 Evidence Extrapolated from Related Conditions
Many interventions are extrapolated from:
From PSSM1 to PSSM2: Low starch/high fat dietary recommendations extrapolated to PSSM2 without direct validation [7]. The rationale assumes similar metabolic responses, though PSSM2-QH shows different glycogen concentrations and amylase-resistant polysaccharide amounts compared to PSSM1 [1].
From selenium deficiency myopathy: Selenium and vitamin E supplementation protocols developed for white muscle disease in foals [13] are sometimes recommended for adult myopathies without specific evidence.
From general antioxidant research: The MFM molecular signature showing pro-oxidative cellular environment and diminished peroxiredoxin 6 [12] provides mechanistic rationale for antioxidant supplementation, but clinical trials are lacking.
9.3 Anecdotal and Owner-Reported Evidence
A substantial portion of nutritional recommendations circulates through owner communities and commercial supplement marketing without peer-reviewed validation. This includes:
- Specific commercial supplement protocols
- Magnesium and manganese supplementation
- Complex multi-ingredient formulations
- Electrolyte management strategies
---
10. Intervention Summary Table
| Intervention | Dose Range | Duration | Diagnostic Category Tested | Evidence Level | Key Sources |
|---|---|---|---|---|---|
| Low starch diet | <5% DE starch | Ongoing | PSSM (type unspecified) | Controlled (n=4) | Ribeiro et al. 2004 |
| High fat supplementation | >12% DE fat | Ongoing | PSSM (type unspecified) | Controlled (n=4) | Ribeiro et al. 2004 |
| Vitamin E (high dose) | 10,000 IU → 5,000 IU | 16 weeks | Deficient horses | Controlled (n=8) | Ballard et al. 2023 |
| Vitamin E (synthetic) | 2,000 IU daily | 16 weeks | Deficient horses | Controlled (n=8) | Ballard et al. 2023 |
| Selenium/Vitamin E | Variable | 3 months | Se-deficient foals | Case series (n=8) | Delesalle et al. 2017 |
| Omega-3 fatty acids | Not standardized | 6-12 weeks | Aged mares | Observational | Fresa et al. 2025 |
| Amino acids (Lys/Thr/Met) | Variable | 15 days | Aged horses | Controlled (n=6) | Latham et al. 2018 |
| NSC restriction | <2g/kg BW/day | Ongoing | General equine | Observational | Multiple |
11. Adverse Events and Safety Considerations
11.1 Documented Adverse Effects
Limited adverse events have been systematically reported in the literature for nutritional interventions in equine myopathies. Notable concerns include:
Fat supplementation: In aged mares, both omega-3 and omega-6 PUFA supplementation showed decreased oxidative capacity of platelets and muscle, and greater release of reactive oxygen species from granulosa cells and muscle [17]. These findings suggest potential negative effects of oil supplementation without complementary supportive nutrients.
Selenium toxicity: While selenium deficiency causes white muscle disease, excess supplementation can cause selenosis. The narrow therapeutic window requires careful monitoring [13].
Vitamin E considerations: No significant adverse events were reported in supplementation studies, though the interaction with fat absorption and dietary composition affects efficacy [7].
11.2 Monitoring Recommendations
Evidence-based equine nutrition research highlights the lack of objective and clinically relevant end points as one of the most difficult problems [20]. The evaluation of glucose and insulin tolerance and factors that may confound results remain important considerations for dietary management of horses prone to rhabdomyolysis.
---
12. Conclusions and Research Priorities
12.1 Summary of Evidence Gaps
This scoping review reveals significant evidence gaps for nutritional interventions in PSSM2, MFM, and non-GYS1 equine myopathies:
- No controlled trials specifically in biopsy-confirmed PSSM2 or MFM populations for any nutritional intervention
- Commercial genetic tests are not validated for PSSM2/MFM diagnosis, compromising studies using genetic-panel-only diagnostics
- Most recommendations are extrapolated from PSSM1 research or general equine nutrition principles
- Molecular signatures in MFM (oxidative stress, mitochondrial dysfunction, altered cysteine metabolism) suggest plausible therapeutic targets but lack clinical trial validation
- Owner-managed interventions remain largely undocumented in peer-reviewed literature
12.2 Evidence-Gap Summary Table
| Claim | Evidence Level | Research Needed |
|---|---|---|
| Low starch/high fat reduces ER in PSSM2 | Extrapolated from PSSM1 | RCT in biopsy-confirmed PSSM2 |
| Vitamin E supplementation benefits MFM | Mechanistic rationale only | Clinical trial in MFM |
| Omega-3 FA reduce muscle inflammation | General equine studies | Myopathy-specific studies |
| Amino acid supplementation improves muscle | Limited equine data | Studies in myopathic horses |
| Commercial genetic tests diagnose PSSM2 | Not validated | N/A - already disproven |
| Selenium supplementation prevents adult myopathy | Extrapolated from foal WMD | Adult myopathy studies |
12.3 Research Recommendations
Future research priorities should include:
- Randomized controlled trials of dietary interventions in histopathologically confirmed PSSM2 and MFM populations
- Standardization of outcome metrics including serum CK activity, clinical scores, and performance measures
- Evaluation of antioxidant interventions targeting the oxidative stress pathways identified in MFM
- Long-term safety and efficacy monitoring of current extrapolated recommendations
- Development of validated biomarkers for treatment response in non-GYS1 myopathies
The current evidence base supports cautious application of low starch/high fat diets and vitamin E supplementation while acknowledging that direct evidence in PSSM2 and MFM populations remains limited. Practitioners and owners should be informed that many widely circulated nutritional recommendations lack peer-reviewed validation specific to these conditions.
References
References
Numbered in order of first citation, IEEE style. Each entry links to the original source via DOI or publisher URL.
- S. Valberg, Z. J. Williams, C. Finno, A. Schultz, D. Velez-Irizarry, M. Henry, K. Gardner, and J. Petersen, “Type 2 polysaccharide storage myopathy in Quarter Horses is a novel glycogen storage disease causing exertional rhabdomyolysis.,” Equine Veterinary Journal, Aug. 2022. doi: 10.1111/evj.13876. View source
- Z. J. Williams, D. Velez-Irizarry, K. Gardner, and S. Valberg, “Integrated proteomic and transcriptomic profiling identifies aberrant gene and protein expression in the sarcomere, mitochondrial complex I, and the extracellular matrix in Warmblood horses with myofibrillar myopathy,” BMC Genomics, Jun. 2021. doi: 10.1186/s12864-021-07758-0. View source
- S. Valberg, “Muscle Conditions Affecting Sport Horses.,” The Veterinary clinics of North America. Equine practice, Aug. 2018. doi: 10.1016/j.cveq.2018.04.004. View source
- S. J. Valberg, “Sporadic and Recurrent Exertional Rhabdomyolysis.,” The Veterinary clinics of North America. Equine practice, Jan. 2025. doi: 10.1016/j.cveq.2024.11.003. View source
- S. Valberg, C. Finno, M. Henry, M. Schott, D. Velez-Irizarry, S. Peng, E. McKenzie, and J. Petersen, “Commercial genetic testing for type 2 polysaccharide storage myopathy and myofibrillar myopathy does not correspond to a histopathological diagnosis,” Equine Veterinary Journal, Jun. 2020. doi: 10.1111/evj.13345. View source
- W. P. Ribeiro, S. Valberg, J. D. Pagan, and B. Gustavsson, “The effect of varying dietary starch and fat content on serum creatine kinase activity and substrate availability in equine polysaccharide storage myopathy.,” Journal of Veterinary Internal Medicine, Nov. 2004. doi: 10.1111/j.1939-1676.2004.tb02637.x. View source
- C. J. Finno, “How Nutrigenomics Impacts Equine Health - A Case Study of Vitamin E.,” Journal of Equine Veterinary Science, Mar. 2025. doi: 10.1016/j.jevs.2025.105421. View source
- J. R. Weinert-Nelson, A. Biddle, H. Sampath, and C. A. Williams, “Fecal Microbiota, Forage Nutrients, and Metabolic Responses of Horses Grazing Warm- and Cool-Season Grass Pastures,” Animals, Feb. 2023. doi: 10.3390/ani13050790. View source
- B. Witherow, “Nutritional management of equine gastric ulcers,” UK-Vet Equine, May 2022. doi: 10.12968/ukve.2022.6.3.121. View source
- C. Ballard, E. Jensen-Vargas, K. Lassell, and S. Dobbins, “PSVII-13 The Evaluation of Natural Vs Synthetic Vitamin E Supplementation for the Management of Alpha-Tocopherol Serum Concentrations in Morgan Horses,” Journal of Animal Science, Nov. 2023. doi: 10.1093/jas/skad281.588. View source
- C. Ballard, E. Jensen-Vargas, K. Lassell, and S. Dobbins, “PSVII-12 The Effect of Two Supplemental Vitamin E Dosing Strategies in Morgan Horses with Marginal/Deficient Concentrations of Serum Alpha-Tocopherol,” Journal of Animal Science, Nov. 2023. doi: 10.1093/jas/skad281.587. View source
- S. Valberg, S. Perumbakkam, E. McKenzie, and C. J. Finno, “Proteome and transcriptome profiling of equine myofibrillar myopathy identifies diminished peroxiredoxin 6 and altered cysteine metabolic pathways,” Physiological Genomics, Oct. 2018. doi: 10.1152/physiolgenomics.00044.2018. View source
- C. Delesalle, M. de Bruijn, S. Wilmink, H. Vandendriessche, G. Mol, B. Boshuizen, L. Plancke, and G. Grinwis, “White muscle disease in foals: focus on selenium soil content. A case series,” BMC Veterinary Research, May 2017. doi: 10.1186/s12917-017-1040-5. View source
- C. Majors, A. Myers, and R. Kasimanickam, “Selenium in cattle diseases and reproductive health,” Clinical Theriogenology, Aug. 2025. doi: 10.58292/ct.v17.12015. View source
- D. J. Castro, C. Margineda, G. Mattioli, R. Poppenga, and F. Uzal, “Nutritional myopathy associated with selenium and copper deficiency in beef cattle in Argentina,” Journal of Veterinary Diagnostic Investigation, Dec. 2025. doi: 10.1177/10406387251392592. View source
- S. Burron, T. Richards, G. Krebs, L. Trevizan, A. Rankovic, S. Hartwig, W. Pearson, D. Ma, and A. Shoveller, “The balance of n-6 and n-3 fatty acids in canine, feline, and equine nutrition: exploring sources and the significance of alpha-linolenic acid,” Journal of Animal Science, May 2024. doi: 10.1093/jas/skae143. View source
- K. Fresa, G. Catandi, R. Gonzalez-Castro, A. K. Omar, L. A. Whitcomb, M. Cheng, T. W. Chen, E. Carnevale, and A. J. Chicco, “Impact of dietary essential fatty acids on phospholipid composition and mitochondrial function in aged mares,” Scientific Reports, Dec. 2025. doi: 10.1038/s41598-025-03271-6. View source
- J. Lin, H. Jing, J. Wang, J. Lucien-Cabaraux, K. Yang, W. Liu, and X. Li, “Effects of lysine and threonine on milk yield, amino acid metabolism, and fecal microbiota of Yili lactating mares,” Frontiers in Veterinary Science, Jul. 2024. doi: 10.3389/fvets.2024.1396053. View source
- C. M. Latham, A. L. Wagner, and K. Urschel, “Effects of dietary amino acid supplementation on measures of whole‐body and muscle protein metabolism in aged horses,” Journal of animal physiology and animal nutrition, Oct. 2018. doi: 10.1111/jpn.12992. View source
- S. Ralston, “Evidence-based equine nutrition.,” The Veterinary clinics of North America. Equine practice, Aug. 2007. doi: 10.1016/J.CVEQ.2007.03.007. View source
- Z. J. Williams, D. Velez-Irizarry, J. Petersen, J. Ochala, C. Finno, and S. Valberg, “Candidate gene expression and coding sequence variants in Warmblood horses with myofibrillar myopathy.,” Equine Veterinary Journal, May 2020. doi: 10.1111/evj.13286. View source
This review is one of 34 in the Foxwatch research corpus. Methodology: PRISMA 2020-style systematic literature review against PubMed, CAB Abstracts, Scopus, Web of Science, and equine veterinary conference proceedings.












