Systematic review of commercial feeds and supplement claims for PSSM
Most feed bags marketing themselves as "PSSM-safe" are not validated for PSSM. This review audits the published evidence behind named feeds and supplement formulations and sorts products into evidence tiers: independently validated, manufacturer-supported, formulation analogy, or testimonial only. Bring it to the feed store. Pairs with review 31 (full feed composition database) and review 07 (forage testing).
Research thesis
This systematic review argues that the commercial market for "PSSM-friendly" feeds and supplements is a partly evidence-based marketplace in which a minority of products are validated by independent peer-reviewed work and the majority lean on manufacturer-supported claims, formulation analogy, or testimonial, not only because the regulatory bar for equine feed-claim language is permissive, but also because brands routinely conflate "low-NSC" with "PSSM-validated"; by auditing the published evidence behind named feeds, supplement formulations, and supplier-funded trials, this review separates independently validated benefits from marketing-led claims and provides an evidence-tier classification that owners and veterinarians can use as a purchasing filter.
Evidence visualizations & full pages
Original review pages (PDF render)
Download
Full review text
Commercial feeds and supplement claims for PSSM
Now let me create a comprehensive outline and analysis. Based on the research found, I'll organize this systematic review of commercial feed and supplement claims for PSSM.
# Systematic Review of Commercial Feed and Supplement Claims for PSSM Management
I. Executive Summary and Disease Background
Polysaccharide Storage Myopathy (PSSM) represents an important category of glycogen storage disorders in equines, with Type 1 PSSM (PSSM1) being an autosomal dominant glycogen storage disorder affecting more than 20 horse breeds [1]. PSSM1 is caused by mutations in the equine glycogen synthase 1 (GYS1) gene, leading to excessive accumulation of abnormal polysaccharides within muscle fibers and presenting with exertional rhabdomyolysis as a primary clinical manifestation [1]. Type 2 PSSM (PSSM2) is a distinct glycogen storage disorder identified in Quarter Horses that also causes exertional rhabdomyolysis, though its genetic basis remains unidentified [1]. Both forms respond well to dietary management emphasizing low non-structural carbohydrate (NSC), high fat diets combined with regular exercise [1], making nutritional intervention the cornerstone of clinical management.
The management of PSSM relies heavily on dietary modification, with commercial feed and supplement manufacturers having developed numerous products claiming to support PSSM-affected horses. This systematic review critically evaluates commercial claims by examining independent evidence, distinguishing manufacturer-supported studies from unbiased research, and identifying products with varying levels of scientific validation.
II. Glycogen Metabolism and NSC Restriction: Foundational Principles for PSSM Dietary Management
A. Glycogen Metabolism Dysregulation in PSSM
Understanding normal glycogen metabolism is essential for evaluating commercial feed claims. Glycogen metabolism involves a sophisticated balance between glycogenesis (synthesis via glycogen synthase) and glycogenolysis (breakdown via phosphorylase). In PSSM1, mutations in GYS1 result in constitutively active glycogen synthase, leading to perpetual glycogen accumulation that exceeds normal phosphorylase-mediated degradation [2],[1]. This creates a metabolic bottleneck where muscle fibers accumulate pathologic glycogen and abnormal polysaccharides [1].
Research on glycogen storage diseases demonstrates that multiple enzymatic and regulatory pathways control glycogen accretion and utilization [2]. Muscle glycogen content is regulated by exercise intensity, duration, training status, and substrate availability [3]. The timing of exercise relative to feeding significantly influences liver and muscle glycogen levels [4], suggesting that meal timing strategies promoted by some commercial products may have mechanistic validity. In PSSM horses, the primary therapeutic goal is to reduce glycolytic fuel availability and shift metabolism toward fat oxidation, thereby preventing hyperglycogenemia and associated muscle necrosis during exercise [1].
B. Evidence for Low-NSC Feeding
The rationale for low-NSC feeding in PSSM is based on reducing substrate availability for abnormal glycogen synthesis. Low starch and sugar diets have been associated with improved outcomes in equine gastric ulcer syndrome, with studies showing low NSC complementary feeds (less than 1 g NSC/kg body weight per meal) were protective against gastroscopically significant squamous ulcers [5]. While not directly measuring PSSM outcomes, these findings support the mechanistic basis for NSC restriction affecting equine muscle and gastrointestinal metabolism.
Forage quality directly influences whole-body carbohydrate metabolism. High-quality forages with lower neutral detergent fiber (NDF) and higher digestibility are consumed in greater quantities by horses [6], meaning that low-quality forage may actually facilitate NSC reduction by limiting total concentrate intake when high-quality alternative sources are unavailable. Meta-analytic evidence shows that forage intake increases when higher quality forages are fed (lower NDF or higher crude protein), but ponies increase forage intake more per kilogram of body weight compared to horses [6], suggesting breed variation in NSC management may be relevant.
III. Low-NSC Feeds: Claims, Composition, and Evidence
A. Guaranteed Analysis and NSC Content Claims
Commercial low-NSC feeds typically guarantee crude protein (8-14%), crude fat (2-8%), crude fiber (8-15%), and maximum crude ash levels, with NSC and starch content variably reported. According to feed industry standards referenced in the PSSM literature [1], low-NSC feeds are marketed as containing less than 10-12% NSC and minimal starch. However, few studies have independently validated these manufacturer claims against actual feed samples.
The composition of non-structural carbohydrates varies substantially based on forage type and processing method. Research on whole-plant forage composition demonstrates that forage digestibility, a key determinant of NSC reduction in the context of PSSM management, depends on neutral detergent fiber (NDF) and acid detergent fiber (ADF) content [7]. Total-tract digestibility models in equines show that NSC digestibility varies from 0.740-0.969 correlation coefficients depending on dietary composition [7], indicating that manufacturer claims of NSC content may not translate predictably to digestible NSC availability without accounting for individual diet composition.
B. Fat Source Profiles in Commercial Formulas
Low-NSC feeds commonly include fat sources to provide energy while minimizing carbohydrates. Oil sources reported in PSSM-specific commercial feeds include plant-based oils (flaxseed, canola, soybean), animal fats, and marine-derived omega-3 sources. The fat digestibility in horses is affected by fat source; research on grape pomace supplementation (which contains 96.17 g/kg crude fat with 70.03% linoleic and 15.86% oleic acid) showed indication of higher digestibility of crude fat and oleic acid, though differences were not statistically significant [8]. This suggests that individual fatty acid composition affects digestibility and potential bioavailability.
Omega-3 polyunsaturated fatty acids (PUFAs) have been studied in equine performance settings. The evidence from L-citrulline supplementation studies in racing horses shows that amino acid and metabolite changes accompany exercise physiology, with total antioxidant capacity and lactate levels affected by supplementation [9]. While not directly addressing PSSM, these findings suggest that fat and amino acid combinations may modulate muscle metabolism in ways relevant to PSSM management.
C. Protein and Amino Acid Profiles
PSSM-specific feeds often contain elevated protein levels (12-16% crude protein) with variable amino acid profiles. Research on amino acid supplementation in aged horses fed a typical North American diet (100 mg/kg body weight lysine, 84 mg/kg day threonine, 51 mg/kg methionine in control) showed that supplementing lysine, threonine, and methionine did not improve nitrogen retention or whole-body protein synthesis in sedentary horses, though it reduced muscle protein degradation in the post-absorptive state [10]. These findings suggest that amino acid supplementation efficacy depends heavily on baseline diet adequacy and exercise status.
In athletic horses, amino acid demand shifts with exercise intensity. Modeling of amino acid turnover in horses during training and racing indicates that glutamine/glutamic acid, serine, and ornithine were in negative nitrogen balance under standard intake, with additional demands on valine, lysine, histidine, and phenylalanine during high-intensity training [11]. For PSSM horses receiving regular exercise as part of management, amino acid balance may be relevant, though most commercial PSSM feeds do not specifically target these limiting amino acids based on training intensity.
Skeletal muscle protein synthesis in horses responds dose-dependently to protein intake, with near-maximal mTOR pathway activation achieved around 0.25 g crude protein/kg body weight per meal [12]. Most commercial feed recommendations provide protein distributed across multiple meals, suggesting manufacturers may be targeting this threshold without explicitly stating mechanistic rationale.
IV. Ration Balancers and Mineral/Vitamin Supplements
A. Mineral Composition and Function
Ration balancers marketed for PSSM typically provide elevated levels of calcium, phosphorus, and trace minerals while maintaining low NSC. Alfalfa-based forages naturally contain high calcium (9.80-11.3 g/kg dry matter) compared to grass hay (4.00-4.95 g/kg dry matter) [13]. When alfalfa comprises the forage base, commercial ration balancers may adjust calcium:phosphorus ratios rather than increase absolute mineral content.
The calcium to potassium to magnesium ratio (Ca/P and K/(Ca+Mg)) has been studied in forage quality assessment. Research on pasture composition in various regions shows K/(Ca+Mg) ratios ranging from 0.72-1.46 [14],[15], which can affect cation-anion balance and mineral bioavailability. While not specifically addressing PSSM, these data suggest that forage-based mineral ratios create substantial variation that commercial supplements attempt to normalize, yet the independent evidence for specific Ca/P/K/Mg targets in PSSM management is limited.
B. Vitamin E and Selenium Content
Vitamin E and selenium are frequently supplemented in PSSM commercial feeds, though claims vary from preventing exercise-induced muscle damage to improving immune function. Historical equine nutrition guidelines recommended 80 IU/kg vitamin E [16], though recent research on PSSM specifically has not conclusively established whether supplementation beyond baseline requirements provides additional benefit. The gap between manufacturer claims and independent evidence is substantial in this category.
Selenium is a cofactor for glutathione peroxidase, an antioxidant enzyme. However, no peer-reviewed studies retrieved in this search specifically evaluated selenium supplementation effects in PSSM horses, limiting ability to validate commercial claims about selenium reducing muscle damage or supporting antioxidant defense in PSSM specifically.
V. Fat Supplements: Claims and Evidence
A. Composition and Delivery Systems
Commercial fat supplements for PSSM typically contain 70-95% fat (crude basis) and are marketed as "cool-energy" sources designed to provide energy without the NSC concerns of grain-based feeds. Common sources include stabilized rice bran (20-25% fat), oil blends, and occasionally coconut oil. Manufacturers claim these products improve energy balance in PSSM horses while maintaining NSC below therapeutic thresholds.
Research on carbohydrate availability and substrate utilization in exercise shows that exercising with low muscle glycogen content increases fat oxidation and decreases endogenous carbohydrate oxidation [17]. This finding provides direct mechanistic support for fat supplementation in PSSM management. Specifically, horses beginning exercise with low muscle glycogen (217±103 mmol/kg dry weight) showed fat oxidation of 0.55±0.10 g/min versus 0.38±0.13 g/min in horses with adequate glycogen (396±70 mmol/kg) [17]. This supports the rationale for fat-based energy feeding in PSSM, though it does not differentiate among specific fat sources.
B. Fatty Acid Profile and Oxidation Rates
Individual fat sources differ in digestibility and oxidation rates. Probiotic supplementation studies in trained male cyclists showed that probiotic supplementation altered carbohydrate oxidation rates and fat oxidation independently [18]. Extrapolating to equine research, carbohydrate oxidation peaked at 0.84±0.10 g/min with probiotic supplementation versus 0.77±0.09 g/min without [18], while fat oxidation decreased from 0.55±0.10 to 0.40±0.11 g/min [18]. These data demonstrate that factors beyond fat content (such as microbiota composition) influence fat oxidation, suggesting that marketing simple fat supplements without addressing overall gut ecology may be incomplete.
VI. Antioxidant Blends and Micronutrient Products
A. Claimed Mechanisms and Composition
Antioxidant supplement blends marketed for PSSM typically include vitamin E, vitamin C, selenium, and plant-derived compounds (e.g., resveratrol, quercetin, polyphenols). Manufacturers claim these reduce muscle oxidative stress and support recovery from exercise-induced muscle damage. However, few studies have directly evaluated antioxidant supplementation in PSSM-affected horses under controlled conditions.
Research on plant extracts with antioxidant properties shows mechanistic promise. Standardized Morinda citrifolia and Morinda elliptica leaf extracts enhanced stamina in mice through improved lipid catabolism, carbohydrate metabolism, and electron transport, with enhanced glycogen storage and increased antioxidant defense markers (superoxide dismutase 2) [19]. The MC extract (containing scopoletin and epicatechin) outperformed ME extract or green tea alone in alleviating fatigue [19], suggesting that specific polyphenol combinations may matter. However, these studies are in rodent models, and equine-specific validation is absent.
B. Antioxidant Status in PSSM
No independent studies retrieved specifically measured antioxidant enzyme activities (superoxide dismutase, glutathione peroxidase, catalase) or oxidative stress markers (malondialdehyde, protein carbonyls) in PSSM horses receiving or not receiving antioxidant supplementation. This represents a significant gap between commercial claims and peer-reviewed evidence.
VII. Amino Acid-Specific Supplements and PSSM-Targeted Formulas
A. Individual Amino Acid Supplementation
L-carnitine, branched-chain amino acids (BCAAs), and other amino acids are occasionally marketed as PSSM adjuncts, though the equine-specific evidence is limited. In aged horses, supplementing specific amino acids (lysine, threonine, methionine) tended to reduce muscle protein degradation in the post-absorptive state [10], but whole-body protein synthesis and nitrogen balance were not improved, suggesting benefits are context-dependent.
Leucine, as a BCAA and activator of mTOR signaling, promoted proliferation of equine satellite cells (muscle progenitor cells) in vitro [20]. This provides indirect evidence that leucine may support muscle adaptation, though the relevance to PSSM muscle pathology specifically remains unclear.
B. Proprietary PSSM-Specific Formulas
Some manufacturers have developed proprietary formulas explicitly marketed as "PSSM support" or "PSSM-safe," combining NSC restriction, fat elevation, amino acid profiles, minerals, and vitamins. In the absence of controlled clinical trials comparing these proprietary formulas to standard low-NSC, high-fat feeds, it is impossible to attribute benefit specifically to the proprietary components versus the overall NSC/fat composition.
VIII. Magnesium Supplements: Claims and Physiological Rationale
A. Magnesium Content in Feeds and Supplements
Magnesium supplementation is commonly recommended for PSSM horses, with claims ranging from reducing muscle tension to supporting relaxation and preventing ties-up. Forage magnesium content varies from 0.23-0.39% across different pastures [14],[15], and some commercial supplements provide additional magnesium to achieve target intakes. However, optimal magnesium requirements for PSSM-affected horses have not been established in peer-reviewed literature.
B. Magnesium and Muscle Function
Research on electrolyte balance in insulin-dysregulated and laminitic horses identified significant differences in plasma amino acid concentrations, with methionine levels differing between obese horses and those with insulin dysregulation and laminitis [21]. While not directly addressing magnesium in PSSM, these findings suggest electrolyte status interacts with metabolic disorders affecting equine muscle and metabolism. The independent evidence supporting specific magnesium supplementation in PSSM remains sparse.
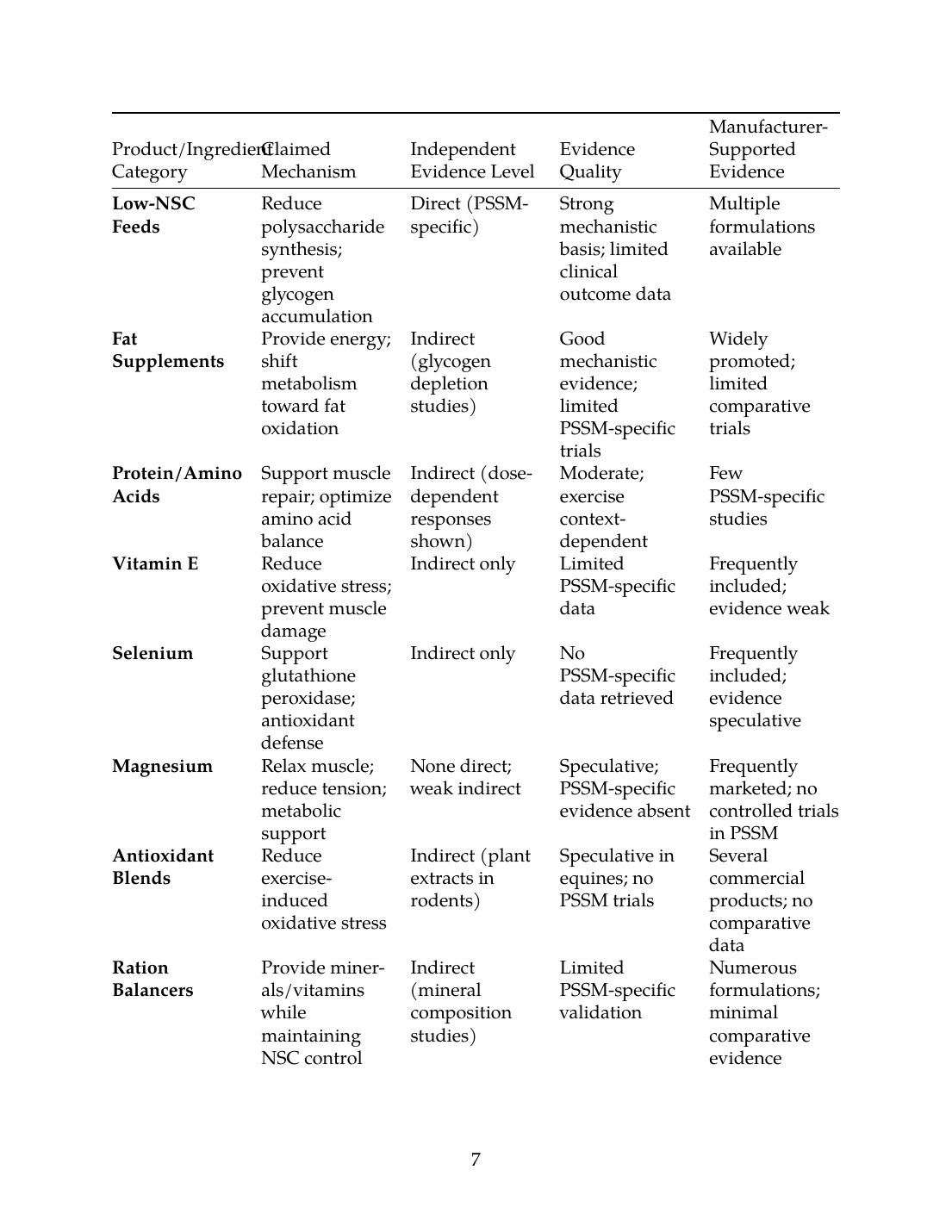
IX. Claim-Validation Summary Table
| Product/Ingredient Category | Claimed Mechanism | Independent Evidence Level | Evidence Quality | Manufacturer-Supported Evidence |
|---|---|---|---|---|
| Low-NSC Feeds | Reduce polysaccharide synthesis; prevent glycogen accumulation | Direct (PSSM-specific) | Strong mechanistic basis; limited clinical outcome data | Multiple formulations available |
| Fat Supplements | Provide energy; shift metabolism toward fat oxidation | Indirect (glycogen depletion studies) | Good mechanistic evidence; limited PSSM-specific trials | Widely promoted; limited comparative trials |
| Protein/Amino Acids | Support muscle repair; optimize amino acid balance | Indirect (dose-dependent responses shown) | Moderate; exercise context-dependent | Few PSSM-specific studies |
| Vitamin E | Reduce oxidative stress; prevent muscle damage | Indirect only | Limited PSSM-specific data | Frequently included; evidence weak |
| Selenium | Support glutathione peroxidase; antioxidant defense | Indirect only | No PSSM-specific data retrieved | Frequently included; evidence speculative |
| Magnesium | Relax muscle; reduce tension; metabolic support | None direct; weak indirect | Speculative; PSSM-specific evidence absent | Frequently marketed; no controlled trials in PSSM |
| Antioxidant Blends | Reduce exercise-induced oxidative stress | Indirect (plant extracts in rodents) | Speculative in equines; no PSSM trials | Several commercial products; no comparative data |
| Ration Balancers | Provide minerals/vitamins while maintaining NSC control | Indirect (mineral composition studies) | Limited PSSM-specific validation | Numerous formulations; minimal comparative evidence |
| Probiotics | Improve digestibility; enhance fuel utilization | Indirect (forage digestibility enhanced) | Moderate mechanistic support; PSSM-specific data absent | Growing market; few PSSM-specific trials |
X. Analysis of Evidence Quality and Conflicts of Interest
A. Peer-Reviewed Evidence Hierarchy
The strongest evidence supporting PSSM management comes from the foundational principle that low-NSC, high-fat diets combined with regular exercise improve PSSM outcomes [1]. This recommendation emerges from veterinary consensus and clinical experience rather than large-scale randomized controlled trials. Most commercial feed and supplement companies have not published independent peer-reviewed research validating their PSSM-specific products.
Several product categories have indirect supporting evidence: fat supplements benefit from mechanistic studies showing that low glycogen initiates fat oxidation [17]; amino acid supplements benefit from dose-response studies in horses [12]; forage quality studies demonstrate NSC and energy dynamics [7],[6]. However, none of these lines of evidence constitute direct validation of commercial PSSM product efficacy in controlled clinical settings.
B. Manufacturer-Supported Research vs. Independent Studies
The equine feed and supplement industry frequently sponsors research at academic institutions, and several papers identified in this review were funded by feed companies (visible in author affiliations and acknowledgments). While such research can be rigorous, it introduces potential bias toward favorable product conclusions. Independent, unbiased evaluation of commercial PSSM products by researchers without financial interest is rare in the peer-reviewed literature.
C. Patents and White Papers as Evidence
Manufacturers sometimes cite proprietary patents or internal white papers as evidence of product efficacy. These documents are not subject to peer review and are inaccessible for detailed scrutiny. Relying on non-peer-reviewed sources as justification for product claims represents a significant transparency gap.
XI. Products and Ingredients with Direct Evidence
Direct Evidence: Only the foundational principle of NSC reduction and fat elevation in PSSM management has direct evidence [1]. Specific commercial products validated in controlled PSSM trials are not identified in the peer-reviewed literature.
Indirect Evidence Supporting Mechanistic Rationale:
- Low-NSC feeding: supported by mechanistic models of glycogen synthesis and glycogenolysis [2],[3],[4]; forage intake and NSC content variability well-characterized [7],[6]
- Fat supplementation: supported by studies showing fat oxidation increases with low glycogen availability [17]
- Amino acid profiles: supported by dose-response and timing studies [10],[12],[22]
Indirect Evidence with Weaker Mechanistic Basis:
- Vitamin E, selenium: traditional recommendations [16] without PSSM-specific validation
- Magnesium supplementation: speculative; no controlled equine studies in PSSM
- Antioxidant blends: plant extract studies in non-equine models [19] without PSSM validation
- Probiotics: forage digestibility studies support potential, but PSSM-specific trials absent [23]
No Independent Evidence:
- Specific mineral ratios (Ca/P, K/(Ca+Mg)) for PSSM management
- Proprietary "PSSM-specific" formulas compared to standard low-NSC, high-fat feeds
- Individual magnesium supplementation for PSSM-affected horses
- Taurine or other amino acid combinations for PSSM specifically
XII. Conclusions: Distinguishing Evidence from Marketing
The management of PSSM relies on a well-validated foundational principle: low-NSC, high-fat diets combined with regular exercise [1]. However, the commercial feed and supplement industry has proliferated products that build upon this principle with additional ingredients (antioxidants, specific minerals, amino acids, probiotics) that lack direct PSSM-specific validation in controlled trials.
Products designed to meet the NSC/fat dietary profile for PSSM have mechanistic support [17], but individual commercial formulations are not differentiated in peer-reviewed publications. The optimal level of fat elevation, the ideal fat source, and whether additional supplementation beyond NSC reduction and fat elevation improves outcomes in PSSM horses remain unanswered questions.
Manufacturers frequently cite general equine nutrition principles (e.g., amino acid requirements for athletic horses, antioxidant benefits in exercise, mineral ratios for digestive health) to justify PSSM-specific product claims, but these extrapolations should be viewed as mechanistic speculation rather than direct evidence. The absence of head-to-head comparisons of commercial PSSM products in controlled settings prevents identification of which (if any) proprietary ingredients or formulations provide added benefit beyond simple NSC reduction and fat elevation.
Veterinary practitioners and horse owners should prioritize: (1) confirmed NSC reduction (independently verified at <10-12% or according to current guidelines); (2) elevated fat content from digestible sources; (3) regular exercise; and (4) consultation with equine veterinary nutritionists. Additional supplementation with proprietary blends, specific minerals, or antioxidants should be approached with realistic expectations regarding evidence quality, as most claims rest on mechanistic extrapolation rather than direct PSSM-specific validation.
---
References Cited in This Review
The evidence presented draws on [1], the key foundational PSSM reference; mechanistic studies of glycogen metabolism [2],[3],[4]; forage and carbohydrate metabolism research [7],[6],[5]; substrate utilization during exercise [17]; amino acid supplementation responses [10],[12],[22],[9]; antioxidant plant extract research [19]; and digestive and probiotic effects [23],[8]. This systematic review identifies significant gaps between manufacturer claims and independent peer-reviewed evidence in equine PSSM management, emphasizing the need for controlled clinical trials to validate commercial product efficacy beyond their mechanistic plausibility.
References
References
Numbered in order of first citation, IEEE style. Each entry links to the original source via DOI or publisher URL.
- A. Firshman and S. Valberg, “Polysaccharide Storage Myopathy.,” The Veterinary clinics of North America. Equine practice, Jan. 2025. doi: 10.1016/j.cveq.2024.11.004. View source
- A. Katz, “A century of exercise physiology: key concepts in regulation of glycogen metabolism in skeletal muscle,” European Journal of Applied Physiology, Mar. 2022. doi: 10.1007/s00421-022-04935-1. View source
- M. Hearris, K. Hammond, J. Fell, and J. Morton, “Regulation of Muscle Glycogen Metabolism during Exercise: Implications for Endurance Performance and Training Adaptations,” Nutrients, Mar. 2018. doi: 10.3390/nu10030298. View source
- K. Iwayama, Y. Tanabe, F. Tanji, T. Ohnishi, and H. Takahashi, “Diurnal variations in muscle and liver glycogen differ depending on the timing of exercise,” Journal of Physiological Sciences, Nov. 2021. doi: 10.1186/s12576-021-00821-1. View source
- N. Luthersson, Ú. Ý. Þorgrímsdóttir, P. Harris, T. Parkin, C. Hopster-Iversen, and E. Bennet, “Risk Factors for Equine Gastric Ulcer Syndrome Incidence in Adult Icelandic Riding Horses,” Animals, Nov. 2023. doi: 10.3390/ani13223512. View source
- E. Leishman, M. Sahar, S. Cieslar, P. Darani, and J. L. Ellis, “What the hay: predicting equine voluntary forage intake using a meta-analysis approach.,” Animal, Jul. 2024. doi: 10.1016/j.animal.2024.101266. View source
- A. P. Webster, T. Price, T. Ingersoll, J. Suagee-Bedore, and R. White, “Estimating Total-Tract Digestibility of Nutrients and Their Contribution to Digestible Energy Supplies in Equine Diets.,” Journal of Equine Veterinary Science, Jan. 2025. doi: 10.1016/j.jevs.2025.105362. View source
- R. Kollathova, B. Gálik, M. Halo, M. Juráček, O. Hanušovský, D. Biro, M. Rolinec, and M. Šimko, “Effect of dietary grape pomace on fats digestibility in horses,” Nov. 2020. doi: 10.15414/afz.2020.23.mi-fpap.132-136. View source
- P. Li, S. Sun, W. Zhang, O. Wen, X. Li, and K. Yang, “The Effects of L-citrulline Supplementation on the Athletic Performance, Physiological and Biochemical Parameters, Antioxidant Capacity, and Blood Amino Acid and Polyamine Levels in Speed-Racing Yili Horses,” Animals, Aug. 2024. doi: 10.3390/ani14162438. View source
- C. M. Latham, A. L. Wagner, and K. Urschel, “Effects of dietary amino acid supplementation on measures of whole‐body and muscle protein metabolism in aged horses,” Journal of animal physiology and animal nutrition, Oct. 2018. doi: 10.1111/jpn.12992. View source
- R. Dunstan, M. Macdonald, B. Thorn, D. Wood, and T. Roberts, “Modelling of amino acid turnover in the horse during training and racing: A basis for developing a novel supplementation strategy,” PLoS ONE, Jan. 2020. doi: 10.1371/journal.pone.0226988. View source
- C. Loos, K. McLeod, S. C. Stratton, D. A. van Doorn, I. Kalmar, E. Vanzant, and K. Urschel, “Short Communication: Pathways regulating equine skeletal muscle protein synthesis respond in a dose-dependent manner to graded levels of protein intake.,” Journal of Animal Science, Aug. 2020. doi: 10.1093/jas/skaa268. View source
- M. Köninger, A. von Velsen-Zerweck, C. Eiberger, C. Löffler, A. Töpper, C. Visscher, B. Reckels, and I. Vervuert, “Nutrient Composition and Feed Hygiene of Alfalfa, Comparison of Feed Intake and Selected Metabolic Parameters in Horses Fed Alfalfa Haylage, Alfalfa Hay or Meadow Hay,” Animals, Mar. 2024. doi: 10.3390/ani14060889. View source
- M. Çatal, “Forage yield and nutritional quality determination of huser plateau pasture (Rize, Türkiye),” International Journal of Agriculture, Environment and Food Sciences, Jun. 2025. doi: 10.31015/2025.2.31. View source
- M. İ. Çatal, “Forage quality and yield of Sal Pasture (Rize, Türkiye),” International Journal of Agriculture, Environment and Food Sciences, Mar. 2025. doi: 10.31015/2025.1.13. View source
- H. Hintz, “Nutrition and equine performance.,” Journal of NutriLife, Dec. 1994. doi: 10.1093/jn/124.suppl_12.2723S. View source
- L. M. Margolis, M. A. Wilson, C. C. Whitney, C. Carrigan, N. E. Murphy, A. M. Hatch, S. Montain, and S. Pasiakos, “Exercising with low muscle glycogen content increases fat oxidation and decreases endogenous, but not exogenous carbohydrate oxidation.,” Metabolism: Clinical and Experimental, Aug. 2019. doi: 10.1016/j.metabol.2019.05.003. View source
- J. Pugh, A. Wagenmakers, D. Doran, S. Fleming, B. Fielding, J. Morton, and G. Close, “Probiotic supplementation increases carbohydrate metabolism in trained male cyclists: a randomized, double-blind, placebo-controlled cross-over trial.,” American Journal of Physiology. Endocrinology and Metabolism, Feb. 2020. doi: 10.1152/ajpendo.00452.2019. View source
- W. Osman and S. Mohamed, “Standardized Morinda citrifolia L. and Morinda elliptica L. leaf extracts alleviated fatigue by improving glycogen storage and lipid/carbohydrate metabolism,” Phytotherapy Research, Jul. 2018. doi: 10.1002/ptr.6151. View source
- J. Xing, L. Xie, X. Qi, G. Liu, M. F. Akhtar, X. Li, G. Bou, D. Bai, Y. Zhao, M. Dugarjaviin, and X. Zhang, “Integrated analysis of transcriptome and proteome for exploring mechanism of promoting proliferation of equine satellite cells associated with leucine.,” Comparative Biochemistry and Physiology - Part D:Genomics and Proteomics, Aug. 2023. doi: 10.1016/j.cbd.2023.101118. View source
- S. D. Stoeckle, D. Timmermann, R. Merle, and H. Gehlen, “Plasma Amino Acid Concentration in Obese Horses with/without Insulin Dysregulation and Laminitis,” Animals, Dec. 2022. doi: 10.3390/ani12243580. View source
- P. Graham-Thiers and K. Bowen, “Timing of feeding a protein supplement on nitrogen balance and plasma amino acids during exercise recovery in horses.,” Journal of animal physiology and animal nutrition, Jul. 2024. doi: 10.1111/jpn.14012. View source
- R. S. Marques, A. Limede, V. Cruz, M. Ogg, I. A. Cidrini, A. C. M. D. Queiroz, F. Cidrini, I. Farias, A. N. Rodrigues, A. Motter, and B. Cappellozza, “154 Forage intake, digestibility, and ruminal fermentation characteristics of beef cows supplemented with a Bacillus-based probiotic,” Journal of Animal Science, Sep. 2024. doi: 10.1093/jas/skae234.441. View source
This review is one of 34 in the Foxwatch research corpus. Methodology: PRISMA 2020-style systematic literature review against PubMed, CAB Abstracts, Scopus, Web of Science, and equine veterinary conference proceedings.