Systematic review of trace minerals in PSSM2/MFM and neuromuscular disease
PSSM Type 2 and MFM horses are not just Type 1 horses with a less strict diet. The biopsy-defined oxidative stress and protein aggregation profile is different, and a ration balancer chosen for a Type 1 horse routinely under-delivers calcium, phosphorus, magnesium, copper, and zinc to a Type 2 horse. This review writes trace-mineral targets specifically for non-GYS1 muscle disease. Builds on reviews 04 (PSSM2 evidence map) and 05 (amino acids).
Research thesis
This systematic review argues that trace mineral status in PSSM Type 2 and myofibrillar myopathy populations is not interchangeable with that of PSSM1, and that calcium, phosphorus, magnesium, copper, and zinc each warrant separate evaluation in non-GYS1 muscle disease, not only because biopsy-defined MFM has a different oxidative stress and protein aggregation profile than glycogen-accumulation PSSM1, but also because high-fat low-NSC ration design routinely under-delivers specific trace minerals when the ration balancer is chosen with Type 1 in mind; by reviewing the equine trace mineral, biopsy-correlated, and supplementation-response literature in PSSM2 and MFM, this review establishes trace mineral targets for non-GYS1 myopathies and identifies where forage-plus-balancer protocols predictably fall short.
Evidence visualizations & full pages
Evidence figures (2)
Figures referenced by the review's Figure N: captions. Click any chart for a full-size view. They are also rendered inline below their respective captions in the full review text further down.
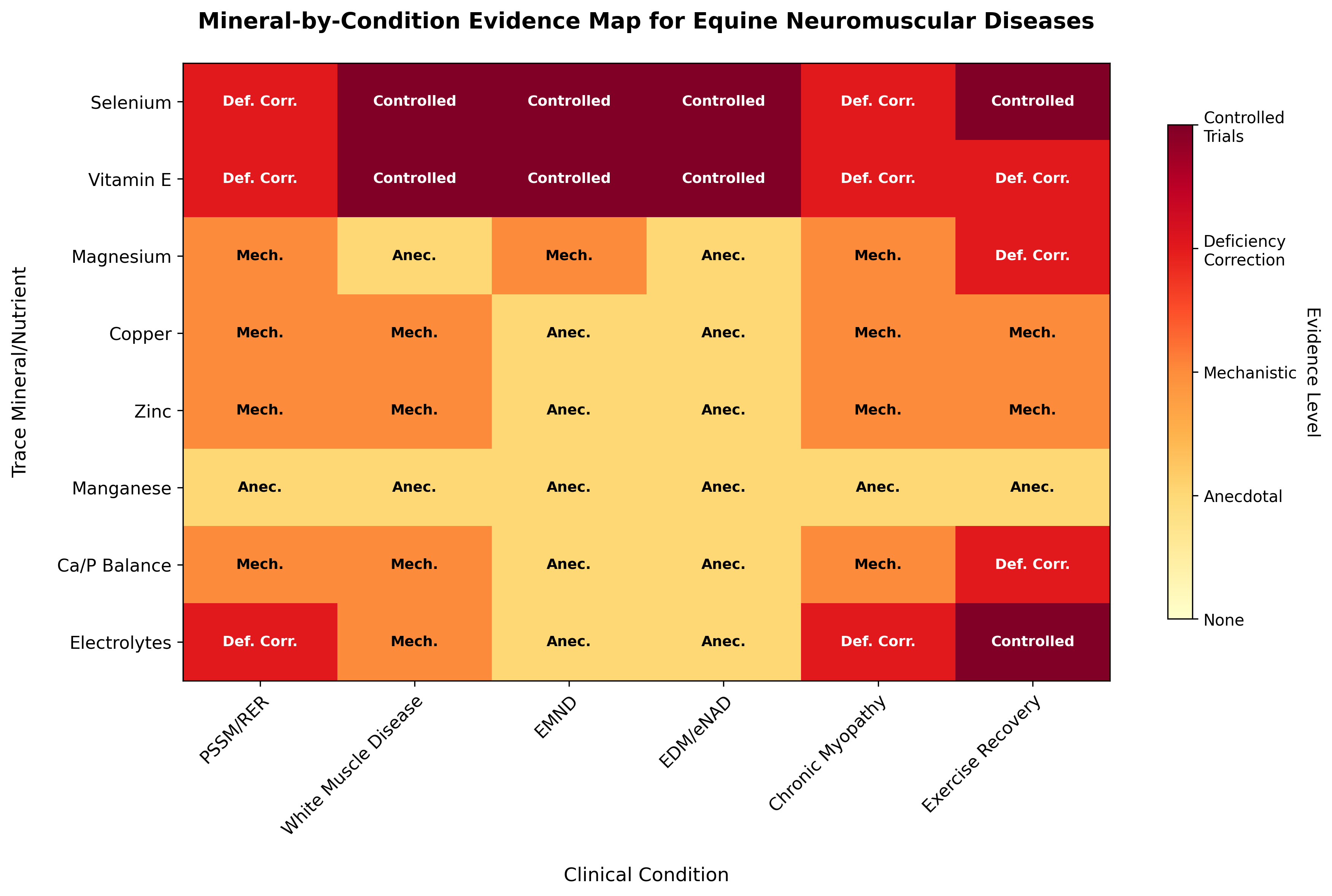 Figure 1: Mineral-by-condition evidence map showing the level of evidence supporting trace mineral interventions in equine neuromuscular diseases. Evidence levels range from anecdotal practice to controlled fe
Figure 1: Mineral-by-condition evidence map showing the level of evidence supporting trace mineral interventions in equine neuromuscular diseases. Evidence levels range from anecdotal practice to controlled fe
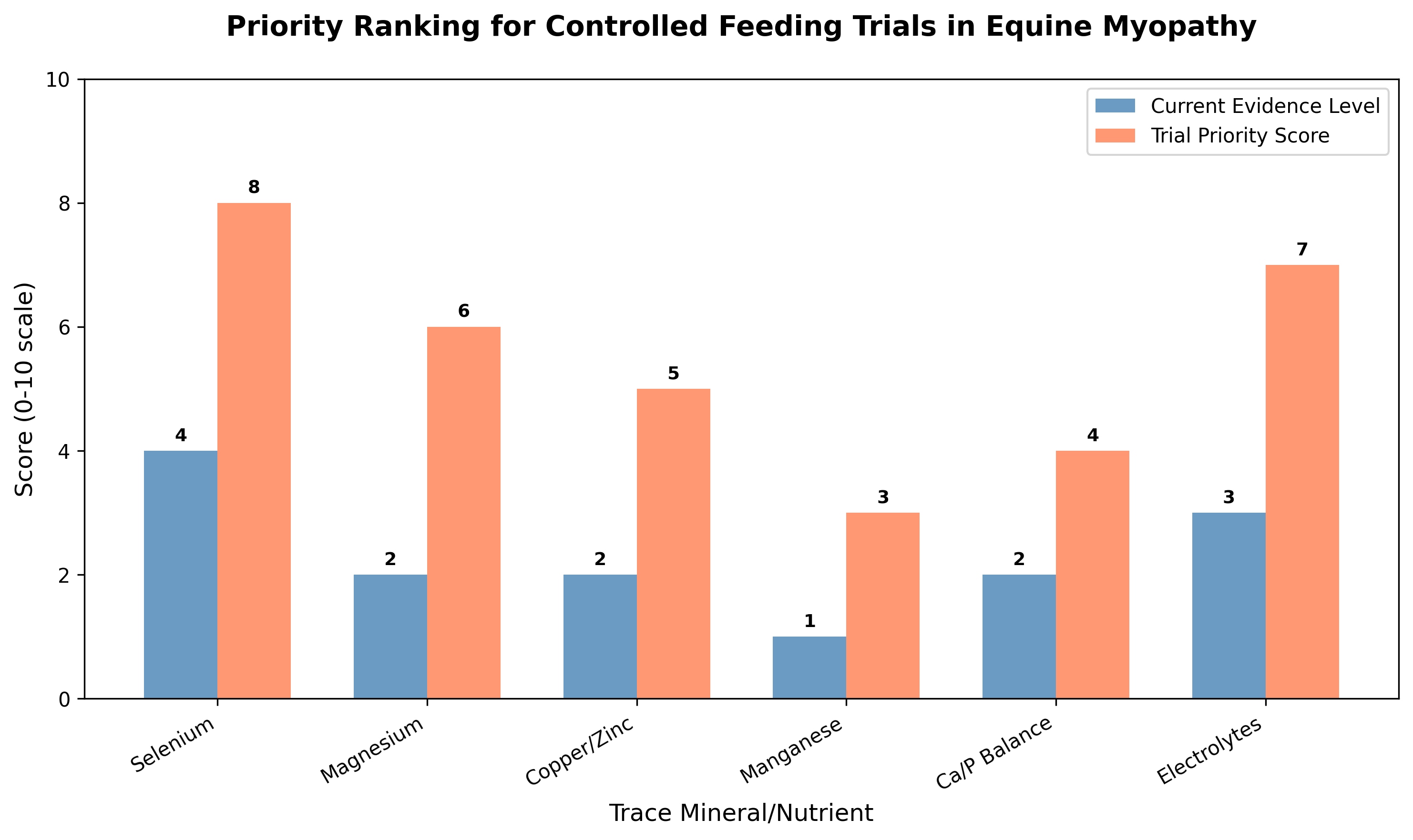 Figure 1: Mineral-by-condition evidence map showing the level of evidence supporting trace mineral interventions in equine neuromuscular diseases. Evidence levels range from anecdotal practice to controlled fe
Figure 1: Mineral-by-condition evidence map showing the level of evidence supporting trace mineral interventions in equine neuromuscular diseases. Evidence levels range from anecdotal practice to controlled fe
Original review pages (PDF render)
Download
Full review text
Trace minerals in PSSM2/MFM and neuromuscular disease
Systematic Review of Trace Minerals in PSSM2/MFM and Neuromuscular Disease in Horses
1. Introduction and Background
1.1 Scope and Rationale
Polysaccharide storage myopathy type 2 (PSSM2), myofibrillar myopathy (MFM), and related chronic equine myopathies represent significant challenges in equine medicine. While dietary management—particularly low-starch, high-fat feeding protocols—has become established for certain myopathies, the role of trace minerals remains incompletely defined. The relationship between mineral status and muscle health has been documented extensively in livestock species, where selenium deficiency causes white muscle disease in foals and other young animals [1]. Understanding the evidence base for mineral interventions is critical because trace mineral supplementation is commonly recommended by practitioners and commercial protocols despite variable supporting data.
This systematic review examines the evidence for trace mineral involvement in equine neuromuscular conditions including PSSM2, MFM, chronic equine myopathy, and neurologic recovery. Selenium is an essential trace element important for antioxidant defense, immune function, and thyroid hormone metabolism, with deficiency manifested by increased susceptibility to infections and muscular disorders [1]. Mineral homeostasis affects multiple physiological systems relevant to muscle function, including antioxidant capacity, energy metabolism, electrolyte balance, and neuromuscular transmission.
1.2 Methodology and Literature Search Strategy
A comprehensive literature search was conducted across multiple databases to identify studies examining trace minerals in equine myopathies and neuromuscular diseases. The review encompasses magnesium, manganese, copper, zinc, selenium, calcium/phosphorus balance, and electrolytes. Data extraction focused on baseline mineral status, forage analysis, diet formulation, supplementation dose, diagnostic certainty, clinical outcomes, biomarkers, and adverse events. Evidence was categorized according to controlled trial data, deficiency correction studies, mechanistic plausibility, and anecdotal practice.
---
2. Selenium and Vitamin E in Equine Myopathies
2.1 Selenium Deficiency and White Muscle Disease
Selenium occupies a unique position among trace minerals due to its well-established role in preventing nutritional muscular dystrophy. In a comprehensive study comparing selenium and vitamin E status in equine farms with and without myopathy occurrence in the Czech Republic, researchers found significantly lower whole blood selenium concentrations (43.09 vs. 117.35 μg/L), serum vitamin E (9.55 vs. 10.75 μmol/L), and glutathione peroxidase (GSH-Px) activities (153.38 vs. 397.41 μkat/L) in horses from farms with myopathies compared to unaffected farms [2]. Nutritional myodegeneration (white muscle disease) was definitively diagnosed in six horses from myopathy-affected farms. Poor selenium and vitamin E saturation represent important factors in white muscle disease, though other myopathies with different etiologies may also be influenced by selenium status.
The Transylvanian enzootic equine rhabdomyolysis syndrome provides a natural experimental model for understanding regional selenium deficiency. In this well-defined region of Harghita county, prevalence of recurrent exertional rhabdomyolysis reaches 17-23% at high-altitude settlements (>800m) compared to <2% in neighboring valley villages [3]. Horses from affected regions had significantly lower GSH-Px levels (P=0.006), with a strong correlation between erythrocyte GSH-Px activity and selenium concentration (r=0.777, P<0.001). Hay samples from affected areas contained lower selenium concentrations, demonstrating the importance of forage analysis in understanding regional myopathy patterns.
2.2 Selenium Supplementation and Antioxidant Response
Dietary selenium supplementation produces measurable effects on antioxidant enzyme activities in equine skeletal muscle. In untrained Thoroughbred horses receiving either 0.1 mg/kg DM (control) or 0.3 mg/kg DM selenium for 36 days, supplementation above NRC requirements increased serum selenium (P=0.011) and thioredoxin reductase (P=0.051), though glutathione peroxidase was not significantly affected at rest [4]. Following prolonged exercise, selenium-supplemented horses had elevated muscle lipid hydroperoxides compared to controls, but this effect was attenuated with selenium supplementation, suggesting benefits for mitigating oxidative damage and aiding post-exercise recovery.
The form of selenium supplementation affects bioavailability. Meta-analysis demonstrates that selenium-enriched foods are more effective than selenomethionine at increasing GPx activity, with tissue-specific sensitivities including red blood cells, kidney, and muscle [5]. Complexed trace mineral supplementation in young Quarter Horses entering training demonstrated higher muscle GPx activity compared to inorganic trace mineral supplementation throughout the study period [6]. While complexed trace minerals did not affect muscle damage markers following a trailer stressor, dietary complexed trace minerals may be useful for improving antioxidant capacity during exercise training.
2.3 Selenium in Neonatal Foals and Rhabdomyolysis
Selenium deficiency poses particular risks in neonatal foals, where marked electrolyte abnormalities including hyperkalemia, hyponatremia, hypocalcemia, and hyperphosphatemia were documented in neonatal foals with acute rhabdomyolysis and pigmenturia [7]. Rhabdomyolysis was attributed to selenium deficiency, with or without vitamin E deficiency, possibly combined with increased oxidant stress from sepsis or hypoxia reperfusion injury during parturition. Destruction of the intracellular compartment through myonecrosis produced life-threatening electrolyte derangements, highlighting the critical importance of adequate maternal selenium status during gestation.
---
3. Vitamin E and Neuromuscular Diseases
3.1 Equine Motor Neuron Disease (EMND) and Vitamin E Deficiency
Vitamin E deficiency plays a central role in the pathogenesis of equine motor neuron disease. Horses obtain vitamin E, an essential nutrient for neuromuscular health, primarily through green pasture, and dietary deficiencies can lead to neuromuscular diseases including equine neuroaxonal dystrophy/equine degenerative myeloencephalopathy (eNAD/EDM), EMND, and vitamin E-deficient myopathy [8]. Vitamin E supplementation is effective at slowing or halting clinical signs of some diseases, though neuromuscular damage is usually irreversible except in vitamin E-deficient myopathy.
A field trial establishing the causal relationship between vitamin E deficiency and EMND demonstrated that horses maintained on vitamin E-deficient diets developed EMND within 44 months of enrollment, with a median time of 38.5 months [9]. There was large variability in levels of vitamins A and C, β-carotene, GSH-Px, and SOD1, but plasma vitamin E levels dropped significantly over time. Ten of eleven horses developed EMND, while none of the control horses developed the disease, confirming vitamin E deficiency as a risk factor.
3.2 Equine Degenerative Myeloencephalopathy (EDM)
Equine degenerative myeloencephalopathy represents a neurologic disease of young horses characterized by insidious development of symmetric ataxia. Decreased or absent cutaneous trunci reflex responses increase clinical suspicion for this disease, along with concurrent predisposing factors including familial history and inadequate access to green pasture [10]. Vitamin E deficiency and hereditary predisposition are currently considered the most significant pathogenic factors. Animals predisposed to EDM or with clinical diagnosis should receive oral alpha-tocopherol acetate supplementation, though prognosis for complete recovery is generally poor.
3.3 Vitamin E-Responsive Myopathy
A distinct vitamin E-deficient myopathy has been characterized histologically, presenting with a moth-eaten appearance in mitochondria and anguloid myofiber atrophy in sacrocaudalis dorsalis medialis muscle biopsy specimens [11]. Horses with clinical signs similar to EMND were highly responsive to vitamin E treatment, suggesting this may represent a specific syndrome or possibly precede development of neurogenic fiber atrophy typical of EMND. This vitamin E-responsive presentation offers a more favorable prognosis than established neurogenic disease.
3.4 Vitamin E Supplementation Forms and Efficacy
The bioavailability of different vitamin E forms has clinical implications. In Morgan horses with marginal or deficient serum alpha-tocopherol (0.94-2.3 μg/mL), both natural (d-alpha-tocopherol acetate) and synthetic (dl-alpha-tocopherol acetate) supplements at 2,000 IU increased serum concentrations to normal ranges (2.0-4.0 μg/mL) by 4 weeks [12]. Horses supplemented with synthetic vitamin E had higher serum alpha-tocopherol than those with natural supplementation (2.61 vs. 2.34 μg/mL, P=0.05), suggesting synthetic forms provide a more affordable option. Oral vitamin E administration in healthy horses resulted in increases in both serum and cerebrospinal fluid alpha-tocopherol concentrations [13].
---
4. Dietary Management of Polysaccharide Storage Myopathy and Recurrent Exertional Rhabdomyolysis
4.1 Low-Carbohydrate, High-Fat Diets
The cornerstone of PSSM dietary management involves restricting dietary starch and increasing fat content. In a case series of 19 horses with exertional rhabdomyolysis, all had evidence of chronic myopathic changes and abnormal glycogen accumulation on muscle biopsy, with 11 also showing complex polysaccharide accumulation [14]. Following adaptation to a low-carbohydrate, high-fat diet over 3-6 months, 16 horses did not have episodes of rhabdomyolysis, with only mild episodes following reduction of dietary fat intake or exercise restriction. Post-exercise serum CK activities were significantly less than initial values.
The effect of varying dietary starch and fat content was systematically evaluated in Quarter Horse-related mares with polysaccharide storage myopathy using a Latin square design [15]. Four isocaloric diets ranging from 21.2% to 3.9% digestible energy from starch and 7.2% to 12.7% from fat were fed in 6-week periods. Postprandial glucose and insulin responses were higher with high-starch diets, while log CK activities varied among diets. Individual variation occurred with differing starch and fat content, but in horses with clinical manifestations of PSSM, diets with <5% starch and >12% fat DE can reduce exertional rhabdomyolysis potentially by increasing fat oxidation metabolism.
4.2 Recurrent Exertional Rhabdomyolysis in Thoroughbreds
Rations high in grain and formulated to exceed daily energy requirements may increase episodes of rhabdomyolysis in Thoroughbred horses susceptible to recurrent exertional rhabdomyolysis (RER) [16]. This finding emphasizes that nutritional management extends beyond macronutrient composition to include appropriate caloric balance relative to workload.
4.3 Fiber vs. Cereal Grain-Based Diets and Welfare
The metabolic consequences of high cereal grain feeding extend beyond myopathy risk to broader welfare concerns. In horses fed high amounts of cereal grains compared to fiber-based diets, researchers documented increased intestinal permeability, altered muscle characteristics, and changes in oxidative status [17]. Horses on high-cereal diets had lower muscle pH, lighter color, and reduced intramuscular fat concentrations. A high-cereal diet can have global impacts on horse physiology and represents a potential welfare threat.
---
5. Magnesium, Copper, Zinc, and Manganese
5.1 Magnesium Status and Neuromuscular Function
Magnesium plays critical roles in nerve transmission and muscle contraction. The 24-hour urinary excretion of magnesium represents the most sensitive indicator of reduced magnesium intake in horses, while spot sample fractional clearance of magnesium can conveniently identify horses consuming a magnesium-deficient diet [18]. Muscle cramps commonly occur as an isolated phenomenon or may be disease-associated, with a neurogenic origin hypothesized based on occurrence in neuromuscular diseases including polyneuropathies and radiculopathies [19]. Magnesium oral supplementation is marketed as over-the-counter prophylaxis for muscle cramps, though the role of supplementation for preventing and treating cramps remains unclear.
Electromyographic studies in horses with induced hypocalcemia and hypomagnesemia demonstrated spontaneous activity in measured muscles indicative of nerve hyperirritability, despite absence of classical clinical signs [20]. Calcium and magnesium deficits appear to have subclinical consequences affecting neuromuscular function, which is of interest for equestrian sports where hypomagnesemia is expected during endurance rides.
5.2 Copper, Zinc, and Selenium Interactions
A study exploring correlations between selenium, copper, and zinc and their biomarkers in adult horses from southern Chile found significant correlations for blood GPx and selenium (r=0.79), blood GPx and SOD (r=-0.6), muscular GPx and SOD (r=0.78), and copper and ceruloplasmin (r=0.48) [21]. These results confirm the strong association between blood GPx and selenium in horses, validating GPx as a diagnostic proxy for selenium deficiency, and suggest significant interactions between GPx and SOD in blood and muscle tissue.
Zinc, copper, and selenium deficiencies were documented in broodmares in southeastern Poland, with zinc concentrations in all studied regions not exceeding 10.7 μmol/L against a reference range of 14.9-29.2 μmol/L [22]. In certain regions, mares had combined selenium, copper, and zinc deficiency. Despite identified deficiencies, mares gave birth on time and registered newborn foals, suggesting subclinical deficiency states may exist without immediate clinical manifestations.
5.3 Trace Element Deficiencies: A Comparative Perspective
A comprehensive review of trace element deficiencies in farm animals emphasizes that selenium contributes to the development and function of cytotoxic T cells, natural killer cells, and helper cells of the immune system [23]. Copper serves as a co-factor for numerous enzymatic processes including collagen synthesis, red blood cell maturation, energy production, and defense against oxidative damage. Zinc is necessary for functioning of proteins, enzymes, and transcription factors. Manganese plays a crucial role in enzyme activation and is involved in glucose and lipid metabolism. Even minerals needed in small quantities can cause diseases in livestock when deficient, leading to significant financial losses.
---
6. Calcium/Phosphorus Balance and Electrolytes
6.1 Electrolyte Disturbances in Exercise and Rhabdomyolysis
Horses that sweat for prolonged periods lose considerable amounts of water and electrolytes, requiring replacement to maintain hydration and prevent dehydration [24]. Effective electrolyte supplements are designed to replace proportions of ions lost through sweating; failure to do so can result in electrolyte imbalance. The small intestine serves as the primary site of rapid absorption of ions and water, which are transported to muscles and skin to replace losses.
Physiological and biochemical evaluation of Quarter Horses during barrel racing demonstrated that after exercise, heart rate, packed cell volume, total plasma proteins, glucose, lactate, creatinine, phosphorus, and sodium increased significantly, with decreased potassium but no hypophosphatemia or hypokalemia [25]. Following one hour of rest, only heart rate, respiratory rate, and body temperature returned to baseline, while lactate and phosphorus did not fully recover, highlighting the importance of adequate recovery periods.
6.2 Phosphorus and Muscle Function
Phosphorus deficiency and hypophosphatemia are believed to be associated with muscle function disturbances in dairy cows, with relevance to equine medicine. In multiparous cows undergoing phosphorus depletion during the transition period, prolonged dietary P deprivation resulted in pronounced sustained decline in plasma phosphate concentration reaching nadir at calving [26]. Hypophosphatemia did not cause signs of clinically apparent muscle weakness or disturbed function of P-containing compounds in muscle tissue. However, electromyographic examination identified subclinical effects on motor unit action potentials indicative of altered neuromuscular functionality, with increasing occurrence of pathologic spontaneous activity suggestive of myopathy.
6.3 Dietary Cation-Anion Balance
The dietary cation-anion balance (DCAB) strongly affects plasma and urine pH, as well as fractional excretion of sodium, potassium, chloride, and phosphorus in both clinically normal horses and horses with recurrent exertional rhabdomyolysis [27]. This finding has implications for formulating diets that maintain appropriate acid-base status while managing mineral excretion.
---
7. Evidence Synthesis and Mineral-by-Condition Evidence Map
7.1 Summary of Evidence by Mineral
The following table summarizes the evidence base for trace mineral interventions in equine neuromuscular conditions:
| Mineral | Condition | Evidence Level | Key Findings | Source |
|---|---|---|---|---|
| Selenium | White Muscle Disease | Controlled trials | Deficiency causally linked; supplementation preventive | Ludvíková et al., Kósa et al. |
| Selenium | PSSM/RER | Deficiency correction | Lower GSH-Px in affected horses; supplementation improves antioxidant capacity | White-Springer et al. |
| Vitamin E | EMND | Controlled trials | Deficiency causes EMND in 38.5 months median | Mohammed et al. |
| Vitamin E | EDM/eNAD | Mechanistic/clinical | Hereditary susceptibility + deficiency; partial response to supplementation | Miller & Collatos |
| Magnesium | Exercise myopathy | Mechanistic plausibility | Subclinical neuromuscular effects documented | Wijnberg et al. |
| Copper/Zinc | Chronic myopathy | Correlation studies | Interactions with selenium; deficiency common but poorly characterized | Deride et al. |
| Electrolytes | Exercise recovery | Controlled trials | Replacement prevents imbalance; composition critical | Lindinger |
7.2 Visual Evidence Map

Figure 1: Mineral-by-condition evidence map showing the level of evidence supporting trace mineral interventions in equine neuromuscular diseases. Evidence levels range from anecdotal practice to controlled feeding trials. Data synthesized from reviewed literature including Kósa et al. (selenium/rhabdomyolysis), Mohammed et al. (vitamin E/EMND), White-Springer et al. (selenium/exercise), and Wijnberg et al. (magnesium/neuromuscular function).
7.3 Gaps in Current Evidence
Critical gaps exist in the evidence base for most trace minerals beyond selenium and vitamin E. The majority of copper, zinc, manganese, and magnesium research consists of correlation studies or mechanistic extrapolations from other species rather than controlled equine feeding trials. Commercial mineral-balancing protocols lack systematic validation for efficacy in PSSM2/MFM specifically, and forage analysis is rarely integrated with clinical outcome data in published studies.
---
8. Priority Ranking for Controlled Feeding Trials
8.1 Criteria for Trial Prioritization
Minerals warranting controlled feeding trials should meet criteria including: (1) mechanistic plausibility linking mineral function to disease pathophysiology; (2) evidence of deficiency prevalence in affected populations; (3) feasibility of intervention and outcome measurement; and (4) gap between current evidence and clinical practice.
8.2 Priority Assessment

Figure 2: Priority ranking for controlled feeding trials in equine myopathy. Current evidence levels and trial priority scores are shown for each mineral category. Higher priority scores indicate greater need for controlled trials based on mechanistic plausibility and evidence gaps. Priority scores derived from evidence gaps identified in reviewed literature including deficiency prevalence data from Kędzierski et al. and mechanistic studies by Wesolowski et al.
Priority 1 - Selenium: Despite strong existing evidence for selenium in white muscle disease, controlled trials examining selenium supplementation specifically in PSSM2/MFM are limited. Beyond antioxidant functions, selenium influences skeletal muscle mitochondrial energetics through selenoproteins H, N, W, and O that affect calcium homeostasis and biogenesis [28]. Selenoproteins play an important role in maintaining muscle function by delaying exercise fatigue and aging.
Priority 2 - Electrolytes: Electrolyte replacement protocols during exercise and recovery have controlled trial support, but optimization for horses with underlying myopathies requires further study. Metabolic syndrome developing after exercise in sport horses includes dehydration, cytolysis syndrome, and electrolyte imbalance as key features [29].
Priority 3 - Magnesium: Given the documented subclinical neuromuscular effects of magnesium deficiency [20] and the widespread use of magnesium supplementation in horses with muscle disorders, controlled trials examining magnesium status and supplementation outcomes in PSSM2/MFM are warranted.
Priority 4 - Copper/Zinc: The interactions between copper, zinc, and selenium documented in equine muscle tissue [21] suggest these minerals may have synergistic or antagonistic effects on antioxidant capacity that require systematic investigation.
8.3 Recommended Trial Design Elements
Future controlled feeding trials should incorporate:
- Baseline mineral assessment: Whole blood selenium, serum zinc and copper, erythrocyte GSH-Px and SOD activities
- Forage analysis: Regional hay and pasture mineral content with correlation to soil surveys
- Diagnostic certainty: Muscle biopsy confirmation of PSSM2/MFM phenotype
- Outcome measures: Serum CK and AST activities, clinical episode frequency, histopathological changes
- Biomarkers: Antioxidant enzyme activities, oxidative stress markers (malondialdehyde, lipid hydroperoxides)
- Adverse event monitoring: Signs of mineral toxicity, changes in coat/hoof quality
---
9. Commercial Mineral-Balancing Protocols
9.1 Current Practice Patterns
Commercial mineral-balancing protocols are widely marketed for horses with myopathies, typically incorporating selenium, vitamin E, magnesium, and various trace mineral complexes. Organic selenium forms, particularly selenomethionine, have been promoted as more bioavailable than inorganic forms. Revisiting oxidative stress and organic selenium use in animal nutrition demonstrates that responses to supplementary selenium in ruminants tend to be more variable than in monogastric species, likely due to different fates in the rumen following ingestion [30]. The reducing conditions in the rumen may markedly lower assimilation of inorganic selenite, predisposing selenite-fed cattle to potential inadequacy.
9.2 Evidence Evaluation
| Protocol Element | Evidence Basis | Recommendation Level |
|---|---|---|
| Selenium supplementation (0.3 mg/kg DM) | Controlled trials in horses | Strongly supported |
| Natural vitamin E (>2000 IU/day) | Controlled trials; superior to synthetic for CNS penetration | Strongly supported |
| Magnesium supplementation | Mechanistic plausibility; limited controlled data | Conditionally supported |
| Complexed trace minerals | Single controlled study showing improved muscle GPx | Emerging support |
| Commercial "muscle support" blends | No controlled trials specific to PSSM2/MFM | Insufficient evidence |
9.3 Forage Analysis Integration
Assessment of mineral deficiency in animal diets emphasizes that macro-microelements must be present in necessary quantities for involvement in formation of hormones, vitamins, and enzymes [31]. With lack or excess of trace elements in soil, feed, and water, specific metabolic diseases arise and develop in animals. Forage mineral content varies substantially by region and season, necessitating periodic analysis rather than reliance on generic supplementation protocols.
---
10. Conclusions and Recommendations
10.1 Summary of Findings
This systematic review reveals a tiered evidence structure for trace mineral interventions in equine neuromuscular diseases. Selenium and vitamin E have the strongest evidence base, with controlled trials demonstrating both causal relationships with deficiency diseases (white muscle disease, EMND, EDM) and benefits of supplementation. The role of selenium and vitamin E in Transylvanian enzootic equine rhabdomyolysis exemplifies how regional mineral deficiencies can produce high-prevalence myopathy syndromes [3]. For PSSM/RER, dietary control emphasizing low-carbohydrate, high-fat feeding has controlled trial support [14], while trace mineral supplementation serves as an adjunctive rather than primary intervention.
Magnesium, copper, zinc, and manganese have mechanistic plausibility for involvement in muscle function and exercise performance, but lack controlled feeding trial data specific to PSSM2/MFM. Calcium/phosphorus balance and electrolyte management have established roles in exercise recovery and acute rhabdomyolysis management but are not primary therapeutic targets for chronic myopathy. Commercial mineral-balancing protocols incorporate multiple elements but require systematic validation for efficacy beyond deficiency correction.
10.2 Clinical Recommendations
- Assess baseline mineral status in horses diagnosed with PSSM2/MFM, including whole blood selenium, serum vitamin E, and GSH-Px activity
- Conduct forage analysis for selenium, zinc, copper, and other trace minerals to identify regional deficiencies
- Supplement selenium to maintain blood levels >120 μg/L and GSH-Px activity within reference ranges
- Provide natural vitamin E at ≥2000 IU/day for horses without pasture access, with higher doses for neurologic presentations
- Consider electrolyte management during exercise and recovery periods
- Avoid over-supplementation of minerals with narrow therapeutic windows (selenium toxicity at >5 mg/day)
10.3 Future Research Priorities
Controlled feeding trials are urgently needed for magnesium supplementation in chronic equine myopathies, given widespread clinical use despite limited evidence. Electrolyte optimization protocols for horses with underlying myopathies warrant investigation, as do potential interactions between copper, zinc, and selenium affecting antioxidant capacity. The genetics of skeletal muscle disorders in horses continue to be defined at the molecular level [32], and integration of nutritional interventions with genetic risk profiling represents a promising direction for personalized management strategies.
Human subjects maintain appropriate physiological mineral levels through balanced diets, with deficiencies of macrominerals leading to osteoporosis, cardiovascular events, and neuromuscular dysfunction [33]. Similar principles apply to equine health, where trace mineral status influences multiple organ systems beyond skeletal muscle. Future research should examine mineral interactions, optimal bioavailable forms, and integration with the broader metabolic management of horses with neuromuscular disease.
References
References
Numbered in order of first citation, IEEE style. Each entry links to the original source via DOI or publisher URL.
- B. Hosnedlová, M. Kepinská, S. Skalíčková, C. Fernández, B. Ruttkay-Nedecký, T. D. Malevu, J. Sochor, M. Baroň, M. Melčová, J. Zídková, and R. Kizek, “A Summary of New Findings on the Biological Effects of Selenium in Selected Animal Species—A Critical Review,” International Journal of Molecular Sciences, Oct. 2017. doi: 10.3390/ijms18102209. View source
- E. Ludvíková, P. Jahn, L. Pavlata, and M. Vyskočil, “Selenium and Vitamin E Status Correlated with Myopathies of Horses Reared in Farms in the Czech Republic,” Acta Veterinaria Brno, Jan. 2005. doi: 10.2754/avb200574030377. View source
- C. Kósa, K. Nagy, O. Szenci, B. Vincze, E. Andrásofszky, R. Szép, Á. Keresztesi, M. Mircean, M. Taulescu, and O. Kutasi, “The role of selenium and vitamin E in a Transylvanian enzootic equine recurrent rhabdomyolysis syndrome,” Acta Veterinaria Hungarica, Sep. 2021. doi: 10.1556/004.2021.00031. View source
- S. H. White-Springer, S. E. Johnson, J. Bobel, and L. Warren, “Dietary selenium and prolonged exercise alter gene expression and activity of antioxidant enzymes in equine skeletal muscle,” Journal of Animal Science, Jun. 2016. doi: 10.2527/jas.2016-0348. View source
- E. N. Bermingham, J. E. Hesketh, B. R. Sinclair, J. Koolaard, and N. C. Roy, “Selenium-Enriched Foods Are More Effective at Increasing Glutathione Peroxidase (GPx) Activity Compared with Selenomethionine: A Meta-Analysis,” Nutrients, Sep. 2014. doi: 10.3390/nu6104002. View source
- C. M. Latham, E. C. Dickson, R. Owen, C. Larson, and S. H. White-Springer, “Complexed trace mineral supplementation alters antioxidant activities and expression in response to trailer stress in yearling horses in training,” Scientific Reports, Apr. 2021. doi: 10.1038/s41598-021-86478-7. View source
- G. A. Perkins, S. J. Valberg, J. M. Madigan, G. P. Carlson, and S. L. Jones, “Electrolyte Disturbances in Foals with Severe Rhabdomyolysis,” Journal of Veterinary Internal Medicine, May 1998. doi: 10.1111/j.1939-1676.1998.tb02114.x. View source
- A. E. Young and C. J. Finno, “The role of vitamin E deficiency in equine neuromuscular disorders,” UK-Vet Equine, Jan. 2025. doi: 10.12968/ukve.2023.0049. View source
- H. Mohammed, T. Divers, B. Summers, and A. de Lahunta, “Vitamin E deficiency and risk of equine motor neuron disease,” Acta Veterinaria Scandinavica, Jul. 2007. doi: 10.1186/1751-0147-49-17. View source
- M. M. Miller and C. Collatos, “Equine degenerative myeloencephalopathy.,” The Veterinary clinics of North America. Equine practice, Apr. 1997. doi: 10.1016/S0749-0739(17)30254-7. View source
- H. E. Bedford, S. J. Valberg, A. M. Firshman, M. Lucio, M. K. Boyce, and T. N. Trumble, “Histopathologic findings in the sacrocaudalis dorsalis medialis muscle of horses with vitamin E–responsive muscle atrophy and weakness,” Journal of the American Veterinary Medical Association, Apr. 2013. doi: 10.2460/javma.242.8.1127. View source
- C. Ballard, E. Jensen-Vargas, K. Lassell, and S. Dobbins, “PSVII-13 The Evaluation of Natural Vs Synthetic Vitamin E Supplementation for the Management of Alpha-Tocopherol Serum Concentrations in Morgan Horses,” Journal of Animal Science, Nov. 2023. doi: 10.1093/jas/skad281.588. View source
- J. K. Higgins, B. Puschner, P. H. Kass, and N. Pusterla, “Assessment of vitamin E concentrations in serum and cerebrospinal fluid of horses following oral administration of vitamin E,” American Journal of Veterinary Research, Jun. 2008. doi: 10.2460/ajvr.69.6.785. View source
- B. A. Valentine, H. F. Hintz, K. M. Freels, A. J. Reynolds, and K. Thompson, “Dietary control of exertional rhabdomyolysis in horses,” Journal of the American Veterinary Medical Association, May 1998. doi: 10.2460/javma.1998.212.10.1588. View source
- W. Ribeiro, S. J. Valberg, J. Pagan, and B. Essen Gustavsson, “The Effect of Varying Dietary Starch and Fat Content on Serum Creatine Kinase Activity and Substrate Availability in Equine Polysaccharide Storage Myopathy,” Journal of Veterinary Internal Medicine, Jan. 2004. doi: 10.1892/0891-6640(2004)18<887:teovds>2.0.co;2. View source
- J. M. MacLeay, S. J. Valberg, J. D. Pagan, J. Xue, F. D. D. L. Côrte, and J. M. Roberts, “Effect of ration and exercise on plasma creatine kinase activity and lactate concentration in Thoroughbred horses with recurrent exertional rhabdomyolysis,” American Journal of Veterinary Research, Nov. 2000. doi: 10.2460/ajvr.2000.61.1390. View source
- F. Raspa, F. R. Dinardo, I. Vervuert, D. Bergero, M. T. Bottero, D. Pattono, A. Dalmasso, M. Vinassa, E. Valvassori, E. Bruno, P. D. Palo, and E. Valle, “A Fibre‐ vs. cereal grain‐based diet: Which is better for horse welfare? Effects on intestinal permeability, muscle characteristics and oxidative status in horses reared for meat production,” Journal of Animal Physiology and Animal Nutrition, Sep. 2021. doi: 10.1111/jpn.13643. View source
- A. J. Stewart, J. Hardy, C. W. Kohn, R. E. Toribio, K. W. Hinchcliff, and B. B. Silver, “Validation of diagnostic tests for determination of magnesium status in horses with reduced magnesium intake,” American Journal of Veterinary Research, Apr. 2004. doi: 10.2460/ajvr.2004.65.422. View source
- A. Moretti, “What is the role of magnesium for skeletal muscle cramps? A Cochrane Review summary with commentary,” Journal of musculoskeletal & neuronal interactions, Mar. 2021. Available online
- I. D. Wijnberg, J. V. D. Kolk, H. Franssen, and H. J. Breukink, “Electromyographic changes of motor unit activity in horses with induced hypocalcemia and hypomagnesemia,” American Journal of Veterinary Research, Jun. 2002. doi: 10.2460/ajvr.2002.63.849. View source
- C. Deride, R. Chihuailaf, V. Arnés, G. Moran, and B. Uberti, “Relationship between selenium, copper, zinc and their biomarkers in blood and skeletal muscle tissue in adult horses from southern Chile.,” Journal of Equine Veterinary Science, Jul. 2023. doi: 10.1016/j.jevs.2023.104881. View source
- W. Kędzierski, A. Chałabis‐Mazurek, Z. Bełkot, I. Janczarek, and S. Kowalik, “Zinc, copper and selenium deficiencies in broodmares in south-eastern Poland,” Polish Journal of Veterinary Sciences, Sep. 2025. doi: 10.24425/pjvs.2025.156074. View source
- “Diseases and Disorders of Trace Elements Deficiency in Farm Animals: An Illustrated Review,” International Journal of Veterinary Science, Jan. 2024. doi: 10.47278/journal.ijvs/2024.139. View source
- M. Lindinger, “Oral Electrolyte and Water Supplementation in Horses,” Veterinary Sciences, Nov. 2022. doi: 10.3390/vetsci9110626. View source
- C. L. N. Gomes, A. M. Alves, J. D. R. Filho, F. D. J. Moraes, R. A. B. Júnior, R. S. Fucuta, B. M. Ribeiro, and L. M. Miranda, “Physiological and biochemical responses and hydration status in equines after two barrel racing courses,” Pesquisa Veterinária Brasileira, Dec. 2020. doi: 10.1590/1678-5150-pvb-6710. View source
- W. Grünberg, P. Scherpenisse, I. Cohrs, L. Golbeck, P. Dobbelaar, L. V. D. Brink, and I. D. Wijnberg, “Phosphorus content of muscle tissue and muscle function in dairy cows fed a phosphorus-deficient diet during the transition period,” Journal of Dairy Science, Mar. 2019. doi: 10.3168/jds.2018-15727. View source
- E. C. McKenzie, S. J. Valberg, S. Godden, J. D. Pagan, G. P. Carlson, J. M. MacLeay, and F. D. DeLaCorte, “Plasma and urine electrolyte and mineral concentrations in Thoroughbred horses with recurrent exertional rhabdomyolysis after consumption of diets varying in cation-anion balance,” American Journal of Veterinary Research, Jul. 2002. doi: 10.2460/ajvr.2002.63.1053. View source
- L. T. Wesolowski, P. L. Semanchik, and S. H. White-Springer, “Beyond antioxidants: Selenium and skeletal muscle mitochondria,” Frontiers in Veterinary Science, Dec. 2022. doi: 10.3389/fvets.2022.1011159. View source
- I. A. Maksymovych and L. G. Slivinskа, “Metabolic responses in endurance horses at exhausted syndrome,” Ukrainian Journal of Veterinary and Agricultural Sciences, Jul. 2018. doi: 10.32718/ujvas1-1.03. View source
- P. F. Surai, I. I. Kochish, В. Фисинин, and D. Juniper, “Revisiting Oxidative Stress and the Use of Organic Selenium in Dairy Cow Nutrition,” Animals, Jul. 2019. doi: 10.3390/ani9070462. View source
- L. Kokolova, L. Gavrilyeva, Е. Sleptsov, and Е. Pribylykh, “Assessment of the effect of mineral deficiency in the diet of animals,” Genetics and breeding of animals, Jan. 2024. doi: 10.31043/2410-2733-2023-4-39-44. View source
- J. R. Mickelson and S. J. Valberg, “The Genetics of Skeletal Muscle Disorders in Horses,” Annual Review of Animal Biosciences, Sep. 2014. doi: 10.1146/annurev-animal-022114-110653. View source
- M. S. Razzaque and S. J. Wimalawansa, “Minerals and Human Health: From Deficiency to Toxicity,” Nutrients, Jan. 2025. doi: 10.3390/nu17030454. View source
This review is one of 34 in the Foxwatch research corpus. Methodology: PRISMA 2020-style systematic literature review against PubMed, CAB Abstracts, Scopus, Web of Science, and equine veterinary conference proceedings.