Systematic review of biomarkers for monitoring dietary response in PSSM
How do you actually know the diet is working? This review identifies the small panel of labs that catches a failing protocol before the next tying-up episode: creatine kinase, AST, glutathione peroxidase, and serum vitamin E. Drives the recheck schedule and reference ranges in the feeding protocol. Read with review 01 (foundation) and review 24 (how strong this evidence really is).
Research thesis
This systematic review argues that monitoring a PSSM horse on a dietary protocol must rest on a defined multi-marker panel, not on creatine kinase alone, not only because CK and AST drift independently with exercise, transport, and concurrent disease, but also because GSH-Px, serum vitamin E, insulin, and clinical-rating scales each capture a different aspect of glycogen storage and antioxidant status; by reviewing the longitudinal-monitoring and reference-interval literature in confirmed PSSM1 and PSSM2 cohorts, this review validates a cost-conscious biomarker panel for routine recheck use and identifies the multi-year trajectory patterns that distinguish a stable horse from one whose protocol is failing.
Evidence visualizations & full pages
Evidence figures (3)
Figures referenced by the review's Figure N: captions. Click any chart for a full-size view. They are also rendered inline below their respective captions in the full review text further down.
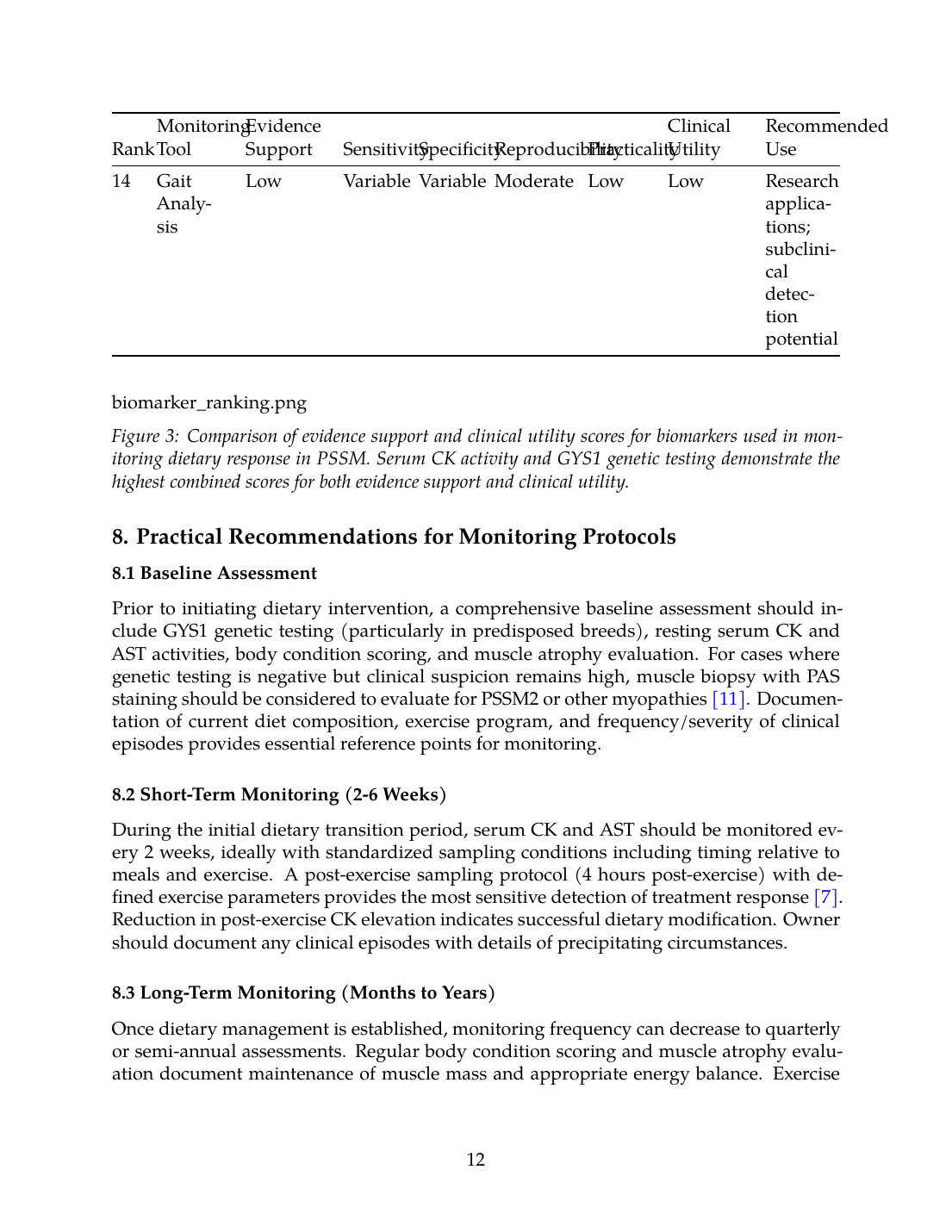
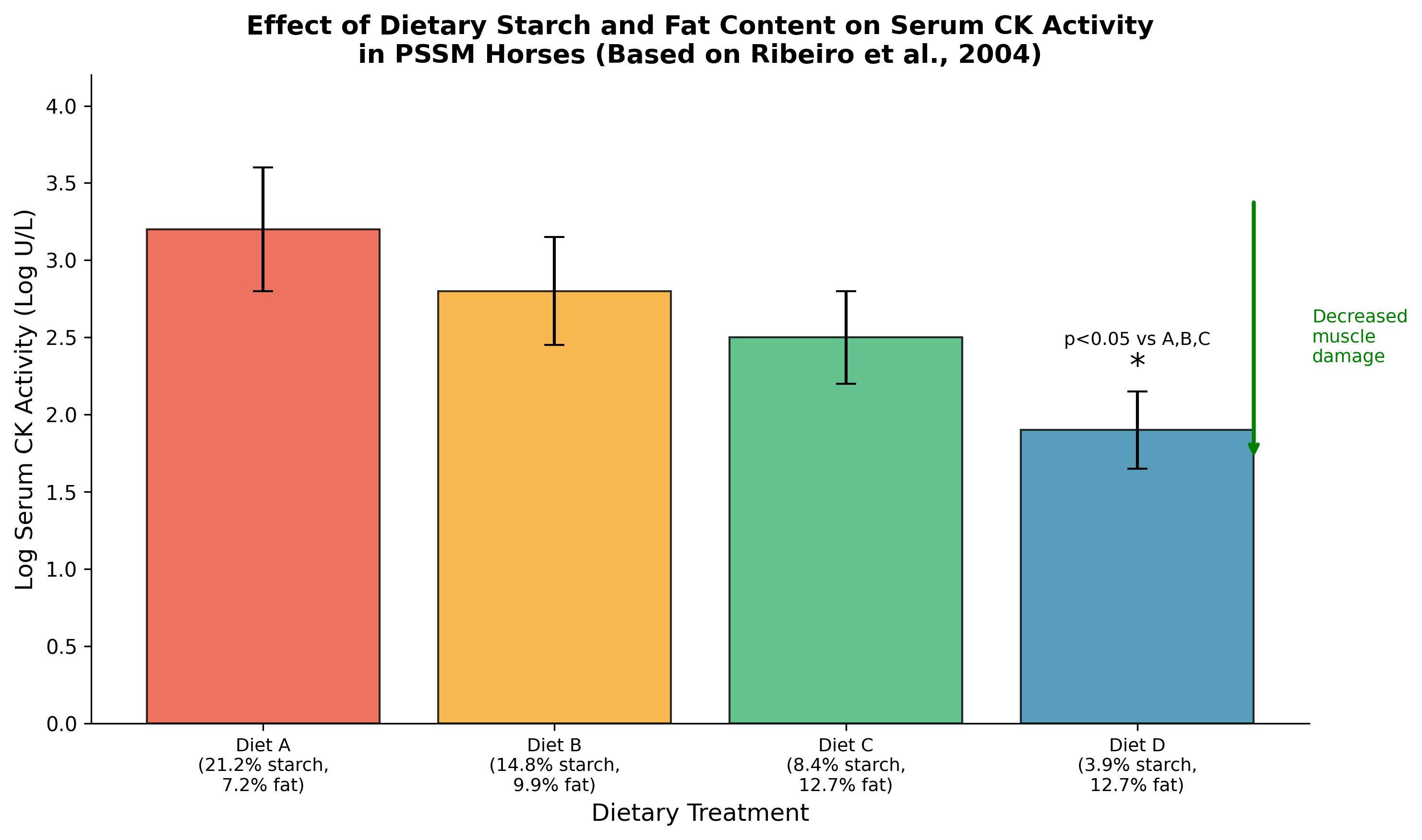 Figure 1: Effect of dietary starch and fat content on serum CK activity in PSSM horses. Diets with <5% DE from starch and >12% DE from fat (Diet D) significantly reduced CK activity compared to higher starch d
Figure 1: Effect of dietary starch and fat content on serum CK activity in PSSM horses. Diets with <5% DE from starch and >12% DE from fat (Diet D) significantly reduced CK activity compared to higher starch d
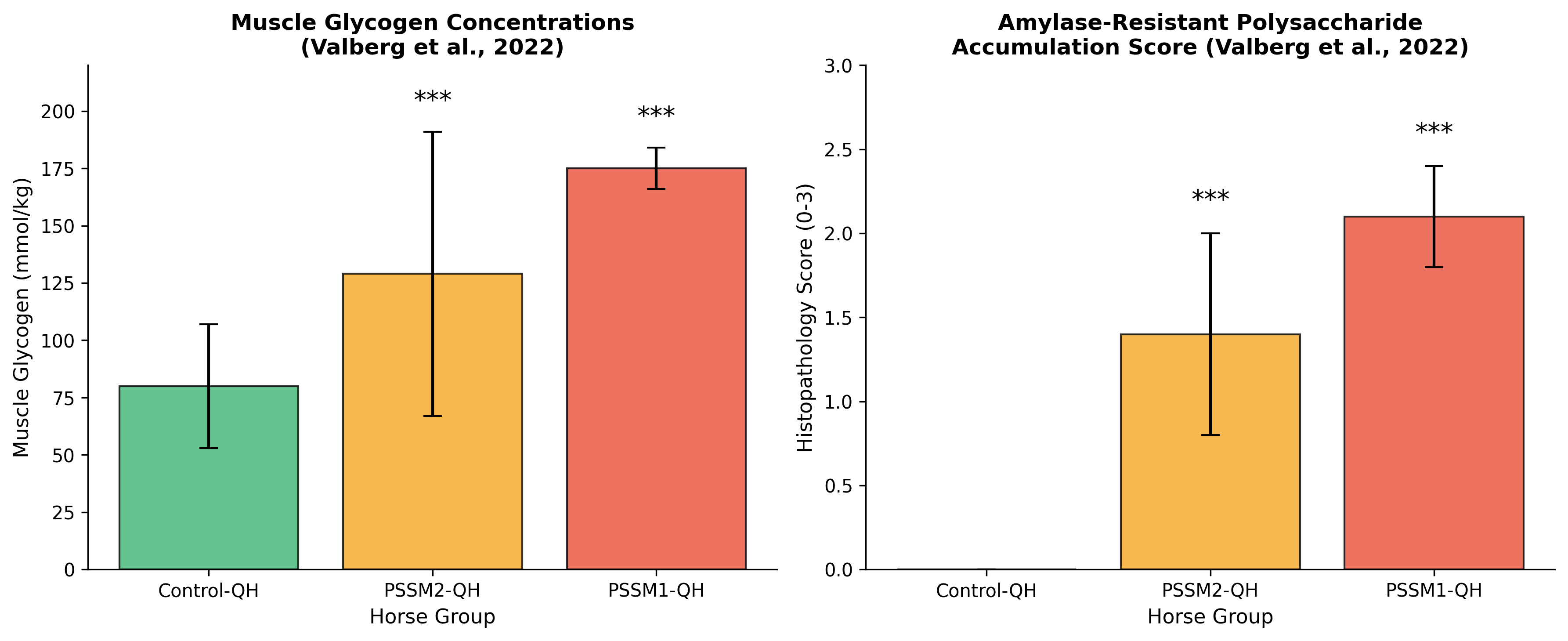 Figure 1: Effect of dietary starch and fat content on serum CK activity in PSSM horses. Diets with <5% DE from starch and >12% DE from fat (Diet D) significantly reduced CK activity compared to higher starch d
Figure 1: Effect of dietary starch and fat content on serum CK activity in PSSM horses. Diets with <5% DE from starch and >12% DE from fat (Diet D) significantly reduced CK activity compared to higher starch d
 Figure 1: Effect of dietary starch and fat content on serum CK activity in PSSM horses. Diets with <5% DE from starch and >12% DE from fat (Diet D) significantly reduced CK activity compared to higher starch d
Figure 1: Effect of dietary starch and fat content on serum CK activity in PSSM horses. Diets with <5% DE from starch and >12% DE from fat (Diet D) significantly reduced CK activity compared to higher starch d
Original review pages (PDF render)
Download
Full review text
Biomarkers for monitoring dietary response in PSSM
Systematic Review of Biomarkers for Monitoring Dietary Response in Polysaccharide Storage Myopathy
1. Introduction and Background
1.1 Overview of Polysaccharide Storage Myopathy Types
Polysaccharide storage myopathy (PSSM) represents a significant group of equine glycogenoses characterized by abnormal accumulation of glycogen and polysaccharides in skeletal muscle fibers [1]. Type 1 PSSM (PSSM1) is caused by a well-characterized R309H gain-of-function mutation in the glycogen synthase 1 gene (GYS1), leading to the accumulation of amylase-resistant alpha-crystalline polysaccharide inclusions within skeletal muscle [2]. This mutation has been identified across numerous breeds worldwide, including Quarter Horses, Appaloosas, Warmbloods, Percherons, Belgians, and various draft breeds, with prevalence rates varying considerably; for example, Percheron horses demonstrate a 90% carrier rate while Russian heavy draft horses show approximately 50% prevalence [3].
Type 2 PSSM (PSSM2) in Quarter Horses has been characterized as a novel glycogen storage disorder distinct from PSSM1, exhibiting intermediate histopathological scores and glycogen concentrations between PSSM1 and control horses. Importantly, whole genome sequencing of candidate genes associated with muscle glycogenoses in other species has failed to identify the causative mutation for PSSM2, indicating that this condition represents a unique glycogenosis not attributable to mutations in genes currently known to cause muscle glycogenoses [4]. Additionally, commercial genetic tests for variants in MYOT, FLNC, and MYOZ3 genes have shown no association with histopathologically-diagnosed PSSM2 in Quarter Horses, with these variants being present in 57% of healthy control horses [5].
1.2 Pathophysiology and Dietary Implications
The pathophysiology of PSSM involves complex interactions between glycogen metabolism, mitochondrial function, and oxidative stress. Gene expression profiling in affected horses has revealed upregulation of inflammatory markers (particularly IL18), inhibition of glycogenesis via glycogen synthase kinase-3 (GSK3β), chronic hypoxia of affected muscles, and significant mitochondrial dysfunction with decreased numbers and structural disorders [6]. These findings provide the mechanistic basis for dietary interventions targeting both substrate availability and metabolic pathways.
Dietary management remains the cornerstone of PSSM treatment, with the primary goal being reduction of dietary starch while increasing fat as an alternative energy source. The landmark study by Ribeiro et al. (2004) demonstrated that diets providing less than 5% digestible energy (DE) from starch and greater than 12% DE from fat significantly reduced serum creatine kinase (CK) activity in PSSM horses, potentially by increasing the availability of free fatty acids for muscle metabolism [7]. This dietary approach has shown benefits across PSSM subtypes, with exertional rhabdomyolysis in PSSM2-QH improving with regular exercise combined with a low starch/fat-supplemented diet [4].
2. Serum Muscle Enzyme Biomarkers
2.1 Creatine Kinase (CK)
Serum creatine kinase activity represents the most extensively validated biomarker for monitoring dietary response in PSSM. The sensitivity of CK to muscle damage makes it an ideal marker for detecting both subclinical myopathy and response to nutritional interventions. In the seminal dietary intervention study, log CK activity was significantly higher with diets containing 21.2%, 14.8%, and 8.4% DE from starch compared to the diet providing only 3.9% DE from starch, demonstrating a clear dose-response relationship between dietary starch content and muscle damage markers [7].
The relationship between CK activity and disease severity has been further elucidated through genotype-phenotype correlation studies. Resting CK activities demonstrate significant differences between PSSM1 genotypes: homozygous affected horses show median CK activities of 364 U/L (interquartile range 332-764), compared to 301 U/L (222-377) for heterozygotes and 260 U/L (216-320) for controls [8]. These baseline differences provide important context for interpreting changes following dietary intervention and establish reference ranges for monitoring therapeutic response.
Optimal sampling protocols for CK measurement require standardization of exercise conditions and timing. The post-exercise sampling window of 4 hours has been utilized in controlled dietary studies, with samples collected following standardized exercise protocols [7]. The influence of hormonal factors on CK activity has also been documented; estrous cycle stage affects resting and post-exercise AST activity but not CK activity in mares susceptible to exertional rhabdomyolysis [9]. This suggests that CK may be more reliable than AST for serial monitoring across the reproductive cycle.
2.2 Aspartate Aminotransferase (AST) and Lactate Dehydrogenase (LDH)
Aspartate aminotransferase provides complementary information to CK for monitoring PSSM, particularly given its longer half-life which allows detection of muscle damage occurring over extended periods. In PSSM1 horses, mean resting AST activities were 502±116 U/L for homozygotes, 357±92 U/L for heterozygotes, and 311±64 U/L for controls, with significant differences between genotypes [8]. Importantly, resting plasma AST activity demonstrated significant correlations with underlying muscle pathology, specifically the severity of subsarcolemmal vacuolation (rho=0.816, P=0.01) and cytoplasmic inclusions (rho=0.766, P=0.01), providing direct linkage between this biomarker and histopathological disease burden.
Lactate dehydrogenase, while less specific for skeletal muscle, has been evaluated as part of comprehensive metabolic panels in PSSM horses. In ultra-endurance exercise settings, LDH shows substantial post-exercise elevation with median increases of 311.20±164.4% compared to pre-exercise values [10]. However, the tissue non-specificity of LDH and the co-measurement of this enzyme in muscle biopsy samples for assessment of oxidative capacity rather than as a damage marker [7] limits its utility as a standalone monitoring tool for dietary response.

Figure 1: Effect of dietary starch and fat content on serum CK activity in PSSM horses. Diets with <5% DE from starch and >12% DE from fat (Diet D) significantly reduced CK activity compared to higher starch diets. Data adapted from Ribeiro et al. (2004).
3. Genetic and Histopathological Diagnostic Methods
3.1 GYS1 Genetic Testing
Genetic testing for the GYS1 R309H mutation represents the gold standard for definitive diagnosis of PSSM1 and provides essential baseline information for interpreting subsequent biomarker responses to dietary intervention. The mutation has been confirmed across multiple geographic populations and breeds. In UK horses with histories of exertional rhabdomyolysis, 21% (10/47) carried the GYS1 mutation, with the mutation exclusively found in horses with grade 2 PSSM on histopathology; notably, 65% of horses with grade 2 (amylase-resistant) PSSM tested positive for the mutation [11].
The genotype demonstrates incomplete dominance for disease phenotype, with homozygous affected horses showing more severe subclinical disease than heterozygotes as evidenced by higher resting enzyme activities and more severe histopathological changes [8]. This genotype-phenotype relationship has practical implications for dietary monitoring, as homozygous horses may require more aggressive nutritional intervention and demonstrate greater magnitude of improvement with appropriate dietary management.
Breed prevalence surveys have revealed substantial variation in GYS1 mutation frequency, which informs population-level screening strategies. The mutation was found in Bashkirskaya horses (3.2%), Buryatskaya (10%), Vyatskaya (9.5%), Soviet heavy draft (30%), Russian heavy draft (49.9%), and Percheronskaya (90%), while Donskaya, Thoroughbred, and Orlov trotter horses showed no PSSM1 defect [3]. This epidemiological data supports targeted genetic screening in high-prevalence breeds and emphasizes that recurrent exertional rhabdomyolysis in certain breeds (notably Standardbreds) has different genetic bases, as all susceptible Standardbreds tested negative for the GYS1 mutation [12].
3.2 Muscle Biopsy and Histopathology
Muscle biopsy with histochemical analysis provides definitive diagnostic confirmation and enables quantification of pathological changes that may not be reflected in serum biomarkers. The diagnostic approach relies on periodic acid-Schiff (PAS) staining with and without amylase digestion to identify abnormal polysaccharide accumulation. In PSSM muscles, histological examination reveals PAS-positive amylase-resistant abnormal polysaccharides, inflammation, necrosis, lipomatosis, and active fiber regeneration [6]. Ultrastructural evaluation demonstrates decreased mitochondrial numbers and structural disorders, with extensive polysaccharide accumulation displacing and partially replacing mitochondria and myofibrils.
Quantitative histopathological scoring provides objective assessment of disease severity and response to treatment. The scoring system (0-3) for amylase-resistant polysaccharide demonstrated significant differences between control, PSSM2, and PSSM1 horses: PSSM2-QH scored 1.4±0.6, PSSM1-QH scored 2.1±0.3, and control-QH scored 0±0, indicating intermediate pathology in PSSM2 [4]. These quantitative measures enable longitudinal monitoring of tissue-level response to dietary intervention, though the invasive nature of biopsy limits frequent serial sampling.
The correlation between GYS1 genotype and histopathological findings has been investigated in endemic populations. Studies in Transylvanian equine populations demonstrated significant association between the number of rhabdomyolysis episodes and GYS1 mutation (P=0.03) and positive amylase-resistant PAS staining (P=0.01), though the correlation between mutation status and PAS staining was weak (P=0.06), suggesting additional factors influence phenotypic expression [13].
3.3 Muscle Glycogen Concentration
Direct measurement of muscle glycogen concentration provides pathophysiologically relevant assessment of the primary metabolic abnormality in PSSM. Glycogen concentrations demonstrate significant differences between affected groups: PSSM2-QH (129±62 mmol/kg), PSSM1-QH (175±9 mmol/kg), and control-QH (80±27 mmol/kg), with all pairwise comparisons reaching statistical significance [4]. These measurements indicate that both PSSM1 and PSSM2 horses accumulate excess muscle glycogen, though the magnitude and variability differ between subtypes.
Importantly, dietary intervention studies have shown that muscle glycogen and abnormal polysaccharide content remain elevated regardless of dietary starch and fat manipulation, suggesting that these tissue parameters may not be responsive to short-term nutritional management even when clinical signs and serum markers improve [7]. This finding has significant implications for monitoring: while serum CK activity responds to dietary changes, tissue glycogen accumulation appears to be a more permanent feature of the disease that may not serve as a practical marker for assessing short-term dietary response.

Figure 2: Muscle glycogen concentrations (left) and amylase-resistant polysaccharide histopathology scores (right) across horse groups. Both PSSM1 and PSSM2 show significantly elevated values compared to controls (*p<0.001). Data from Valberg et al. (2022).
4. Metabolic Response Markers
4.1 Glucose and Insulin Responses
Postprandial glucose and insulin responses represent important metabolic markers for assessing the effectiveness of low-starch dietary interventions in PSSM. The mechanistic rationale centers on reducing the glycemic stimulus that drives muscle glycogen synthesis via insulin signaling. In controlled dietary studies, postprandial insulin and glucose responses were significantly higher following high-starch diets compared to low-starch alternatives [7]. Daily insulin concentrations were elevated and free fatty acid (FFA) availability reduced on high-starch diets, while glucose varied only slightly, suggesting insulin as the more sensitive metabolic indicator.
The interaction between fitness level and metabolic response to diet has been characterized in Thoroughbreds with recurrent exertional rhabdomyolysis (RER). Fit horses fed high non-structural carbohydrate (NSC) diets demonstrated higher postprandial glucose, insulin, and insulin:glucose ratios compared to unfit horses, suggesting that training-induced metabolic adaptations may amplify the metabolic response to dietary starch [14]. Critically, these heightened responses were not observed when horses were fed low-starch, high-fat diets, supporting the use of such diets to mitigate metabolic triggers for rhabdomyolysis.
Practical assessment of glucose and insulin dynamics can be accomplished using oral tolerance testing, similar to diagnostic approaches for equine metabolic syndrome. The oral sugar test has been established as reliable for detecting exaggerated postprandial insulin responses in horses at risk for metabolic dysfunction [15]. Serial assessment before and after dietary intervention provides objective evidence of metabolic improvement.
4.2 Free Fatty Acids and Ketone Bodies
Free fatty acid (FFA) and beta-hydroxybutyrate (β-HBA) concentrations reflect the metabolic shift toward fat oxidation that represents the therapeutic goal of high-fat, low-starch dietary management in PSSM. In dietary intervention studies, FFA concentrations increased with decreasing dietary starch and increasing fat content, while post-exercise β-HBA concentrations also rose with fat-supplemented diets [7]. These metabolic changes indicate successful substrate shifting from carbohydrate toward lipid metabolism.
The importance of adequate FFA availability extends beyond simple energy provision. PSSM horses demonstrate low muscle oxidative capacity and reduced lipid stores, suggesting impaired ability to utilize fat as a metabolic fuel [7]. Dietary fat supplementation may therefore serve both to reduce glycemic stimulus and to enhance the capacity for fatty acid oxidation through adaptive enzyme upregulation. Serial measurement of circulating FFA and ketone bodies can provide evidence that metabolic adaptation to high-fat feeding is occurring.
The forage-only diet approach has demonstrated metabolic effects that align with these principles. Horses fed high-energy forage-only diets showed increased plasma acetate concentrations before and after exercise, along with higher non-esterified fatty acid (NEFA) levels the day after exercise, compared to traditional high-starch, low-energy forage diets [16]. These findings support the concept that fiber-based diets may provide metabolic benefits similar to fat supplementation through hindgut fermentation products.
5. Nutritional Status and Antioxidant Markers
5.1 Vitamin E and Selenium Status
Assessment of vitamin E and selenium status is relevant to PSSM monitoring given the role of oxidative stress in muscle pathology and the documented deficiencies that can contribute to nutritional myopathies. White muscle disease (WMD) in foals, caused by selenium deficiency, presents with muscle weakness, inability to rise, and elevated serum muscle enzymes with low glutathione peroxidase (GSH-Px) levels [17]. While WMD represents a distinct pathological entity from PSSM, both conditions share features of muscle dysfunction that may be exacerbated by inadequate antioxidant status.
Selenium status assessment typically employs measurement of glutathione peroxidase activity as a functional marker of selenium availability, rather than total selenium concentration alone. In foals with WMD, low GSH-Px levels normalized after appropriate vitamin E and selenium supplementation, providing direct evidence of therapeutic response [17]. For PSSM horses, ensuring adequate antioxidant status may complement dietary starch restriction by reducing oxidative damage to already compromised muscle fibers.
The importance of antioxidant supplementation has been demonstrated in human myopathy populations with potential relevance to equine conditions. In patients with facioscapulohumeral muscular dystrophy, personalized antioxidant supplementation with vitamins C and E, zinc, copper, and selenomethionine produced significant improvements in muscle strength and quality of life [18]. While direct extrapolation to equine PSSM requires caution, these findings support evaluation of antioxidant status as part of comprehensive nutritional assessment.
5.2 Body Condition and Muscle Scoring
Body condition scoring (BCS) and muscle atrophy assessment provide practical, non-invasive monitoring tools for evaluating the overall effectiveness of nutritional management in PSSM horses. The Muscle Atrophy Scoring System (MASS) has been specifically developed and validated for horses, with inter-rater agreement in the good-to-excellent range for neck (ICC=0.62), back (ICC=0.62), and hindquarter (ICC=0.76) regions [19]. Weight was inversely associated with muscle atrophy scores at all regions, while age showed positive correlation with atrophy scores, reflecting the expected relationships between these parameters.
The integration of morphometric measurements with BCS and muscle atrophy scoring provides a comprehensive framework for body composition assessment. Research has demonstrated that body weight of horses can be predicted with high reproducibility (concordance correlation coefficient=0.97), accuracy (0.99), and precision (0.97) using height at withers, chest circumference, cane circumference, body length, body circumference, along with BCS, cresty neck score (CNS), and MASS [20]. These objective measurements can track response to dietary intervention over time.
For PSSM horses specifically, monitoring body condition is important to ensure that dietary modifications designed to reduce starch do not inadvertently result in inadequate energy intake. The goal is to maintain appropriate body condition while shifting metabolic substrate utilization, which may require careful adjustment of fat supplementation to meet energy requirements without excessive weight gain.
6. Functional Assessment Methods
6.1 Exercise Tolerance Testing
Exercise tolerance represents a functionally meaningful outcome for assessing dietary response in PSSM, as reduction of exercise-induced rhabdomyolysis episodes is the primary clinical goal of nutritional management. Standardized exercise testing protocols enable quantification of exercise capacity and detection of muscle damage thresholds. The controlled dietary intervention studies employed exercise protocols with sampling at defined post-exercise time points (4 hours) to assess muscle enzyme release under standardized conditions [7].
The relationship between dietary intervention and exercise response has been documented in fitness-controlled studies. Thoroughbreds with recurrent exertional rhabdomyolysis showed that the combination of fitness and high NSC diet amplified metabolic responses that may trigger rhabdomyolysis, while low NSC, high fat diets prevented this effect regardless of fitness level [14]. This suggests that dietary management may be particularly important for horses in active training programs where fitness-induced metabolic changes could otherwise predispose to muscle damage.
Practical assessment of exercise tolerance in clinical settings may rely on owner-reported observations of performance and post-exercise signs rather than formal treadmill testing. Documentation of the intensity, duration, and type of exercise that can be performed without triggering clinical signs provides clinically relevant outcome data for monitoring dietary response over time.
6.2 Gait Analysis and Locomotion Assessment
Objective gait analysis provides quantitative assessment of locomotor function that may detect subclinical effects of muscle pathology not apparent on subjective examination. Inertial measurement unit (IMU) systems have been validated for equine gait analysis, offering good repeatability across trials and functionality in varying environmental conditions [21]. These systems provide objective assessment of movement asymmetries and can detect changes after interventions such as diagnostic analgesia, suggesting utility for monitoring therapeutic responses.
The application of surface electromyography (sEMG) has been specifically investigated in horses with PSSM. Studies of Haflinger and Noriker horses with known GYS1 mutation status demonstrated that genetically affected horses (both heterozygous GA and homozygous AA) showed significantly reduced density of muscle activity compared to non-affected horses during walk and trot [22]. Importantly, this reduction in functional muscle fiber activity detected by sEMG was present even in the absence of overt clinical signs of myopathy, suggesting potential utility for subclinical monitoring.
The integration of kinematic and kinetic assessment provides complementary information about gait function. While kinematic evaluations focus on movement asymmetries detectable by position sensors, kinetic examinations based on pressure measurement under hooves can quantify force distribution changes [23]. Both approaches may have value for detecting functional improvements following dietary intervention, though formal validation in PSSM populations is needed.
6.3 Owner-Reported Outcomes
Owner-reported outcomes capture clinically meaningful aspects of disease impact and treatment response that may not be fully reflected in objective biomarkers. The frequency and severity of rhabdomyolysis episodes, willingness to work, behavioral changes, and overall quality of life represent important dimensions of treatment success from the horse owner's perspective. Standardized questionnaire approaches similar to those used in epidemiological studies of exertional rhabdomyolysis could be adapted for monitoring dietary response [12].
Recurrence frequency represents a particularly important owner-reported outcome. Documentation of the number, timing, and precipitating circumstances of rhabdomyolysis episodes provides direct evidence of treatment effectiveness. Successful dietary management would be expected to reduce both the frequency and severity of clinical episodes, with the ultimate goal of complete prevention during appropriate exercise.
The epidemiological approach to characterizing risk factors and outcomes in exertional rhabdomyolysis provides a framework for structured owner reporting. Factors such as sex (female horses at significantly greater risk), temperament (nervous horses at greater risk), and management conditions have been systematically evaluated and could be incorporated into monitoring protocols [12].
7. Ranked Table of Monitoring Tools
The following table synthesizes the evidence reviewed above to provide a ranked assessment of biomarkers and outcome measures for monitoring dietary response in PSSM, considering evidence support, sensitivity/specificity, reproducibility, practicality, and clinical utility.
| Rank | Monitoring Tool | Evidence Support | Sensitivity | Specificity | Reproducibility | Practicality | Clinical Utility | Recommended Use |
|---|---|---|---|---|---|---|---|---|
| 1 | Serum CK Activity | High | High | Moderate | High | High | High | Primary monitoring marker; serial pre/post-exercise sampling |
| 2 | GYS1 Genetic Testing | High | High (PSSM1) | High | High | Moderate | High | Baseline diagnosis; genotype-specific dietary targets |
| 3 | Muscle Biopsy (PAS) | High | High | High | Moderate | Low | Moderate | Baseline diagnosis; confirmatory when diagnosis uncertain |
| 4 | Serum AST Activity | High | Moderate | Moderate | High | High | High | Complementary to CK; longer half-life for historical damage |
| 5 | Postprandial Insulin Response | Moderate | Moderate | Moderate | Moderate | Moderate | Moderate | Assessment of dietary compliance and metabolic response |
| 6 | Exercise Tolerance Testing | Moderate | Moderate | High | Moderate | Moderate | High | Functional outcome assessment; standardized protocols preferred |
| 7 | Muscle Glycogen Concentration | Moderate | High | High | Moderate | Low | Low | Research applications; limited responsiveness to diet |
| 8 | Body Condition Score | Moderate | Moderate | Low | Moderate | High | High | Overall nutritional status; energy balance monitoring |
| 9 | Recurrence Frequency | Moderate | High | High | High | High | High | Primary clinical endpoint; owner-reported |
| 10 | Owner-Reported Outcomes | Moderate | Variable | Variable | Moderate | High | High | Quality of life assessment; treatment satisfaction |
| 11 | Muscle Atrophy Scoring (MASS) | Low-Moderate | Moderate | Moderate | Moderate | High | Moderate | Long-term monitoring of muscle mass preservation |
| 12 | Vitamin E/Selenium Status | Low-Moderate | Variable | Low | Moderate | Moderate | Moderate | Adjunctive nutritional assessment; deficiency correction |
| 13 | Serum LDH Activity | Low | Low | Low | High | High | Low | Non-specific; limited utility for PSSM monitoring |
| 14 | Gait Analysis | Low | Variable | Variable | Moderate | Low | Low | Research applications; subclinical detection potential |

Figure 3: Comparison of evidence support and clinical utility scores for biomarkers used in monitoring dietary response in PSSM. Serum CK activity and GYS1 genetic testing demonstrate the highest combined scores for both evidence support and clinical utility.
8. Practical Recommendations for Monitoring Protocols
8.1 Baseline Assessment
Prior to initiating dietary intervention, a comprehensive baseline assessment should include GYS1 genetic testing (particularly in predisposed breeds), resting serum CK and AST activities, body condition scoring, and muscle atrophy evaluation. For cases where genetic testing is negative but clinical suspicion remains high, muscle biopsy with PAS staining should be considered to evaluate for PSSM2 or other myopathies [11]. Documentation of current diet composition, exercise program, and frequency/severity of clinical episodes provides essential reference points for monitoring.
8.2 Short-Term Monitoring (2-6 Weeks)
During the initial dietary transition period, serum CK and AST should be monitored every 2 weeks, ideally with standardized sampling conditions including timing relative to meals and exercise. A post-exercise sampling protocol (4 hours post-exercise) with defined exercise parameters provides the most sensitive detection of treatment response [7]. Reduction in post-exercise CK elevation indicates successful dietary modification. Owner should document any clinical episodes with details of precipitating circumstances.
8.3 Long-Term Monitoring (Months to Years)
Once dietary management is established, monitoring frequency can decrease to quarterly or semi-annual assessments. Regular body condition scoring and muscle atrophy evaluation document maintenance of muscle mass and appropriate energy balance. Exercise tolerance should progressively improve, allowing gradual advancement of training intensity. Annual reassessment of metabolic markers (postprandial insulin, FFA) may be appropriate for horses on high-fat diets to ensure continued metabolic adaptation. Vitamin E and selenium status should be evaluated annually, particularly in areas with known soil deficiency [17].
8.4 Threshold Values for Clinical Decision-Making
Based on available evidence, the following threshold values provide guidance for interpreting monitoring results:
- Resting CK: Normal <350 U/L; mild elevation 350-1000 U/L; significant elevation >1000 U/L [8]
- Post-exercise CK increase: Successful dietary management suggested by <2-fold increase from resting values
- Dietary targets: <5% DE from starch, >12% DE from fat for optimal response [7]
- Histopathology scores: Reduction in amylase-resistant polysaccharide score indicates tissue-level improvement
8.5 Disease Subtype-Specific Considerations
PSSM1 homozygous horses demonstrate more severe subclinical disease and may require more aggressive dietary modification and closer monitoring than heterozygotes [8]. PSSM2 horses show intermediate glycogen accumulation and histopathology scores between PSSM1 and controls, potentially allowing slightly more flexibility in dietary management [4]. For cases without identified genetic basis (PSSM2, idiopathic exertional rhabdomyolysis), clinical response to dietary intervention becomes the primary measure of success.
9. Conclusions
This systematic review identifies serum creatine kinase activity as the most well-validated and practical biomarker for monitoring dietary response in PSSM, supported by strong evidence demonstrating dose-response relationships between dietary starch content and CK activity. GYS1 genetic testing provides essential diagnostic confirmation for PSSM1 and enables genotype-specific monitoring approaches. Muscle biopsy with PAS staining remains important for diagnostic confirmation, particularly for PSSM2 and genetically-negative cases, though the invasive nature limits utility for serial monitoring.
Metabolic markers including postprandial glucose and insulin responses provide mechanistic assessment of dietary compliance and metabolic adaptation, while functional outcomes including exercise tolerance and owner-reported recurrence frequency capture clinically meaningful treatment success. The integration of multiple assessment approaches—combining serum enzymes, metabolic markers, body condition assessment, and functional outcomes—provides the most comprehensive evaluation of dietary response.
Future research priorities should include validation of novel biomarkers with improved sensitivity and specificity, development of standardized exercise testing protocols for PSSM monitoring, investigation of molecular markers that may predict individual response to dietary intervention, and long-term outcome studies correlating biomarker changes with clinical outcomes across disease subtypes. The continued refinement of monitoring strategies will support optimization of nutritional management for this significant cause of equine morbidity.
References
References
Numbered in order of first citation, IEEE style. Each entry links to the original source via DOI or publisher URL.
- M. B. S. Teixeira, G. R. Bonatto, T. G. Cristo, D. Hemckmeier, N. L. Pisetta, and R. Casagrande, “Polysaccharide storage myopathy in two Percheron horses,” Revista de Ciências Agroveterinárias, Mar. 2023. doi: 10.5965/223811712212023078. View source
- R. Naylor, V. Luis-Fuentes, L. Livesey, C. Mobley, N. Henke, K. Brock, M. Fernández-Fuente, and R. Piercy, “Evaluation of cardiac phenotype in horses with type 1 polysaccharide storage myopathy.,” Journal of Veterinary Internal Medicine, Nov. 2012. doi: 10.1111/j.1939-1676.2012.00988.x. View source
- N. Blohina, S. I. Sorokin, L. Khrabrova, A. Zelenchenkova, and B. Bazaron, “Biodiversity and functional variability of the allele pool of horse breed populations,” IOP Conference Series: Earth and Environment, Sep. 2021. doi: 10.1088/1755-1315/848/1/012229. View source
- S. Valberg, Z. J. Williams, C. Finno, A. Schultz, D. Velez-Irizarry, M. Henry, K. Gardner, and J. Petersen, “Type 2 polysaccharide storage myopathy in Quarter Horses is a novel glycogen storage disease causing exertional rhabdomyolysis.,” Equine Veterinary Journal, Aug. 2022. doi: 10.1111/evj.13876. View source
- S. Valberg, M. Henry, K. L. Herrick, D. Velez-Irizarry, C. Finno, and J. Petersen, “Absence of myofibrillar myopathy in Quarter Horses with a histopathological diagnosis of type 2 polysaccharide storage myopathy and lack of association with commercial genetic tests,” Equine Veterinary Journal, Mar. 2022. doi: 10.1111/evj.13574. View source
- E. Barrey, E. Mucher, N. Jeansoule, T. Larcher, L. Guigand, B. Herszberg, S. Chaffaux, G. Guérin, X. Mata, P. Benech, M. Canale, O. Alibert, P. Maltere, and X. Gidrol, “Gene expression profiling in equine polysaccharide storage myopathy revealed inflammation, glycogenesis inhibition, hypoxia and mitochondrial dysfunctions,” BMC Veterinary Research, Aug. 2009. doi: 10.1186/1746-6148-5-29. View source
- W. P. Ribeiro, S. Valberg, J. D. Pagan, and B. Gustavsson, “The effect of varying dietary starch and fat content on serum creatine kinase activity and substrate availability in equine polysaccharide storage myopathy.,” Journal of Veterinary Internal Medicine, Nov. 2004. doi: 10.1111/j.1939-1676.2004.tb02637.x. View source
- R. Naylor, L. Livesey, J. Schumacher, N. Henke, C. Massey, K. Brock, M. Fernández-Fuente, and R. Piercy, “Allele Copy Number and Underlying Pathology Are Associated with Subclinical Severity in Equine Type 1 Polysaccharide Storage Myopathy (PSSM1),” PLoS ONE, Jul. 2012. doi: 10.1371/journal.pone.0042317. View source
- M. Pasolini, R. Pezzella, P. Santoro, N. Cocchia, M. Greco, C. D. Prete, G. D. Valle, and L. Auletta, “Correlation Between Serum Activity of Muscle Enzymes and Stage of the Estrous Cycle in Italian Standardbred Horses Susceptible to Exertional Rhabdomyolysis.,” Journal of Equine Veterinary Science, Sep. 2020. doi: 10.1016/j.jevs.2020.103175. View source
- M. Lecina, C. Castellar-Otín, A. García-Giménez, and F. Pradas, “Exertional Rhabdomyolysis and Ultra-Trail Races: A Systematic Review Highlighting the Significant Impact of Eccentric Load,” Muscles, Aug. 2024. doi: 10.3390/muscles3030022. View source
- R. Stanley, M. E. McCue, S. Valberg, J. R. Mickelson, I. G. Mayhew, C. M. McGowan, C. Hahn, J. C. Patterson-Kane, and R. J. Piercy, “A glycogen synthase 1 mutation associated with equine polysaccharide storage myopathy and exertional rhabdomyolysis occurs in a variety of UK breeds.,” Equine Veterinary Journal, Jul. 2009. doi: 10.2746/042516409X407611. View source
- C. Isgren, M. Upjohn, M. Fernández-Fuente, C. Massey, G. Pollott, K. Verheyen, and R. Piercy, “Epidemiology of Exertional Rhabdomyolysis Susceptibility in Standardbred Horses Reveals Associated Risk Factors and Underlying Enhanced Performance,” PLoS ONE, Jul. 2010. doi: 10.1371/journal.pone.0011594. View source
- C. A. Kósa, O. Szenci, L. Lénárt, I. Biksi, R. Szép, Á. Keresztesi, M. Mircean, M. Tăulescu, and O. Kutasi, “Genetic background of the Transylvanian endemic equine recurrent rhabdomyolysis.,” Acta Veterinaria Hungarica, Feb. 2026. doi: 10.1556/004.2026.01199. View source
- C. Finno, E. McKenzie, S. Valberg, and J. D. Pagan, “Effect of fitness on glucose, insulin and cortisol responses to diets varying in starch and fat content in Thoroughbred horses with recurrent exertional rhabdomyolysis.,” Equine veterinary journal. Supplement, Nov. 2010. doi: 10.1111/j.2042-3306.2010.00199.x. View source
- F. Bertin, “Diagnosis and management of equine metabolic syndrome,” Equine Veterinary Education, Mar. 2026. doi: 10.1111/eve.70094. View source
- A. Jansson and J. Lindberg, “A forage-only diet alters the metabolic response of horses in training.,” Animal, Dec. 2012. doi: 10.1017/S1751731112000948. View source
- C. Delesalle, M. de Bruijn, S. Wilmink, H. Vandendriessche, G. Mol, B. Boshuizen, L. Plancke, and G. Grinwis, “White muscle disease in foals: focus on selenium soil content. A case series,” BMC Veterinary Research, May 2017. doi: 10.1186/s12917-017-1040-5. View source
- D. Laoudj-Chenivesse, S. Arbogast, E. Raynaud de Mauverger, C. Fédou, E. Debroize, G. Hugon, J. Pincemail, M. Picot, J. Cristol, J. Mercier, and F. Portet, “Personalized Antioxidant Supplementation Improves Muscle Strength, Physical Activity, and Quality of Life in Patients with FSHD1: A Real-World Longitudinal Study.,” Free Radical Biology & Medicine, Feb. 2026. doi: 10.1016/j.freeradbiomed.2026.02.071. View source
- A. Herbst, M. Johnson, H. Gammons, S. Reedy, K. Urschel, P. Harris, and A. Adams, “Development and Evaluation of a Muscle Atrophy Scoring System (MASS) for Horses.,” Journal of Equine Veterinary Science, Sep. 2021. doi: 10.1016/j.jevs.2021.103771. View source
- N. Urbanek and Q. Zebeli, “Morphometric Measurements and Muscle Atrophy Scoring as a Tool to Predict Body Weight and Condition of Horses,” Veterinary Sciences, Aug. 2023. doi: 10.3390/vetsci10080515. View source
- K. V. Zullo, J. G. Davis, and K. Bubeck, “Objective gait analysis methods for assessing movement asymmetry in the equine patient,” Journal of the American Veterinary Medical Association, Apr. 2026. doi: 10.2460/javma.25.12.0784. View source
- R. Zsoldos, N. Khayatzadeh, J. Soelkner, U. Schroeder, C. Hahn, and T. Licka, “Comparison of gluteus medius muscle activity in Haflinger and Noriker horses with polysaccharide storage myopathy,” Journal of animal physiology and animal nutrition, Feb. 2021. doi: 10.1111/jpn.13504. View source
- C. Crecan and C. Peștean, “Inertial Sensor Technologies—Their Role in Equine Gait Analysis, a Review,” Italian National Conference on Sensors, Jul. 2023. doi: 10.3390/s23146301. View source
This review is one of 34 in the Foxwatch research corpus. Methodology: PRISMA 2020-style systematic literature review against PubMed, CAB Abstracts, Scopus, Web of Science, and equine veterinary conference proceedings.