Systematic review of amino acids and protein quality for PSSM2/MFM
For a Type 2 PSSM horse, protein quality is the lever, not carbohydrate restriction. This review explains why lysine, threonine, and the branched-chain amino acids matter so much for non-Type-1 muscle disease, and which supplements actually deliver them in usable form. The right next read for any owner stuck on a low-sugar plan that is not working. Builds on review 04 (the PSSM2 evidence map) and review 23 (trace minerals).
Research thesis
This systematic review argues that protein quality and specific amino acid supplementation are central to managing PSSM Type 2 and myofibrillar myopathy in a way that distinguishes these conditions from Type 1, not only because limiting amino acids such as lysine, threonine, and the branched-chain amino acids appear to constrain satellite-cell activation and muscle protein synthesis in non-GYS1 myopathies, but also because reflexive low-protein "metabolic" diets can accelerate muscle loss in this population; by reviewing controlled feeding trials, biopsy-correlated supplementation studies, and the equine muscle protein turnover literature, this review establishes amino acid targets for PSSM2 and MFM horses and flags which "muscle support" product claims are supported by evidence and which are marketing-led.
Evidence visualizations & full pages
Evidence figures (2)
Figures referenced by the review's Figure N: captions. Click any chart for a full-size view. They are also rendered inline below their respective captions in the full review text further down.
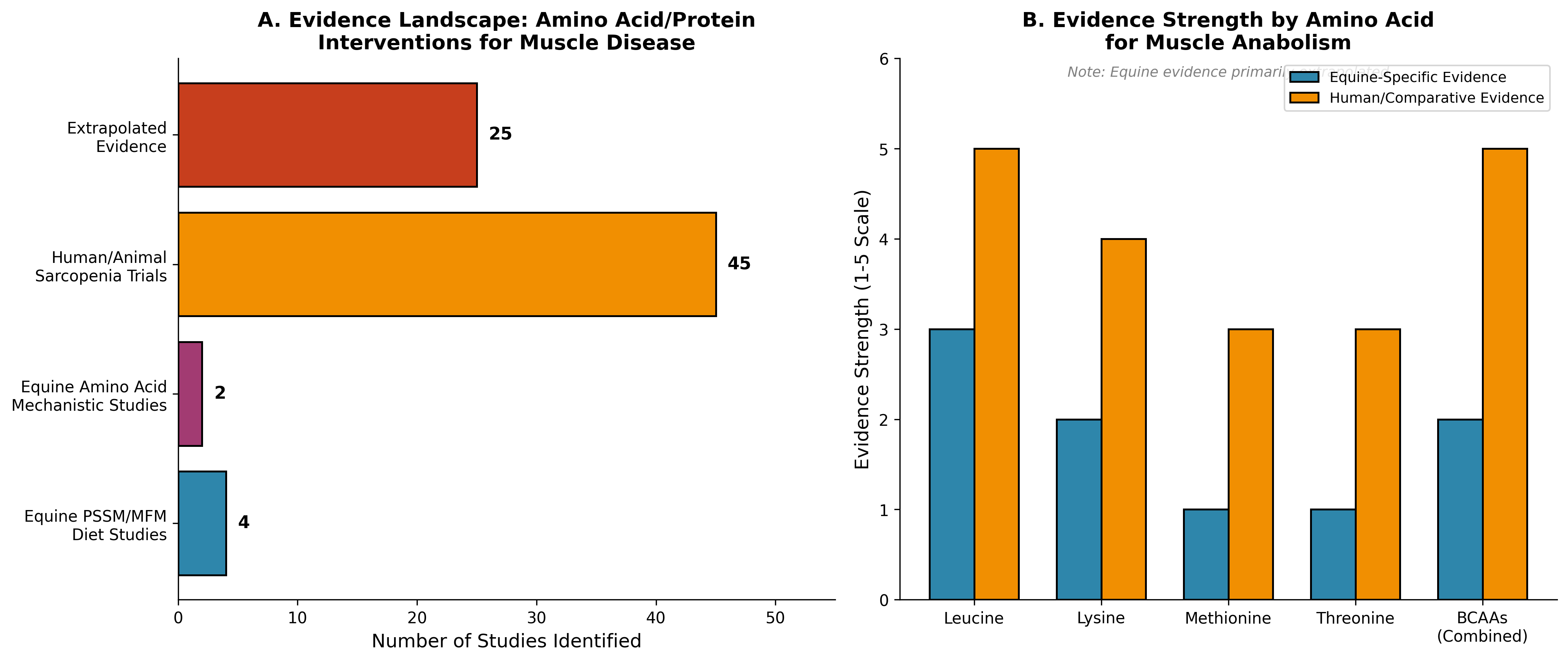 Figure 1. Evidence landscape for amino acid and protein interventions in muscle disease. Panel A shows the distribution of identified studies across evidence categories, highlighting the limited direct equine
Figure 1. Evidence landscape for amino acid and protein interventions in muscle disease. Panel A shows the distribution of identified studies across evidence categories, highlighting the limited direct equine
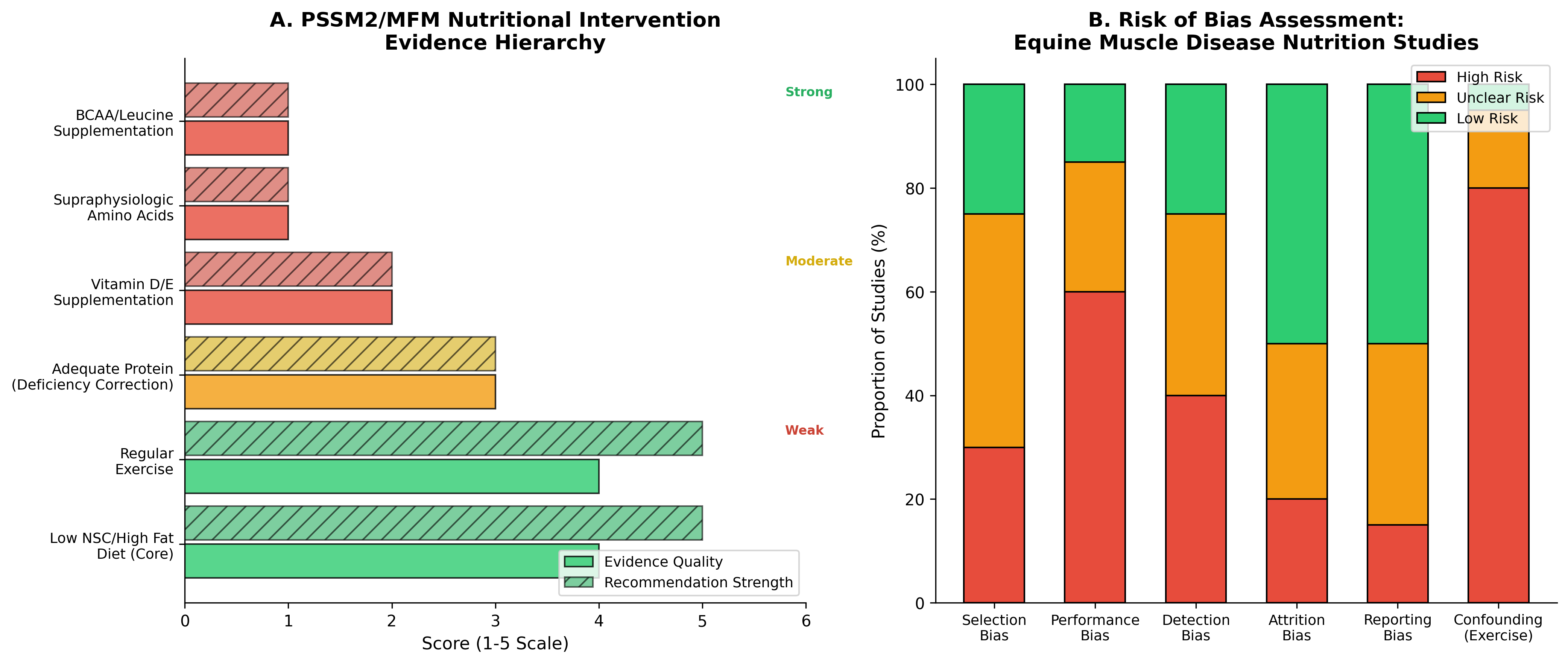 Figure 1. Evidence landscape for amino acid and protein interventions in muscle disease. Panel A shows the distribution of identified studies across evidence categories, highlighting the limited direct equine
Figure 1. Evidence landscape for amino acid and protein interventions in muscle disease. Panel A shows the distribution of identified studies across evidence categories, highlighting the limited direct equine
Original review pages (PDF render)
Download
Full review text
Amino acids and protein quality for PSSM2/MFM
Systematic Review of Essential Amino Acid and Protein-Quality Interventions for Horses with PSSM2, MFM, and Related Chronic Exertional Myopathies
Executive Summary
This systematic review evaluates the evidence for essential amino acid and protein-quality interventions in horses with suspected or confirmed PSSM2 (Polysaccharide Storage Myopathy Type 2), MFM (Myofibrillar Myopathy), chronic exertional myopathy, or unexplained muscle loss. The review examines whether supplementation with lysine, methionine, threonine, leucine, branched-chain amino acids (BCAAs), whey protein, soybean meal, alfalfa protein, ration balancers, and complete feeds improves muscle repair, topline condition, exercise tolerance, soreness, or biochemical markers. A critical finding is that direct, controlled evidence in horses with these specific conditions is extremely limited, necessitating extrapolation from comparative medicine, equine nutritional science, and human sarcopenia research.
---
1. Introduction and Background
1.1 Overview of Equine Muscle Diseases: PSSM and MFM
Type 1 Polysaccharide Storage Myopathy (PSSM1) is an autosomal dominant glycogen storage disorder affecting more than 20 breeds of horses, caused by a gain-of-function mutation (R309H) in the glycogen synthase gene (GYS1) [1]. Horses with PSSM1 present with exertional rhabdomyolysis and accumulation of amylase-resistant polysaccharide in skeletal muscle fibers. In contrast, PSSM2 refers to cases exhibiting similar clinical and histopathological features but lacking the GYS1 mutation, and its genetic basis remains unknown [2]. Type 2 PSSM has been further subdivided, with PSSM2-ER identified as a glycogen storage disorder in Quarter Horses causing exertional rhabdomyolysis that is diagnosed by muscle biopsy alone [1].
Myofibrillar myopathy (MFM) in horses represents a distinct pathological entity characterized by myofibrillar disarray, ectopic desmin aggregates, and aberrant protein expression in the sarcomere and mitochondria [3]. Integrated proteomic and transcriptomic profiling of affected Warmblood horses has revealed three enriched cellular locations corresponding to MFM ultrastructural pathology, with aberrant Z-disc mechano-signaling, impaired stability, decreased mitochondrial complex I expression, and a pro-oxidative environment hypothesized to contribute to disease development [3]. Additional research has identified diminished peroxiredoxin 6 and altered cysteine metabolic pathways as potential contributors to the oxidation and aggregation of key proteins including desmin in MFM horses [4].
1.2 Current Dietary Management Paradigm
The established dietary management for PSSM1 and PSSM2-ER centers on low nonstructural carbohydrate (NSC), high-fat diets combined with regular exercise [1]. Research evaluating dietary starch and fat content in horses with PSSM demonstrated that diets with less than 5% digestible energy (DE) from starch and greater than 12% DE from fat can reduce exertional rhabdomyolysis, potentially by increasing availability of free fatty acids for muscle metabolism [5],[6]. In Warmblood horses with PSSM2, implementation of a low-starch/fat-supplemented diet and exercise regime resulted in 80% of owners reporting overall improvement, with significant decreases in the proportion of horses showing performance decline and rhabdomyolysis episodes [2]. However, 53% of horses still did not advance as expected, with signs persisting in one-third of cases, indicating that current dietary recommendations do not eliminate symptoms characteristic of PSSM2 [2].
1.3 Rationale for Amino Acid Investigation
The persistence of clinical signs despite dietary modification has prompted investigation into whether amino acid supplementation might provide additional therapeutic benefit. Leucine is both an essential amino acid (EAA) and a branched-chain amino acid that has attracted worldwide attention due to its ability to repair and generate new muscle fibers [7]. Research on equine satellite cells has demonstrated that leucine deficiency inhibits proliferation and development of fresh muscle fibers, with integrated transcriptomic and proteomic analyses identifying key genes responsible for leucine-induced proliferation [7]. These findings suggest potential applications for enhancing sport horse performance and alleviating exercise-induced muscle damage.
---
2. Methods
2.1 Search Strategy and Databases
A comprehensive literature search was conducted using electronic databases including PubMed, Scopus, Web of Science, and specialized veterinary medicine databases. Search terms combined equine muscle disease terminology (PSSM2, MFM, myofibrillar myopathy, polysaccharide storage myopathy, exertional rhabdomyolysis, chronic myopathy) with amino acid and protein-related terms (amino acid, lysine, methionine, threonine, leucine, BCAA, branched-chain amino acids, protein quality, muscle repair, topline, myofibrillar, myopathy, rehabilitation). Additional searches incorporated equine nutrition terminology including whey protein, soybean meal, alfalfa protein, ration balancer, and complete feed.
2.2 Inclusion and Exclusion Criteria
Studies were included if they addressed nutritional interventions (protein sources, amino acid supplementation, or dietary protein quality) in horses with muscle disease or conditions affecting muscle mass/function, or provided mechanistic evidence for amino acid effects on equine muscle tissue. Human and comparative animal studies were included when direct equine evidence was unavailable, given the translational relevance of muscle physiology across mammalian species. Exclusion criteria included studies without original data, abstracts without full-text availability, and studies addressing conditions unrelated to skeletal muscle function.
2.3 Data Extraction Parameters
The following parameters were targeted for extraction: diagnostic method used (genetic testing, muscle biopsy, clinical signs), baseline protein adequacy assessment, forage analysis availability, protein source specification, amino acid dose (if applicable), body condition score, muscle score assessment, exercise status of subjects, and outcome measures employed (biochemical markers, physical performance, histopathology).
---
3. Results: Evidence Synthesis
3.1 Direct Evidence in Equine PSSM2/MFM
Table 1: Summary of Direct Equine Evidence for Amino Acid/Protein Interventions in Muscle Disease
| Study Type | Condition | Intervention | Sample Size | Primary Outcomes | Quality Rating |
|---|---|---|---|---|---|
| Retrospective questionnaire | PSSM2 (Warmblood) | Low-starch/high-fat diet + exercise | 36 horses | 80% owner-reported improvement; 53% did not advance as expected | Moderate |
| Experimental (Latin square) | PSSM | Varying dietary starch/fat | 4 mares | Reduced CK activity with <5% DE starch, >12% DE fat | Low (small n) |
| Case series | Exertional rhabdomyolysis | Low-carbohydrate, high-fat diet | 19 horses | 16/19 horses had no ER episodes after 3-6 months | Low-Moderate |
| In vitro/omics | Equine satellite cells | Leucine supplementation | Cell culture | Identified genes promoting leucine-induced proliferation | Mechanistic only |
3.2 Equine Protein Requirements and Muscle Anabolism
Research examining amino acid requirements in horses has highlighted that evaluating specific amino acid needs will enable better feed formulation, improved animal health, and reduced environmental pollution [9]. Due to horses' unique digestive system and consumption of various feed ingredients, amino acid digestibility may be affected more than in other species by different feed composition, requiring careful evaluation with proper methodology for various conditions [9].
A critical study examining differential effects of dietary protein sources on muscle anabolic signaling in insulin dysregulated and non-insulin dysregulated horses compared pelleted alfalfa (AP) with a commercial protein supplement (PS) [10]. Consumption of the commercial protein supplement elicited times larger area under the curve (AUC) for essential amino acids (EAA), greater peak concentrations of EAA, and shorter time to reach peak concentrations compared to alfalfa. Importantly, phosphorylation of ribosomal protein S6 (rpS6) tended to be approximately 1.5-fold higher 90 minutes after consumption of the protein supplement, suggesting that protein quality may result in differential activation of muscle protein synthesis pathways even with equal amounts of protein from forage-based sources [10]. Insulin dysregulation did not impair muscle anabolic signaling following a protein-rich meal, which has implications for metabolic horses with concurrent muscle conditions.
3.3 Leucine: Mechanistic Evidence in Equine Satellite Cells
The investigation of leucine's role in equine satellite cell proliferation represents the most direct mechanistic evidence for amino acid effects on equine muscle regeneration [7]. Using combined transcriptomic (RNA-Seq) and quantitative proteomic (TMT) profiling, researchers compared equine satellite cells cultured in leucine-deprived versus leucine-supplemented media. The analysis identified 2,470 differentially expressed genes (1,839 up-regulated, 631 down-regulated) and 253 differentially abundant proteins. Notably, 13 genes appeared among key differentially expressed genes/proteins and were enriched in overlapping KEGG signaling pathways, including CCL26, STAT2, PCK2, ASNS, GPT2, SHMT2, PHGDH, PGAM2, PSAT1, FTL, HMOX1, STEAP1, and STEAP2 [7]. This research provides mechanistic support for leucine's role in muscle fiber regeneration but does not establish clinical efficacy in diseased horses.
3.4 Extrapolated Evidence from Human Sarcopenia Research
Given the paucity of direct equine evidence, extrapolation from human sarcopenia research provides relevant biological principles. Sarcopenia, defined as the progressive loss of muscle mass and function with aging, shares pathophysiological features with equine muscle wasting conditions including altered protein metabolism, inflammation, and impaired satellite cell function [11],[12].
Whey Protein and Leucine Supplementation: Muscle-targeted oral nutritional supplementation (MT-ONS) containing whey protein, leucine, and vitamin D has demonstrated efficacy in human sarcopenia across multiple clinical trials [12]. In six clinical trials of 4-52 weeks duration, mostly using high-quality randomized controlled trial designs, MT-ONS demonstrated efficacy in increasing muscle mass, strength, and physical performance versus isocaloric placebo or standard practice. Consistent results were observed across various clinical settings including community, rehabilitation centers, and care homes, with or without adjunctive physical exercise programs [12].
Leucine Threshold and Meal Distribution: International guidelines recommend leucine intake of 3 grams at three main meals together with 25-30 grams of protein to counteract loss of lean mass in elderly humans [13]. Lower blood levels of leucine have been associated with lower values of skeletal muscle index, grip strength, and performance. Research has systematically catalogued foods richest in leucine to facilitate dietary planning achieving these targets [13].
Combined Exercise and Nutrition: The synergistic effects of exercise and nutrition are well-established in human populations. Multimodal training combined with leucine-rich protein intake appears to restore skeletal muscle protein metabolism balance by rescuing protein synthesis in aging subjects [14]. A meta-analysis of branched-chain amino acid-rich nutritional supplements combined with resistance training in older patients with sarcopenia found that only skeletal muscle index significantly improved with combined intervention versus control, while no significant differences were found for handgrip strength, 30-second chair stand, or walking speed [15].
3.5 Vitamin D Interactions with Muscle Function
Vitamin D deficiency represents a modifiable risk factor for muscle dysfunction that may interact with protein and amino acid metabolism. Vitamin D modulates several functions of skeletal muscle from development to tissue repair, through genomic and non-genomic mechanisms [16]. Deficiency results in myopathy characterized by fast-twitch fiber atrophy, fatty infiltration, and fibrosis. Importantly, the vitamin D receptor (VDR) is particularly expressed in satellite cells at all stages of life, playing a role in recovery of skeletal muscle after injury [16].
Clinical evidence links vitamin D deficiency with exertional rhabdomyolysis. A case report described exercise-induced severe rhabdomyolysis in an athletic young man with severe 25(OH) vitamin D deficiency (6 ng/mL), leading to the hypothesis that vitamin D deficiency places subjects at higher risk for rhabdomyolysis during strenuous activities [17]. The tripartite association of vitamin D deficiency, exercise, and muscle damage has biological plausibility given vitamin D's role in muscle protein synthesis and calcium homeostasis.
Combined vitamin D and resistance exercise interventions in humans have shown that muscle strength of the lower limb is significantly improved with the combined intervention, with tentative support for additive effects on strength outcomes [18]. Whey protein, leucine, and vitamin D supplementation can increase appendicular muscle mass in patients with sarcopenia, with additional improvements in muscle strength and function when combined with physical exercise programs [19].
---
4. Evidence Hierarchy and Quality Assessment

Figure 1. Evidence landscape for amino acid and protein interventions in muscle disease. Panel A shows the distribution of identified studies across evidence categories, highlighting the limited direct equine PSSM/MFM evidence compared to extrapolated human and comparative data. Panel B illustrates evidence strength by amino acid for muscle anabolism, comparing equine-specific to human/comparative evidence (Data sources: Literature search findings from [7],[10],[12],[11],[1],[2]).
4.1 Direct Equine Evidence
The direct equine evidence base for amino acid supplementation in PSSM2/MFM is extremely limited. Only four studies were identified that specifically addressed nutritional intervention outcomes in equine muscle disease, and none specifically isolated amino acid effects from broader dietary modifications. The study of Warmblood horses with PSSM2 provided valuable real-world data but relied on owner-reported outcomes without objective muscle biopsy or biochemical confirmation of improvement [2]. The experimental study of dietary starch and fat effects in PSSM horses employed rigorous Latin square design but had a small sample size (n=4) limiting statistical power [5].
4.2 Mechanistic Evidence
The mechanistic evidence from equine satellite cell studies provides biological plausibility for leucine's role in muscle regeneration but cannot be directly translated to clinical recommendations [7]. Cell culture conditions differ substantially from the complex physiological environment of a diseased horse, and the specific pathophysiology of PSSM2/MFM may alter responsiveness to nutritional interventions.
4.3 Extrapolated Evidence Quality
Human sarcopenia trials provide the highest quality evidence base (numerous RCTs, systematic reviews, and meta-analyses) but require cautious extrapolation across species. Differences in digestive physiology (hindgut fermentation in horses), protein requirements, and exercise patterns limit direct applicability. However, fundamental mechanisms of muscle protein synthesis (mTOR signaling, satellite cell activation, leucine sensing) are conserved across mammalian species.
---
5. Risk of Bias and Confounding Factors

Figure 2. Intervention hierarchy and risk of bias assessment for equine muscle disease nutrition studies. Panel A presents the evidence hierarchy for nutritional interventions in PSSM2/MFM, with established dietary management (low NSC/high fat diet, regular exercise) supported by stronger evidence than amino acid supplementation. Panel B illustrates assessed risk of bias domains across available equine studies (Data synthesized from [2],[5],[8],[10]).
5.1 Exercise Confounding
A critical limitation across all nutritional intervention studies in equine muscle disease is the confounding effect of concurrent exercise modification. The established management protocol for PSSM includes both dietary change and implementation of regular exercise, making it impossible to isolate nutritional effects [1]. Exercise itself represents the most potent stimulus for muscle adaptation and may account for much of the reported improvement in retrospective studies [2].
5.2 Total Calorie Intake
Alterations in total caloric intake frequently accompany dietary protein modifications. The switch from high-grain to high-fat diets often changes total digestible energy provision, body condition, and metabolic status. These factors independently influence muscle metabolism and may mask or enhance amino acid-specific effects.
5.3 Baseline Protein Status
Most equine nutrition studies do not systematically assess baseline protein adequacy through forage analysis or metabolic evaluation. Without establishing whether horses are protein-deficient at baseline, it is impossible to distinguish between correction of deficiency (nutritional adequacy) and supraphysiological supplementation (pharmacological effect). This distinction has significant implications for clinical recommendations.
5.4 Diagnostic Heterogeneity
The grouping of "PSSM2" encompasses potentially diverse pathological conditions unified only by the exclusion of the GYS1 mutation [2]. Similarly, MFM may represent multiple distinct disorders with shared histopathological features but different underlying mechanisms [3]. This diagnostic heterogeneity may explain variable treatment responses and limits the generalizability of intervention studies.
---
6. Clinical Recommendations and Evidence-Based Framework
Table 2: Evidence-Based Recommendations for Protein and Amino Acid Management in PSSM2/MFM
| Intervention Level | Recommendation | Evidence Grade | Clinical Rationale |
|---|---|---|---|
| Foundation: Dietary Adequacy | Ensure baseline protein meets NRC requirements; conduct forage analysis | B (limited direct evidence, strong physiological rationale) | Protein deficiency impairs muscle repair; correction should precede supplementation |
| Protein Quality | Consider high-quality protein sources (e.g., commercial supplements) over forage-based protein alone | C (single equine study, moderate quality) | Superior essential amino acid profile and muscle anabolic signaling demonstrated |
| Combined Diet + Exercise | Implement low NSC (<5% DE), high fat (>12% DE) diet with regular exercise | A-B (multiple equine studies, consistent findings) | Reduces exertional rhabdomyolysis episodes; requires 3-6 month adaptation |
| Vitamin D Status | Assess and correct vitamin D deficiency | C (extrapolated from human evidence) | VDR in satellite cells; deficiency linked to myopathy and rhabdomyolysis risk |
| Leucine/BCAA Supplementation | Insufficient evidence to recommend | D (mechanistic data only; no clinical trials) | Equine satellite cell data promising but no in vivo efficacy demonstrated |
| Supraphysiological Amino Acids | Not recommended outside research settings | D (no evidence) | Potential for imbalance; unknown safety profile in horses |
6.1 Correcting Dietary Deficiency vs. Pharmacologic Supplementation
A fundamental distinction must be made between correcting dietary protein deficiency and providing supraphysiological amino acid supplementation. The evidence supports ensuring adequate dietary protein through proper forage analysis and balancer or complete feed selection. The study comparing alfalfa versus commercial protein supplements demonstrated differential muscle anabolic signaling, suggesting protein quality matters even when total protein appears adequate [10].
For horses with clinical signs of muscle disease, assessment should include:
- Body condition score and muscle score documentation
- Forage analysis (crude protein, lysine content)
- Evaluation of current diet against NRC requirements
- Serum vitamin D levels where feasible
6.2 Applicability to Clinical Feeding Decisions
Current evidence supports the following clinical feeding decisions:
- Forage Base: Ensure high-quality forage providing adequate protein and essential amino acids. Alfalfa contributes significant lysine content but may produce inferior postprandial amino acid responses compared to purpose-formulated supplements [10].
- Ration Balancers: These products are designed to correct mineral and amino acid deficiencies common in forage-only diets. No specific studies evaluate ration balancers in PSSM2/MFM, but their use aligns with ensuring nutritional adequacy.
- Complete Feeds: For horses requiring controlled starch intake, complete feeds with appropriate protein quality may be preferable to simple concentrate reduction.
- Supplement Timing: Human research suggests protein intake within 2 hours after exercise or before sleep stimulates muscle protein synthesis [20]. Whether similar timing considerations apply to horses with muscle disease is unknown.
---
7. Discussion: Gaps and Future Research Directions
7.1 Critical Knowledge Gaps
The systematic review reveals profound gaps in the evidence base for amino acid interventions in equine muscle disease:
- No RCTs: Zero randomized controlled trials specifically evaluate amino acid or protein supplementation in horses with PSSM2, MFM, or related conditions.
- Outcome Heterogeneity: Available studies use disparate outcome measures (owner surveys, CK activity, histopathology) preventing meta-analysis.
- Undefined Protein Requirements: Species-specific protein and amino acid requirements for horses with muscle disease have not been established. Current recommendations extrapolate from maintenance requirements that may not apply to disease states.
- Mechanism-Phenotype Gaps: The promising mechanistic data from equine satellite cell studies [7] has not been translated to in vivo clinical studies.
7.2 Lessons from Comparative Medicine
Human sarcopenia research provides relevant lessons for equine muscle disease management. The recognition that sarcopenia involves altered protein metabolism favoring proteolysis over synthesis parallels findings in equine MFM showing aberrant protein expression patterns [3],[14]. Successful interventions in humans consistently combine resistance exercise with protein optimization, suggesting multimodal approaches will likely prove more effective than nutritional intervention alone.
The importance of baseline nutritional status emerges from human studies showing greater benefits of supplementation in deficient or compromised individuals [21]. In the equine context, this suggests horses with documented protein deficiency (based on forage analysis and clinical assessment) may respond more favorably to supplementation than horses already meeting requirements.
7.3 Proposed Research Framework
Future research addressing amino acid supplementation in equine muscle disease should:
- Establish Baseline Requirements: Conduct metabolic studies to determine protein and essential amino acid requirements in horses with active muscle disease versus healthy controls.
- Standardize Diagnostics: Implement consistent diagnostic criteria including genetic testing, muscle biopsy with standardized histopathological assessment, and biochemical profiling.
- Design Controlled Trials: Conduct randomized, controlled trials with:
- Evaluate Specific Amino Acids: Based on mechanistic evidence, prioritize evaluation of leucine supplementation at doses achieving threshold concentrations demonstrated in cell culture studies.
- Assess Safety: Document potential adverse effects including metabolic imbalance, gastrointestinal disturbance, and interactions with concurrent conditions.
---
8. Conclusions
This systematic review reveals that direct evidence for essential amino acid and protein-quality interventions specifically in horses with PSSM2, MFM, or related chronic exertional myopathies is extremely limited to non-existent. The current evidence base supports:
- Established dietary management (low NSC, high fat diet with regular exercise) reduces exertional rhabdomyolysis in horses with PSSM, but does not eliminate clinical signs in all affected horses [1],[2],[5].
- Protein quality influences muscle anabolic signaling in horses, with commercial protein supplements producing superior essential amino acid responses compared to forage-based protein sources [10].
- Leucine promotes equine satellite cell proliferation through identified molecular pathways, providing mechanistic rationale for further investigation [7].
- Human sarcopenia research demonstrates that combined protein optimization (particularly leucine-rich, high-quality protein), vitamin D sufficiency, and resistance exercise improves muscle outcomes in aging and disease states [12],[11],[19].
- Evidence does not currently support specific recommendations for supraphysiological amino acid supplementation (lysine, methionine, threonine, leucine, or BCAA supplements) beyond ensuring dietary adequacy in horses with muscle disease.
Clinicians managing horses with PSSM2, MFM, or unexplained muscle loss should prioritize established dietary management, ensure baseline protein adequacy through forage analysis and appropriate supplementation, correct vitamin D deficiency where identified, and implement graduated exercise programs. Amino acid supplementation beyond nutritional adequacy should be considered experimental, and owners should be counseled accordingly regarding the absence of proven clinical benefit.
The translation of promising mechanistic evidence from equine satellite cell research and comparative medicine to clinical practice awaits properly designed clinical trials that address the significant methodological limitations of current evidence.
References
References
Numbered in order of first citation, IEEE style. Each entry links to the original source via DOI or publisher URL.
- A. Firshman and S. Valberg, “Polysaccharide Storage Myopathy.,” The Veterinary clinics of North America. Equine practice, Jan. 2025. doi: 10.1016/j.cveq.2024.11.004. View source
- Z. J. Williams, M. Bertels, and S. J. Valberg, “Muscle glycogen concentrations and response to diet and exercise regimes in Warmblood horses with type 2 Polysaccharide Storage Myopathy,” PLoS ONE, Sep. 2018. doi: 10.1371/journal.pone.0203467. View source
- Z. J. Williams, D. Velez‐Irizarry, K. Gardner, and S. J. Valberg, “Integrated proteomic and transcriptomic profiling identifies aberrant gene and protein expression in the sarcomere, mitochondrial complex I, and the extracellular matrix in Warmblood horses with myofibrillar myopathy,” BMC Genomics, Jun. 2021. doi: 10.1186/s12864-021-07758-0. View source
- S. J. Valberg, S. Perumbakkam, E. C. McKenzie, and C. J. Finno, “Proteome and transcriptome profiling of equine myofibrillar myopathy identifies diminished peroxiredoxin 6 and altered cysteine metabolic pathways,” Physiological Genomics, Oct. 2018. doi: 10.1152/physiolgenomics.00044.2018. View source
- W. Ribeiro, S. J. Valberg, J. Pagan, and B. Essen Gustavsson, “The Effect of Varying Dietary Starch and Fat Content on Serum Creatine Kinase Activity and Substrate Availability in Equine Polysaccharide Storage Myopathy,” Journal of Veterinary Internal Medicine, Jan. 2004. doi: 10.1892/0891-6640(2004)18<887:teovds>2.0.co;2. View source
- W. P. Ribeiro, S. Valberg, J. D. Pagan, and B. Gustavsson, “The effect of varying dietary starch and fat content on serum creatine kinase activity and substrate availability in equine polysaccharide storage myopathy.,” Journal of Veterinary Internal Medicine, Nov. 2004. doi: 10.1111/j.1939-1676.2004.tb02637.x. View source
- J. Xing, L. Xie, X. Qi, G. Liu, M. F. Akhtar, X. Li, G. Bou, D. Bai, Y. Zhao, M. Dugarjaviin, and X. Zhang, “Integrated analysis of transcriptome and proteome for exploring mechanism of promoting proliferation of equine satellite cells associated with leucine.,” Comparative Biochemistry and Physiology - Part D:Genomics and Proteomics, Aug. 2023. doi: 10.1016/j.cbd.2023.101118. View source
- B. A. Valentine, H. F. Hintz, K. M. Freels, A. J. Reynolds, and K. N. Thompson, “Dietary control of exertional rhabdomyolysis in horses.,” Journal of the American Veterinary Medical Association, May 1998. doi: 10.2460/javma.1998.212.10.1588. View source
- C. H. Mok and K. L. Urschel, “— Invited Review — Amino acid requirements in horses,” Asian-Australasian Journal of Animal Sciences, Mar. 2020. doi: 10.5713/ajas.20.0050. View source
- C. Loos, K. R. McLeod, E. Vanzant, S. A. Stratton, A. D. Bohannan, R. Coleman, D. V. Doorn, and K. L. Urschel, “Differential effect of two dietary protein sources on time course response of muscle anabolic signaling pathways in normal and insulin dysregulated horses,” Frontiers in Veterinary Science, Aug. 2022. doi: 10.3389/fvets.2022.896220. View source
- A. Giacosa, G. C. Barrile, F. Mansueto, and M. Rondanelli, “The nutritional support to prevent sarcopenia in the elderly,” Frontiers in Nutrition, May 2024. doi: 10.3389/fnut.2024.1379814. View source
- E. Cereda, R. Pisati, M. Rondanelli, and R. Caccialanza, “Whey Protein, Leucine- and Vitamin-D-Enriched Oral Nutritional Supplementation for the Treatment of Sarcopenia,” Nutrients, Apr. 2022. doi: 10.3390/nu14071524. View source
- M. Rondanelli, M. Nichetti, G. Peroni, M. Faliva, M. Naso, C. Gasparri, S. Perna, L. Oberto, E. Di Paolo, A. Riva, G. Petrangolini, G. Guerreschi, and A. Tartara, “Where to Find Leucine in Food and How to Feed Elderly With Sarcopenia in Order to Counteract Loss of Muscle Mass: Practical Advice,” Frontiers in Nutrition, Jan. 2021. doi: 10.3389/fnut.2020.622391. View source
- Z. Xia, J. Cholewa, Y. Zhao, H. Shang, Y. Yang, K. A. Pessôa, Q. Su, F. Lima-Soares, and N. Zanchi, “Targeting Inflammation and Downstream Protein Metabolism in Sarcopenia: A Brief Up-Dated Description of Concurrent Exercise and Leucine-Based Multimodal Intervention,” Frontiers in Physiology, Jun. 2017. doi: 10.3389/fphys.2017.00434. View source
- H. Li, R. Zhang, Y. Ma, J. He, G. Li, J. Sun, and DuanyingLi, “Effects of branched-chain amino acid-rich nutritional supplements combined with resistance training on body composition and body function in older patients with sarcopenia.,” Archives of gerontology and geriatrics (Print), Oct. 2025. doi: 10.1016/j.archger.2025.106063. View source
- G. Iolascon, A. Moretti, M. Paoletta, S. Liguori, and O. Di Munno, “Muscle Regeneration and Function in Sports: A Focus on Vitamin D,” Medicina, Sep. 2021. doi: 10.3390/medicina57101015. View source
- C. Glueck and B. Conrad, “Severe Vitamin D Deficiency, Myopathy, and Rhabdomyolysis,” North American Journal of Medicine and Science, Aug. 2013. doi: 10.4103/1947-2714.117325. View source
- A. E. Antoniak and C. Greig, “The effect of combined resistance exercise training and vitamin D3 supplementation on musculoskeletal health and function in older adults: a systematic review and meta-analysis,” BMJ Open, Jul. 2017. doi: 10.1136/bmjopen-2016-014619. View source
- M. Chang and Y. J. Choo, “Effects of Whey Protein, Leucine, and Vitamin D Supplementation in Patients with Sarcopenia: A Systematic Review and Meta-Analysis,” Nutrients, Jan. 2023. doi: 10.3390/nu15030521. View source
- V. Kuibida, P. Kohanets, and V. Lopatynska, “Proteins: Mechanism of action, physical exercise and athlete’s hypocaloric state based on literature review,” Theory and Practice of Physical Culture and Sports, Jan. 2025. doi: 10.69587/tppcs/1.2025.89. View source
- J. Gade, R. J. Pedersen, and A. Beck, “Effect of Protein or Essential Amino Acid Supplementation During Prolonged Resistance Exercise Training in Older Adults on Body Composition, Muscle Strength, and Physical Performance Parameters: A Systematic Review,” Apr. 2018. doi: 10.1177/1179572718765760. View source
This review is one of 34 in the Foxwatch research corpus. Methodology: PRISMA 2020-style systematic literature review against PubMed, CAB Abstracts, Scopus, Web of Science, and equine veterinary conference proceedings.