Systematic review of body condition, obesity, insulin dysregulation, and PSSM feeding
A "low-sugar" diet does not exempt a horse from being too fat. PSSM horses with metabolic syndrome have a worse time of it because the muscle is already insulin-sensitive and obesity drives the substrate load the diet is trying to restrict. This review integrates body-condition management, EMS screening, and PSSM dietary strategy into a single plan. Read with review 17 (the opposite problem, the underweight horse) and review 09 (meal timing).
Research thesis
This systematic review argues that body condition, insulin dysregulation, and PSSM are linked metabolic problems, and that a "low-NSC" diet without simultaneous attention to body condition can fail or even worsen the horse, not only because the GYS1 mutation drives accelerated glycogen storage in already insulin-sensitive muscle, but also because obesity and pasture-driven insulin spikes raise the substrate load that the diet is meant to restrict; by reviewing the equine metabolic syndrome, insulin dynamics, and PSSM management literature with attention to body-condition targets and metabolic comorbidity, this review integrates body-condition management, EMS screening, and the PSSM dietary protocol into a single coherent treatment plan.
Evidence visualizations & full pages
Evidence figures (2)
Figures referenced by the review's Figure N: captions. Click any chart for a full-size view. They are also rendered inline below their respective captions in the full review text further down.
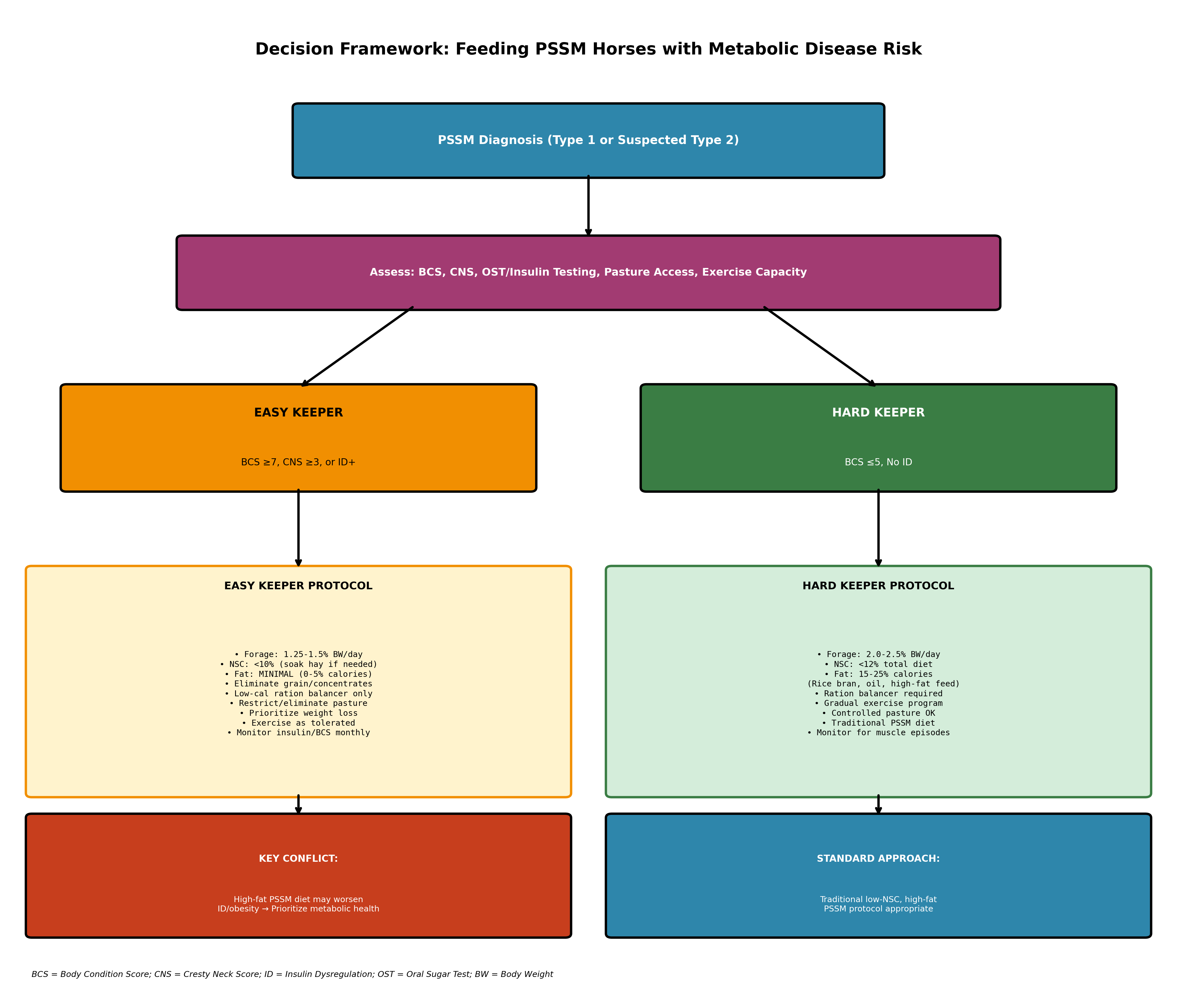 Figure 1. Decision framework for feeding PSSM horses with metabolic disease risk. The algorithm stratifies horses based on body condition score (BCS), cresty neck score (CNS), and insulin dysregulation (ID) st
Figure 1. Decision framework for feeding PSSM horses with metabolic disease risk. The algorithm stratifies horses based on body condition score (BCS), cresty neck score (CNS), and insulin dysregulation (ID) st
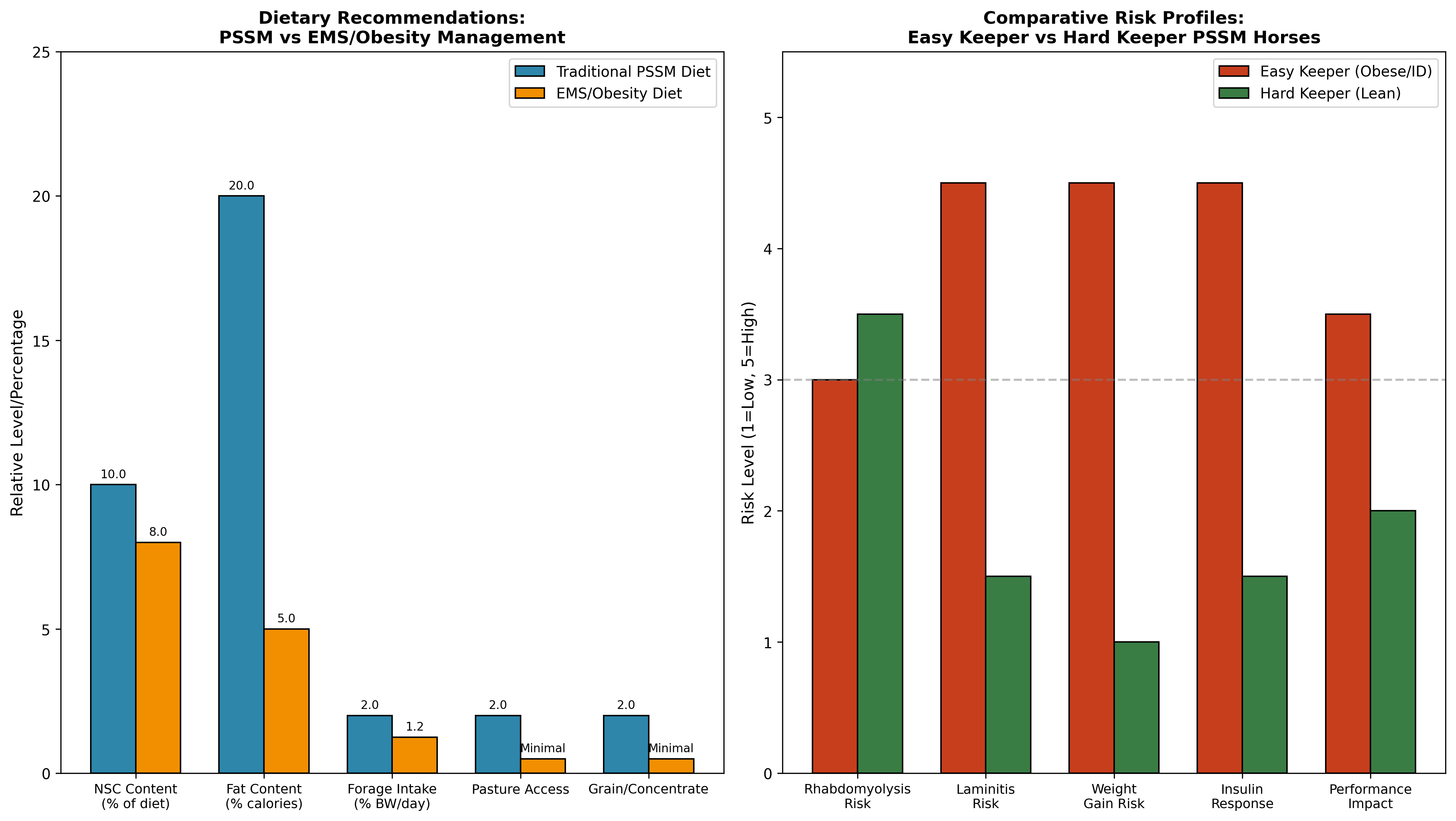 Figure 1. Decision framework for feeding PSSM horses with metabolic disease risk. The algorithm stratifies horses based on body condition score (BCS), cresty neck score (CNS), and insulin dysregulation (ID) st
Figure 1. Decision framework for feeding PSSM horses with metabolic disease risk. The algorithm stratifies horses based on body condition score (BCS), cresty neck score (CNS), and insulin dysregulation (ID) st
Original review pages (PDF render)
Download
Full review text
Body condition, obesity, insulin dysregulation, and PSSM feeding
Systematic Review: Feeding PSSM Horses with Obesity, Insulin Dysregulation, and Metabolic Disease Risk
1. Introduction and Background
1.1 Overview of PSSM and Metabolic Syndrome in Horses
Polysaccharide storage myopathy (PSSM) represents a significant glycogen storage disorder affecting multiple horse breeds, with Type 1 PSSM (PSSM1) being an autosomal dominant condition affecting more than 20 breeds and presenting with exertional rhabdomyolysis as a primary manifestation [1]. The condition is diagnosed through genetic testing or muscle biopsies revealing abnormal amylase-resistant polysaccharide accumulation in muscle fibers. Type 2 PSSM (PSSM2) has recently been subdivided, with PSSM2-ER representing a glycogen storage disorder identified primarily in Quarter Horses that causes exertional rhabdomyolysis but has an unknown genetic basis. Both PSSM1 and PSSM2-ER traditionally respond well to a low nonstructural carbohydrate (NSC), high-fat diet combined with regular exercise.
Equine metabolic syndrome (EMS) presents a complex challenge when occurring concurrently with PSSM, as it represents a prevalent endocrine disorder characterized by insulin dysregulation (ID), which encompasses basal or postprandial hyperinsulinemia and tissue insulin resistance [2]. The central concern with EMS is the dramatically increased risk of hyperinsulinemia-associated laminitis, the most common and clinically significant form of laminitis in horses. While obesity is frequently associated with EMS, the condition can occur in lean animals, and not all obese horses are affected, necessitating objective diagnostic testing rather than reliance on body condition alone. Environmental and management factors, particularly high-NSC diets, unrestricted pasture access, and physical inactivity, are the primary drivers of EMS development.
1.2 The Clinical Challenge of Comorbid Conditions
The intersection of PSSM and metabolic disease creates a unique therapeutic dilemma for veterinarians and horse owners. EMS is a health problem that particularly affects horses considered "easy keepers," with the disease consisting of large regional fat deposits, obesity, insulin resistance, and if not controlled, recurrent episodes of laminitis [3]. Despite education by veterinarians and nutritionists to maintain horses at moderate body weight, horse owners continue to feed high-calorie concentrates and hay, often resulting in overweight middle-aged horses that struggle with mild exercise and are predisposed to metabolic complications.
The traditional high-fat PSSM diet recommendation directly conflicts with the dietary management of obesity and insulin dysregulation, which emphasizes calorie restriction and limited fat intake. This review systematically examines the evidence base for balancing these competing dietary requirements, with specific attention to body condition scoring, cresty neck assessment, insulin and glucose testing, diet composition, forage intake, and clinical outcomes in horses affected by both PSSM and metabolic disease.
2. Assessment and Diagnostic Considerations
2.1 Body Condition Scoring and Adiposity Assessment
Body condition assessment forms the foundation of metabolic evaluation in horses with concurrent myopathy and metabolic concerns. Obesity in horses increases the risk of developing insulin dysregulation, reduced athleticism, colic, abnormal reproductive performance, laminitis, endotoxemia, diabetes mellitus, hyperlipemia, impaired thermoregulation, and pituitary pars intermedia dysfunction [4]. Weight gain occurs fundamentally when energy consumption exceeds physical energy needs, making accurate body condition assessment essential for developing appropriate feeding strategies. In practice, obesity is assessed through body condition scoring using the Henneke system, cresty neck scoring, ultrasonographic assessment, morphometric measurements, or biochemical indicators in blood.
Research examining obese horses demonstrated significantly elevated leptin levels and significantly lower adiponectin concentrations compared to controls with normal body condition scores of 4-6 on the Henneke scale [5]. This resulted in more than a threefold increase in the leptin-to-adiponectin ratio (LAR), indicating marked disruption of hormonal regulation of adipose tissue. Additionally, elevated glycemia, hyperinsulinemia, and decreased reciprocal of the square root of insulin (RISQI) values confirmed insulin resistance in overweight horses. These findings suggest that LAR can serve as a sensitive early biomarker of metabolic distress and insulin resistance, with use recommended for early identification of at-risk animals.
2.2 Cresty Neck Score as an Independent Predictor
The cresty neck score has emerged as a particularly valuable assessment tool that may be more predictive of insulin dysregulation than general body condition score alone. Research demonstrated that post-prandial serum insulin was positively associated with CNS, and ponies with a CNS ≥3 had 5 times greater odds of being insulin-dysregulated [6]. Ponies with high CNS but without generalized obesity (BCS ≤7 and CNS ≥3) showed a greater insulin response to oral glucose testing than those in the normal/fleshy group, whereas obese ponies did not differ significantly from other groups. Basal high-molecular-weight adiponectin was negatively correlated with post-prandial insulin concentrations and was decreased in the group with high CNS compared to the obese group.
Studies in Connemara ponies found that 29.5% had a BCS ≥7, 29.0% had a CNS ≥2.5, and 67.5% had regionalized adiposity, with 68.5% of ponies having at least one of these abnormalities [7]. Owner-reported history or clinical evidence of chronic laminitis was found in 46.0% of ponies, and the odds of hyperinsulinemia increased by a factor of 6.53 when BCS was ≥7. These findings emphasize the importance of comprehensive adiposity assessment in PSSM horses, particularly those belonging to breeds predisposed to metabolic disease.
2.3 Insulin and Glucose Testing Protocols
Dynamic testing for insulin dysregulation is essential for risk stratification in PSSM horses with suspected metabolic comorbidities. The oral sugar test (OST) represents the most commonly used diagnostic tool for identifying ID and EMS [8]. In longitudinal research, horses with ID had lower serum adiponectin concentrations, longer neck circumference, and larger height than horses without ID. Importantly, the risk of ID was substantially higher in horses with no exercise compared to horses with exercise (OR 7.6, 95% CI 1.2-49.3, P = 0.03), highlighting the critical role of physical activity in metabolic regulation.
Epidemiological investigation in ponies revealed that among 167 ponies tested, the prevalence of ID was 61% (95% CI 53-68%) [9]. Factors associated with insulin concentrations 60 minutes post-OST included age, CNS ≥3/5, and owner-perceived obesity. Critically, factors associated with laminitis included dynamic hyperinsulinemia (OR 4.60), insulin resistance (OR 3.66), and pituitary pars intermedia dysfunction (OR 11.75). This emphasizes the need for concurrent PPID screening in older horses presenting with PSSM and metabolic concerns.
2.4 Relationship Between PPID, EMS, and Metabolic Risk
Pituitary pars intermedia dysfunction frequently complicates the management of PSSM horses, particularly in older populations. PPID is a prevalent, age-related chronic disorder with a prevalence of 21-27% in equids aged ≥15 years [10]. The clinical presentation includes hypertrichosis or delayed/incomplete hair coat shedding, which provides a high index of clinical suspicion for PPID. The combination of clinical signs and age informs the index of clinical suspicion prior to diagnostic testing, and importantly, equids with PPID and hyperinsulinemia appear to be at higher risk of laminitis.
Research examining PPID cases across Australian latitudes found that laminitis was diagnosed in 89.9% of cases and ID was present in 76.5% of cases in which these were investigated [11]. Being a pony, having a higher body condition score, and pergolide administration were associated with survival, while adequate body condition and administration of pergolide were identified as fundamental in PPID management. The clinical presentation of PPID changed with latitude and climate, with anhidrosis and polyuria/polydipsia more commonly recognized at lower latitudes.
3. Dietary Management Principles
3.1 NSC Restriction: Evidence and Recommendations
Nonstructural carbohydrate restriction forms a cornerstone of management for both PSSM and metabolic disease, though the degree of restriction may differ. For PSSM management, both PSSM1 and PSSM2-ER respond well to a low NSC diet, typically targeting less than 10-12% of the total diet [1]. However, in metabolic disease, more stringent restriction may be necessary. Management of EMS is centered on strict dietary NSC restriction, controlled forage intake, and elimination or careful management of pasture access [2].
The importance of NSC restriction is supported by evidence that excessive dietary nonstructural carbohydrates contribute to sustained hyperinsulinemia which induces laminitis. Dietary adjustments are the key to weight loss in the majority of EMS horses, and to encourage weight loss, horses should be given approximately 1.25% of body weight per day [12]. Exercise not only aids weight loss but also improves insulin sensitivity, though exercise must be tailored to the specific horse, taking into account its breed, fitness level, and owner's resources. When an obese horse is unable to exercise owing to laminitis or when obesity persists after extensive exercise and dietary management, pharmacological interventions such as levothyroxine sodium and metformin hydrochloride can be considered.
3.2 Fat Supplementation: The Central Conflict
The fundamental conflict between traditional PSSM dietary recommendations and obesity/insulin management centers on fat supplementation. Traditional PSSM diets recommend that 15-25% of dietary calories come from fat sources to provide alternative energy substrates and reduce reliance on glycogen [1]. This high-fat approach has shown benefits for reducing rhabdomyolysis episodes and improving exercise tolerance in affected horses.
However, research examining Warmblood horses with type 2 PSSM found that while 80% of owners reported an overall improvement with the recommended low-starch/fat-supplemented diet and exercise regime, 53% of horses were still not advancing as expected, with reluctance to go forward and collect persisting in approximately one-third of horses [13]. Median muscle glycogen concentrations did not differ between PSSM2 horses and controls without evident myopathy, though horses with the highest glycogen concentrations were significantly more likely to show a decline in performance. This suggests that diet and exercise recommendations ideal for PSSM1 improve but do not eliminate the decline in performance characteristic of PSSM2.
For easy keeper horses with concurrent obesity and insulin dysregulation, high-fat diets may exacerbate metabolic dysfunction by contributing excess calories and potentially worsening insulin resistance. In such cases, the priority must shift toward metabolic health, with fat supplementation minimized or eliminated until body condition and insulin sensitivity improve.
3.3 Caloric Restriction and Weight Management
Effective caloric restriction requires careful balancing of energy intake against nutritional requirements and PSSM management needs. The current management of EMS is based on caloric restriction and increased physical activity [14]. Research has demonstrated that horses fed hypercaloric diets develop hypercholesterolemia concomitantly with insulin dysregulation, with cholesterol, LDL, HDL, and fructosamine blood concentrations increasing significantly and correlating positively with blood insulin response to glucose tolerance testing [15].
Studies examining dietary supplementation with a synergistic polyphenol and amino acid blend including leucine showed that horses after supplementation weighed significantly less, had significantly higher baseline high-molecular-weight adiponectin concentrations, and had significantly lower insulin concentrations at 60- and 75-minute time points [16]. An increased HMW adiponectin level supports increasing insulin sensitivity after supplementation, suggesting that targeted nutritional interventions may improve the clinical manifestations of EMS/insulin dysregulation while potentially reducing laminitis risk.
3.4 Forage Management and Pasture Considerations
Forage intake represents the foundation of equine nutrition and requires careful management in horses with PSSM and metabolic disease. For obese or insulin-dysregulated horses, forage should be restricted to 1.25-1.5% of body weight daily to promote weight loss while meeting minimum fiber requirements. Hay soaking can reduce NSC content, though the degree of reduction varies based on soaking duration, water temperature, and initial hay composition.
Pasture access presents particular challenges for easy keeper horses with metabolic concerns. The prevalence of obesity in leisure horses was found to be 21.4% compared to only 3.3% in polo horses, with factors associated with increased odds of obesity including horses not being exercised (66.7%), horses tethered to a stake in the ground (23.2%), and horses fed ad libitum on concentrates (21.4%) [17]. These findings emphasize that both pasture management and exercise are critical components of metabolic disease prevention.
4. Exercise and Physical Activity
4.1 Exercise and Insulin Sensitivity
Regular exercise represents perhaps the most important non-dietary intervention for managing both PSSM and metabolic disease. Research has consistently demonstrated that lack of exercise is a significant risk factor for insulin dysregulation [8]. The benefits extend beyond weight management to include direct improvements in insulin sensitivity, glucose tolerance, and adipokine profiles.
In horses with PSSM, gradual introduction of regular exercise is essential for improving muscle function and reducing rhabdomyolysis risk. However, exercise must be carefully balanced against the risk of triggering episodes in horses that are not adequately conditioned or whose diets have not been appropriately modified. For horses with concurrent laminitis, exercise may be contraindicated until hoof pathology has been addressed.
4.2 Balancing Exercise with Laminitis Risk
The high prevalence of laminitis in metabolically compromised horses presents a significant barrier to implementing exercise programs. Approximately 90% of laminitis cases are associated with insulin dysregulation, making exercise management particularly challenging in these populations [18]. In horses with active laminitis, pharmacological interventions may be necessary to improve metabolic parameters before exercise can be safely introduced.
Sodium-glucose cotransporter 2 inhibitors (SGLT2i) represent a novel class of agents that have been explored for managing equine ID. These drugs increase urinary glucose excretion by suppressing glucose reabsorption from the glomerular filtrate, resulting in urinary calorie loss with consequent weight loss and improvements in ID. While there are currently no licensed veterinary drugs available for treating ID and preventing insulin-associated laminitis in horses, the use of SGLT2i for control of equine hyperinsulinemia has been advocated as an adjunctive therapy.
5. Clinical Outcomes and Prognosis
5.1 Response to Dietary and Exercise Interventions
Clinical outcomes in PSSM horses with metabolic disease depend significantly on the type of PSSM, degree of metabolic dysfunction, and compliance with management recommendations. Research on Warmblood horses with PSSM2 found that abnormal exercise responses reported by owners began at approximately 6 years of age and included a decline in performance, reluctance to collect, and reluctance to go forward in over 50% of horses [13]. With the recommended diet and exercise regime, 80% of owners reported an overall improvement with significant decreases in the proportion of horses showing a decline in performance and rhabdomyolysis.
For horses with concurrent EMS, case reports demonstrate that diet considering adequate energy and nutrient intake combined with an exercise regime can improve behavior, work under saddle, and insulin levels [19]. In one reported case, a mare with EMS showed signs of estrus again and serum hormone levels returned to within normal limits following dietary and exercise intervention, demonstrating the potential for significant clinical improvement with appropriate management.
5.2 Long-term Management Considerations
EMS is controlled mainly by dietary strategies and exercise programs that aim to improve insulin regulation and decrease obesity where present, representing a long-term management strategy requiring diligence and discipline by the horse's carer [20]. The ECEIM consensus statement emphasizes that several choices of test exist for examining different facets of ID and related metabolic disturbances, and that pharmacologic aids might be useful in some cases.
Novel dietary supplements have shown promise in improving metabolic parameters. Supplementation with Arthrospira platensis enriched with chromium, magnesium, and manganese ions has been shown to reduce baseline insulin and glucose levels, decrease body weight, and improve overall body condition scores and cresty neck scores in EMS-affected horses [21]. Additionally, oligosaccharide supplementation significantly reduced insulin in geldings with EMS, suggesting that gut microbiota-targeted interventions may complement conventional management strategies [22].
6. Decision Framework: Easy Keepers vs. Hard Keepers
6.1 Assessment Algorithm
The management of PSSM horses requires stratification based on metabolic phenotype. The following decision framework provides guidance for clinical decision-making:

Figure 1. Decision framework for feeding PSSM horses with metabolic disease risk. The algorithm stratifies horses based on body condition score (BCS), cresty neck score (CNS), and insulin dysregulation (ID) status to guide dietary modifications. Easy keepers require prioritization of metabolic health with reduced fat supplementation, while hard keepers can follow traditional PSSM protocols.
6.2 Dietary Comparison: PSSM vs. EMS Management
The fundamental differences between traditional PSSM dietary recommendations and EMS/obesity management create therapeutic tension that must be resolved on an individual basis:

Figure 2. Comparison of dietary recommendations and risk profiles for PSSM versus EMS/obesity management (left panel) and easy keeper versus hard keeper PSSM horses (right panel). Easy keepers with metabolic disease face elevated risks of laminitis and insulin dysregulation that must be prioritized over traditional PSSM high-fat protocols.
6.3 Summary of Management Recommendations
| Parameter | Easy Keeper (BCS ≥7, ID+) | Hard Keeper (BCS ≤5, No ID) |
|---|---|---|
| Forage Intake | 1.25-1.5% BW/day | 2.0-2.5% BW/day |
| NSC Target | <10% (soak hay if needed) | <12% total diet |
| Fat Calories | 0-5% (minimal) | 15-25% (traditional PSSM) |
| Grain/Concentrate | Eliminate; low-cal ration balancer only | Permitted; ration balancer required |
| Pasture Access | Restrict or eliminate | Controlled access acceptable |
| Exercise | As tolerated; priority after laminitis resolves | Gradual program; essential for PSSM management |
| Monitoring | Monthly: BCS, CNS, insulin | Monitor for muscle episodes |
| Primary Goal | Weight loss and insulin sensitivity | Muscle function and energy supply |
| Pharmacological Support | Consider levothyroxine, metformin, SGLT2i | Generally not indicated |
6.4 Key Clinical Considerations
The management of PSSM horses with metabolic disease requires several critical considerations:
- Prioritization of metabolic health in easy keepers: For horses with BCS ≥7, CNS ≥3, or confirmed insulin dysregulation, the traditional high-fat PSSM diet is contraindicated. Weight loss and insulin sensitivity improvement must take precedence, even if this means accepting some increased risk of mild muscle episodes during the transition period.
- Protein considerations: Research has shown that horses with EMS had a 9-fold greater insulinemic response to consumption of a high protein meal compared with controls, with post-prandial levels of several amino acids being higher in EMS horses [23]. This suggests that dietary protein content should be taken into consideration in management of horses with insulin dysregulation, and excessive protein should be avoided in easy keeper PSSM horses.
- Breed predisposition: Certain breeds are disproportionately affected by both PSSM and EMS. Diagnosis is complicated by the presence of concurrent conditions like pituitary pars intermedia dysfunction, and owner-assigned assessments of body condition were found to be one full score lower than scores assigned by researchers, indicating differing perceptions of healthy equine body weight [24].
- Individualized approach: The relationship between obesity, hyperinsulinemia, and metabolic abnormalities is complex. Research has demonstrated that obesity, whether assessed by rump fat or overweight, is not always associated with hyperinsulinemia or metabolic abnormalities, and conversely, hyperinsulinemia is not always associated with obesity but is related to insulin resistance and dysregulation [25]. This emphasizes the need for individual assessment rather than assumptions based on body condition alone.
- Adipose tissue dysfunction: Horses with obesity and EMS have significant dysfunction of peri-renal and retroperitoneal adipose tissue characterized by marked adipocyte hypertrophy and increased expression of inflammatory cytokines, which may contribute to whole-body insulin dysregulation [26]. This supports the importance of targeting adiposity reduction in easy keeper horses.
- Seasonal considerations: Neither OST results nor adiponectin varied significantly with season in longitudinal studies, though a substantial number of horses had variable ID status throughout the year, suggesting that repeated testing may be beneficial [8].
- Gut microbiome: Obese horses show differences in gut microbiome composition with higher relative abundance of Firmicutes and lower numbers of Bacteroidetes and Actinobacteria compared to lean horses [27]. This suggests potential future therapeutic targets through microbiome-directed interventions.
Conclusions
The management of PSSM horses with concurrent obesity, insulin dysregulation, or metabolic syndrome presents a significant clinical challenge requiring individualized assessment and treatment approaches. The traditional high-fat, low-NSC diet recommended for PSSM directly conflicts with the calorie-restricted, low-fat approach required for metabolic disease management. This review demonstrates that horses must be stratified into easy keeper and hard keeper phenotypes, with dietary recommendations tailored accordingly.
For easy keeper horses (BCS ≥7, CNS ≥3, or confirmed ID), metabolic health must take precedence over traditional PSSM management. Fat supplementation should be minimized or eliminated, forage restricted to 1.25-1.5% of body weight daily, pasture access eliminated or strictly controlled, and NSC content maintained below 10%. Exercise should be introduced as tolerated, with consideration of pharmacological support for severe cases. For hard keeper horses (BCS ≤5, no ID), traditional PSSM protocols with 15-25% fat calories and adequate forage intake remain appropriate.
The evidence supports regular monitoring of body condition score, cresty neck score, and insulin status in all PSSM horses, with particular attention to breeds predisposed to metabolic disease. The presence of PPID significantly increases laminitis risk and should be assessed in older horses. Future research should focus on developing evidence-based guidelines specific to horses with comorbid PSSM and metabolic disease, including evaluation of novel therapeutic agents and dietary supplements that may improve outcomes in this challenging patient population.
References
References
Numbered in order of first citation, IEEE style. Each entry links to the original source via DOI or publisher URL.
- A. Firshman and S. Valberg, “Polysaccharide Storage Myopathy.,” The Veterinary clinics of North America. Equine practice, Jan. 2025. doi: 10.1016/j.cveq.2024.11.004. View source
- F. Bertin, “Diagnosis and management of equine metabolic syndrome,” Equine Veterinary Education, Mar. 2026. doi: 10.1111/eve.70094. View source
- A. Cristian, A. Dutulescu, and M. Codreanu, “Equine metabolic syndrome,” Practica Veterinara ro, 2024. doi: 10.26416/pv.43.1.2024.9642. View source
- O. Akinniyi, A. Sackey, G. E. Ochube, P. Mshelia, and K. O. Jolayemi, “Definition, Assessment, Health Consequences and Management of Equine Obesity: A Review,” Folia Veterinaria, Jun. 2023. doi: 10.2478/fv-2023-0011. View source
- S. Borovkov, V. Boiko, and V. Borovkova, “THE ROLE OF ADIPONECTIN, LEPTIN, AND THEIR RATIO IN THE PATHOGENESIS OF OBESITY AND INSULIN RESISTANCE IN HORSES,” The Scientific and Technical Bulletin of the Institute of Animal Science NAAS of Ukraine, Aug. 2025. doi: 10.32900/2312-8402-2025-134-251-262. View source
- D. Fitzgerald, S. T. Anderson, M. Sillence, and M. D. de Laat, “The cresty neck score is an independent predictor of insulin dysregulation in ponies,” PLoS ONE, Jul. 2019. doi: 10.1371/journal.pone.0220203. View source
- A. S. Al-Ansari, E. Golding, N. Walshe, C. T. Mooney, and V. Duggan, “Obesity and obesity-associated metabolic disease conditions in Connemara ponies in Ireland.,” Equine Veterinary Journal, Nov. 2023. doi: 10.1111/evj.14029. View source
- N. Karikoski, J. R. Box, A. Mykkänen, V. V. Kotiranta, and M. Raekallio, “Variation in insulin response to oral sugar test in a cohort of horses throughout the year and evaluation of risk factors for insulin dysregulation,” Equine Veterinary Journal, May 2021. doi: 10.1111/evj.13529. View source
- B. Clark, E. Norton, N. Bamford, I. Randhawa, K. Kemp, M. E. McCue, F. Bertin, and A. Stewart, “Epidemiological investigation of insulin dysregulation in Shetland and Welsh ponies in Australia.,” Equine Veterinary Journal, Jan. 2024. doi: 10.1111/evj.14044. View source
- N. Menzies‐Gow, H. Banse, A. J. A. Duff, N. Hart, J. L. Ireland, E. Knowles, D. McFarlane, and D. Rendle, “BEVA primary care clinical guidelines: Diagnosis and management of equine pituitary pars intermedia dysfunction.,” Equine Veterinary Journal, Oct. 2023. doi: 10.1111/evj.14009. View source
- R. Horn, N. Bamford, T. Afonso, M. Sutherland, J. Buckerfield, R. Tan, C. Secombe, A. J. Stewart, and F. Bertin, “Factors associated with survival, laminitis and insulin dysregulation in horses diagnosed with equine pituitary pars intermedia dysfunction.,” Equine Veterinary Journal, Dec. 2018. doi: 10.1111/evj.13041. View source
- O. O. Akinniyi, “Focus on the epidemiology, pathophysiology, diagnosis, and management of insulin dysregulation in horses,” Nutrition and Food Processing, Feb. 2024. doi: 10.31579/2637-8914/191. View source
- Z. J. Williams, M. Bertels, and S. Valberg, “Muscle glycogen concentrations and response to diet and exercise regimes in Warmblood horses with type 2 Polysaccharide Storage Myopathy,” PLoS ONE, Sep. 2018. doi: 10.1371/journal.pone.0203467. View source
- K. Marycz, J. Szłapka-Kosarzewska, F. Geburek, and K. Kornicka-Garbowska, “Systemic Administration of Rejuvenated Adipose-Derived Mesenchymal Stem Cells Improves Liver Metabolism in Equine Metabolic Syndrome (EMS)- New Approach in Veterinary Regenerative Medicine,” Stem Cell Reviews and Reports, Oct. 2019. doi: 10.1007/s12015-019-09913-3. View source
- R. M. Ribeiro, D. Ribeiro, L. O. Cota, F. Leme, A. M. Carvalho, and R. R. Faleiros, “Changes in metabolic and physiological biomarkers in Mangalarga Marchador horses with induced obesity.,” The Veterinary Journal, Apr. 2021. doi: 10.1016/j.tvjl.2021.105627. View source
- J. Manfredi, E. Stapley, J. Nadeau, and D. Nash, “Investigation of the Effects of a Dietary Supplement on Insulin and Adipokine Concentrations in Equine Metabolic Syndrome/Insulin Dysregulation.,” Journal of Equine Veterinary Science, May 2020. doi: 10.1016/j.jevs.2020.102930. View source
- O. Akinniyi, A. Sackey, G. E. Ochube, and P. Mshelia, “A study on obesity and risk factors among leisure and polo horses in Kaduna State, Nigeria,” Ukrainian Journal of Veterinary and Agricultural Sciences, Aug. 2023. doi: 10.32718/ujvas6-2.05. View source
- N. Menzies‐Gow and E. Knowles, “Sodium‐glucose transport protein 2 inhibitor use in the management of insulin dysregulation in ponies and horses,” Journal of Veterinary Pharmacology and Therapeutics, Jul. 2024. doi: 10.1111/jvp.13470. View source
- K. Shell, A. Rijkenhuizen, and I. Vervuert, “Hyperandrogenemia and behavioural changes in a mare with equine metabolic syndrome,” Equine Veterinary Education, Jan. 2026. doi: 10.1111/eve.70048. View source
- A. Durham, N. Frank, C. McGowan, N. Menzies‐Gow, E. Roelfsema, I. Vervuert, K. Feige, and K. Fey, “ECEIM consensus statement on equine metabolic syndrome,” Journal of Veterinary Internal Medicine, Feb. 2019. doi: 10.1111/jvim.15423. View source
- A. Tomal, J. Szłapka-Kosarzewska, M. Mironiuk, I. Michalak, and K. Marycz, “Arthrospira platensis enriched with Cr(III), Mg(II), and Mn(II) ions improves insulin sensitivity and reduces systemic inflammation in equine metabolic affected horses,” Frontiers in Endocrinology, Apr. 2024. doi: 10.3389/fendo.2024.1382844. View source
- A. V. Münchow, S. T. Yttergren, R. R. Jakobsen, N. Luthersson, A. K. Hansen, and F. Lindenberg, “Oligosaccharide feed supplementation reduces plasma insulin in geldings with Equine Metabolic Syndrome,” Frontiers in microbiomes, Aug. 2023. doi: 10.3389/frmbi.2023.1194705. View source
- C. Loos, S. Dorsch, S. Elzinga, T. Brewster-Barnes, E. Vanzant, A. Adams, and K. Urschel, “A high protein meal affects plasma insulin concentrations and amino acid metabolism in horses with equine metabolic syndrome.,” The Veterinary Journal, Jul. 2019. doi: 10.1016/J.TVJL.2019.105341. View source
- S. L. Lewis, H. Holl, M. Long, M. Mallicote, and S. Brooks, “Use of principle component analysis to quantitatively score the equine metabolic syndrome phenotype in an Arabian horse population,” PLoS ONE, Jul. 2018. doi: 10.1371/journal.pone.0200583. View source
- A. M. Aboelmaaty, A. M. Ahdy, S. El-khodery, and M. Elgioushy, “Investigations on metabolic diseases of horses in Egypt,” Frontiers in Veterinary Science, Aug. 2025. doi: 10.3389/fvets.2025.1591090. View source
- A. Reynolds, J. Keen, T. Fordham, and R. Morgan, “Adipose tissue dysfunction in obese horses with equine metabolic syndrome,” Equine Veterinary Journal, Apr. 2019. doi: 10.1111/evj.13097. View source
- A. Biddle, J. Tomb, and Z. Fan, “Microbiome and Blood Analyte Differences Point to Community and Metabolic Signatures in Lean and Obese Horses,” Frontiers in Veterinary Science, Sep. 2018. doi: 10.3389/fvets.2018.00225. View source
This review is one of 34 in the Foxwatch research corpus. Methodology: PRISMA 2020-style systematic literature review against PubMed, CAB Abstracts, Scopus, Web of Science, and equine veterinary conference proceedings.











