Mechanistic review of carbohydrate metabolism in PSSM
Why does low-sugar feeding actually work? This review reads the molecular biology of the GYS1 mutation, explains how a low-sugar high-fat ration shifts skeletal muscle toward fatty acid oxidation, and connects each clinical recommendation back to its underlying biochemistry. Useful for veterinarians explaining the disease to owners and for owners who want the science behind the rules. Pairs with reviews 01 (foundation) and 03 (high-fat).
Research thesis
This mechanistic review argues that the GYS1 R309H gain-of-function mutation is the primary biochemical driver of PSSM1, and that insulin sensitivity, post-prandial glucose dynamics, fiber-type composition, and mitochondrial substrate utilization together explain why low-NSC, high-fat feeding actually works, not only because the mutation produces a constitutively active glycogen synthase that converts excess glucose into amylase-resistant polysaccharide whether or not insulin signals it to, but also because muscle fat-oxidation capacity in affected horses is preserved and can be deliberately exploited by ration design; by reviewing the molecular biology, exercise physiology, and clinical feeding literature, this review constructs a mechanistic pathway connecting each clinical recommendation to its underlying biochemistry and identifies the unresolved mechanistic questions that current dietary guidance leaves unanswered.
Evidence visualizations & full pages
Evidence figures (1)
Figures referenced by the review's Figure N: captions. Click any chart for a full-size view. They are also rendered inline below their respective captions in the full review text further down.
Original review pages (PDF render)
Download
Full review text
Carbohydrate metabolism in PSSM
Mechanistic Review of Carbohydrate Metabolism in Polysaccharide Storage Myopathy (PSSM) and Dietary Management Implications
Executive Summary
Polysaccharide storage myopathy (PSSM) represents a glycogen storage disorder characterized by abnormal glycogen accumulation in equine skeletal muscle, leading to recurrent episodes of exertional rhabdomyolysis. This mechanistic review synthesizes current understanding of the molecular pathophysiology underlying PSSM, with particular emphasis on the GYS1 mutation effects, insulin dynamics, mitochondrial dysfunction, and their collective implications for evidence-based dietary and exercise management strategies.
---
1. Genetic Basis and Glycogen Synthase Dysfunction
1.1 The GYS1 R309H Mutation
Type 1 PSSM (PSSM1) is caused by a dominant point mutation in the glycogen synthase 1 gene (GYS1), which encodes the skeletal muscle isoform of glycogen synthase (GS), the rate-limiting enzyme in glycogen synthesis [1]. Genome-wide association studies identified this mutation as a G-to-A base substitution resulting in an arginine-to-histidine amino acid change at codon 309 (R309H) in a highly conserved region of the enzyme [1]. This represents the first documented gain-of-function mutation in GYS1 resulting in a glycogenosis, distinguishing PSSM from other glycogen storage diseases that typically involve loss-of-function mutations in glycogenolytic or glycolytic enzymes [2].
The R309H mutation causes constitutive activation of glycogen synthase, shifting the equilibrium of glycogen synthesis and breakdown decidedly toward synthesis [3]. Functional analysis has demonstrated significantly elevated GS activity in PSSM-affected horses compared to controls, even in the absence of the allosteric regulator glucose-6-phosphate (G6P) [4]. This constitutive activation occurs despite compensatory increases in inhibitory phosphorylation at sites 2+2a and elevated AMPKα1 expression, reflecting physiological attempts to reduce enzyme activity that ultimately prove insufficient [4].
1.2 Prevalence and Breed Distribution
The GYS1 mutation has been identified in more than 20 horse breeds, with prevalence estimates ranging from 6-10% in Quarter Horses to up to 36% in draft breeds [5]. Within Quarter Horse subpopulations, halter horses show the highest prevalence at approximately 28% [6]. The mutation is believed to represent an ancient polymorphism that predates the establishment of modern horse breeds, potentially having provided a selective advantage under historical management conditions involving daily work and limited feed availability [7]. The presence of this allele across diverse breeds suggests it was positively selected for as a "thrifty" genotype that enhanced glycogen storage capacity during periods of nutritional scarcity [8].
1.3 PSSM Type 2: A Distinct Entity
Horses diagnosed with PSSM by muscle histopathology who lack the GYS1 mutation are classified as having Type 2 PSSM (PSSM2) [9]. Unlike PSSM1, where muscle glycogen concentrations average 1.8-fold higher than normal, PSSM2 horses do not consistently demonstrate significantly elevated glycogen levels above reference ranges [10]. The specific molecular cause of PSSM2 remains unknown, raising questions about whether current dietary recommendations optimally address the underlying pathophysiology in these horses [10].
---
2. Insulin Response and Postprandial Glucose Dynamics
2.1 Enhanced Insulin Sensitivity in PSSM1
Research utilizing euglycemic hyperinsulinemic clamp studies has established that PSSM horses exhibit enhanced whole-body insulin sensitivity, requiring higher rates of glucose infusion to maintain euglycemia throughout the clamp procedure [11]. This heightened insulin sensitivity manifests as early as 6 months of age and persists throughout life [12]. When insulin is released in response to carbohydrate consumption, it stimulates the already overactive glycogen synthase enzyme in PSSM1 muscle, further exacerbating glycogen accumulation [13].
The molecular basis for enhanced glucose uptake involves the glucose transporter system, with PSSM horses demonstrating increased glucose uptake into muscle despite lower GLUT1 content in some studies [11]. The insulin receptor content and GLUT4 transporter dynamics facilitate rapid glucose clearance from circulation, providing substrate for the constitutively active glycogen synthase. Importantly, muscle can also take up glucose through insulin-independent pathways, meaning the enhanced GYS1 activity creates a continuous "hunger" for glucose in the muscle cell regardless of circulating insulin levels [5].
2.2 Postprandial Glycemic Response
The postprandial glycemic and insulinemic responses in PSSM horses are significantly influenced by dietary composition. Horses with PSSM fed hay containing 17% non-structural carbohydrates (NSC) demonstrate substantially elevated insulin concentrations compared to those fed hay with 12% or 4% NSC [13]. These elevated postprandial insulin concentrations directly stimulate glycogen synthase activity, creating a mechanistic link between dietary carbohydrate content and disease severity. The threshold effect observed around 12% NSC provides a rational basis for dietary recommendations targeting this level or below.
Studies examining oral glucose tolerance tests in Haflinger horses with the GYS1 mutation have not demonstrated significant differences between affected and control groups in standardized testing conditions [14]. However, clinical observations consistently support that high-starch diets exacerbate episodes of exertional rhabdomyolysis [15]. This apparent paradox may reflect differences between acute glucose challenge responses and chronic dietary exposure effects on muscle glycogen accumulation and cellular function.
---
3. Muscle Glycogen Accumulation and Polysaccharide Abnormalities
3.1 Quantitative Glycogen Excess
Muscle glycogen concentrations in PSSM1 horses average 1.8-fold higher than in unaffected horses [16]. This excessive accumulation occurs despite apparently normal glycogenolytic enzyme activities, indicating that the primary defect lies in synthesis rather than utilization pathways [17]. The accumulated glycogen displays abnormal structural characteristics, with iodine absorption spectra analysis revealing less-branched polysaccharide compared to normal glycogen [11].
The abnormal polysaccharide formed in PSSM1 is often resistant to amylase digestion, appearing as crystalline inclusions on histopathological examination [18]. This amylase-resistant polysaccharide likely results from the relative overactivity of glycogen synthase compared to branching enzyme, producing straight-chain polymers rather than the highly branched normal glycogen structure [18]. Grade 2 polysaccharide accumulation (typically amylase-resistant) shows the strongest association with the GYS1 mutation, with prevalences of 68-87% in Quarter Horse-related breeds with this grade classification [18].
3.2 Histopathological Features
Histological evaluation of PSSM muscle reveals periodic acid-Schiff (PAS)-positive aggregates in the cytoplasm or subsarcolemmal regions of muscle fibers [19]. These accumulations can displace and partially replace mitochondria and myofibrils, contributing to structural disruption of the contractile apparatus. Ultrastructural evaluation demonstrates decreased mitochondrial number alongside accumulation of abnormal polysaccharide [19]. Chronic myopathic changes including fiber necrosis, anisocytosis, endomysial fibrosis, and fatty infiltration may be observed, particularly in homozygous affected horses who typically exhibit more severe histological disorders [19].
---
4. Skeletal Muscle Fiber Type Composition
4.1 Fiber Type Distribution in PSSM
The relationship between muscle fiber type and PSSM pathophysiology is complex and potentially breed-specific. Studies in Haflinger horses with PSSM1 have demonstrated significantly more type 2A fibers and fewer type 2X fibers in affected semitendinosus muscle compared to controls [14]. Type 2A fibers possess greater oxidative capacity than type 2X fibers and may represent an adaptive response or predisposing factor for disease expression.
Muscle fiber type composition influences metabolic substrate utilization patterns. Type I (slow-twitch oxidative) fibers have the highest mitochondrial density and rely predominantly on fatty acid oxidation, while type IIX (fast-twitch glycolytic) fibers have the lowest oxidative capacity and depend more heavily on glycogenolysis for energy production [20]. The altered fiber type distribution observed in PSSM muscles may reflect chronic metabolic stress or represent a compensatory shift toward oxidative metabolism.
4.2 Implications for Energy Metabolism
The citrate synthase (CS) activity, a marker of mitochondrial density, correlates positively with type I fiber proportion and negatively with type 2X fiber percentage [20]. However, substantial variability exists, with some horses demonstrating similar CS activities despite markedly different fiber type compositions. This suggests that training status and other factors beyond fiber type determine oxidative capacity. Exercise training increases mitochondrial volume density by approximately 50% in unfit Quarter Horses [20], highlighting the plasticity of muscle oxidative capacity and its potential for therapeutic modulation.
---
5. Mitochondrial Metabolism and Oxidative Dysfunction
5.1 Impaired Oxidative Phosphorylation
High-resolution respirometry studies have revealed significant mitochondrial dysfunction in PSSM-affected horses. The gluteus medius muscle from PSSM horses demonstrates decreased oxidative phosphorylation (OXPHOS) and electron transfer (ET) capacities compared to healthy controls when assessed with complex I substrates (glutamate, malate, and succinate) [21]. These deficits indicate impaired function of the electron transport chain, specifically involving complex I-linked respiration.
When mitochondrial capacities are expressed as ratios, both PSSM and other rhabdomyolysis-affected horses show greater contribution of the NADH pathway (feeding electrons into Complex I) to maximal OXPHOS or ET capacity compared to healthy horses [21]. This altered substrate preference suggests compensatory metabolic adaptations that may prove insufficient under exercise stress. The specific modifications in mitochondrial function likely contribute to the pathogenesis of PSSM and explain the energy deficit observed during submaximal exercise [22].
5.2 Ultrastructural Mitochondrial Changes
Ultrastructural evaluation reveals decreased mitochondrial numbers and structural abnormalities in PSSM muscle [19]. The accumulation of abnormal polysaccharide physically displaces mitochondria within muscle fibers, potentially compromising energy production capacity independent of intrinsic mitochondrial dysfunction. This combined structural and functional mitochondrial impairment creates a significant barrier to adequate ATP generation during exercise.
The relationship between glycogen accumulation and mitochondrial dysfunction may be bidirectional. Gene expression analysis has identified modulation of genes involved in oxidative energy metabolism, with evidence of chronic hypoxia-like signaling associated with very low activity of oxidative energetic pathways in PSSM muscle [19]. Whether mitochondrial dysfunction is a primary pathogenic factor or a secondary consequence of glycogen accumulation remains an important mechanistic question.
---
6. Fat Oxidation and Alternative Fuel Utilization
6.1 Impaired Fatty Acid Metabolism in High-Carbohydrate Conditions
When PSSM horses are fed grain-based diets high in non-structural carbohydrates, fatty acid oxidation becomes significantly impaired through multiple mechanisms [23]. High insulin concentrations following carbohydrate meals suppress lipolysis, reducing the availability of free fatty acids as fuel substrates. Additionally, elevated muscle citrate concentrations activate acetyl-CoA carboxylase, which converts acetyl-CoA to malonyl-CoA—the committed step for fatty acid synthesis.
Accumulation of malonyl-CoA inhibits carnitine palmitoyltransferase (CPT), the key enzyme required to transport long-chain fatty acids into mitochondria for β-oxidation [23]. This biochemical cascade effectively "locks" the muscle into carbohydrate-dependent metabolism while simultaneously impairing the ability to utilize stored glycogen effectively. The result is an energy substrate paradox where fuel is abundant but unavailable for oxidation.
6.2 Benefits of Long-Chain Fatty Acid Supplementation
Provision of long-chain fatty acids through corn oil or other fat supplements significantly decreases exercise intolerance and rhabdomyolysis in PSSM horses [23]. Fat supplementation increases plasma non-esterified fatty acid (NEFA) concentrations and plasma acylcarnitine levels, indicating enhanced availability of fats for muscle oxidation. Increased β-oxidation of long-chain fats during exercise is suggested by elevated acetyl-carnitine concentrations post-exercise in fat-supplemented horses.
The long-chain fat diets also appear to improve glycogenolytic-glycolytic and oxidative flux in PSSM muscle, as indicated by higher glucose-6-phosphate concentrations, lower lactate concentrations, and higher pyruvate concentrations [23]. This suggests that fat supplementation may "unlock" metabolic pathways that become dysfunctional when excessive glycogen accumulation and high insulin signaling dominate the metabolic landscape.
---
7. Exercise Adaptation and Metabolic Response
7.1 Acute Exercise Response in PSSM
During submaximal exercise, PSSM horses demonstrate an energy deficit that likely underlies muscle fiber damage [24]. The degradation of adenine nucleotides in individual muscle fibers indicates inadequate ATP regeneration despite abundant glycogen stores. Importantly, rhabdomyolysis in PSSM is not characterized by marked accumulation of lactic acid, suggesting that the glycolytic pathway itself functions normally [17].
Exercise testing protocols typically reveal subclinical rhabdomyolysis in PSSM horses, with CK elevations of 1000 U/L or greater occurring 4-6 hours post-exercise in affected but not healthy horses [25]. The dissociation between glycogen availability and energy production supports the concept that PSSM reflects a regulatory defect affecting both glycogen synthesis and metabolic flux rather than a simple storage problem.
7.2 Training Adaptations and Mitochondrial Plasticity
Exercise training induces substantial mitochondrial adaptations that may benefit PSSM horses. Submaximal exercise training increases citrate synthase activity and mitochondrial volume density in equine skeletal muscle [26]. Complex I-linked respiratory capacity increases relative to total electron transport system capacity with improved fitness level, suggesting qualitative improvements in mitochondrial function beyond simple biogenesis [27].
Diet alone will not improve muscle function in horses with PSSM; exercise is crucial for increasing mitochondrial capacity to oxidize fats [28]. Daily exercise enhances glucose utilization and improves energy metabolism in skeletal muscle, providing mechanistic rationale for combined dietary and exercise interventions [13]. Even 10 minutes of daily exercise has been shown to be extremely valuable in reducing muscle damage in PSSM horses [12].
---
8. Pathophysiology of Rhabdomyolysis
8.1 Mechanisms of Muscle Cell Damage
The pathophysiology of rhabdomyolysis in PSSM involves ATP depletion leading to failure of ATP-dependent ion pumps, particularly the Na⁺/K⁺-ATPase and Ca²⁺-ATPase [29]. Dysfunction of these pumps results in intracellular sodium and calcium influx, with water drawn into the cell along with sodium, causing cellular swelling and disruption of intracellular structures. Excessive intracellular calcium activates actin-myosin crosslinkage, myofibrillar contraction, and further ATP depletion in a destructive positive feedback loop.
Elevated intracellular calcium also activates calcium-dependent phospholipases and proteases, promoting cell membrane dissolution and disruption of ion channels [29]. With reperfusion following an ischemic or energy-depleted state, leukocytes migrate into damaged muscle, releasing cytokines, prostaglandins, and free radicals that cause further myolysis and necrosis. The released muscle breakdown products include potassium, myoglobin, creatine kinase, phosphate, uric acid, and various organic acids.
8.2 Clinical Manifestations and Complications
Clinical signs of exertional rhabdomyolysis include excessive sweating, tachypnea, tachycardia, muscle fasciculations, reluctance to move, and firm painful lumbar and gluteal musculature [30]. Episodes range from subclinical enzyme elevations to severe muscle necrosis with recumbency. Serum CK activities may exceed 200,000 U/L in severe cases [25]. Myoglobinuric renal failure represents the most serious potential complication, occurring when the kidney's filtering capacity becomes overwhelmed by excess myoglobin [29].
Unlike other forms of rhabdomyolysis, PSSM horses stall-rested after an initial episode may continue to show elevated or rising serum CK due to ongoing subclinical episodes [25]. This persistent elevation differentiates PSSM from sporadic exertional rhabdomyolysis and supports the recommendation against prolonged rest following episodes [31].
---
9. Dietary Management: Mechanistic Rationale
9.1 Low Non-Structural Carbohydrate Diets
The primary dietary intervention for PSSM involves reducing hydrolyzable carbohydrates to less than 10% of digestible energy [32]. This recommendation targets the mechanistic link between postprandial insulin secretion and glycogen synthase activation. Hydrolyzable carbohydrates (HC) include starch and sugars that stimulate insulin secretion, distinguishing them from non-structural carbohydrates that include compounds like fructans which do not stimulate insulin release [32].
Forage selection should prioritize hay containing less than 12% NSC on a dry matter basis to minimize post-feeding glycemic responses [33]. When hay analysis reveals higher NSC levels, soaking can effectively reduce sugar content. For easy keeper horses requiring caloric restriction, selecting hay with 4% NSC provides room to add adequate fat without exceeding energy requirements [13].
9.2 Grain Elimination and Starch Avoidance
Effective dietary management requires complete elimination of grains including wheat, barley, oats, corn, and molasses [33]. These high-starch ingredients produce rapid and substantial postprandial glucose and insulin elevations that directly stimulate the already overactive glycogen synthase [12]. Sweet feeds are particularly problematic due to their combination of cereal grains and added molasses.
The historical context for these recommendations is important: horses with PSSM fed high-NSC diets demonstrate increased severity of rhabdomyolysis with exercise [15]. Approximately 50% of horses improve with dietary modification alone, but 90% improve when both diet and exercise regimens are implemented [13].
9.3 Fat Supplementation Strategies
For horses requiring additional calories beyond what low-NSC forage provides, fat should replace starch as the primary concentrated energy source [34]. Current recommendations suggest providing 15-25% of digestible energy as fat [33]. Vegetable oil can be added gradually, starting with small amounts and adjusted according to weight and exercise tolerance [35].
Fat supplementation serves multiple mechanistic purposes: it provides an alternative fuel source that bypasses the dysfunctional glycogen synthesis pathway, reduces insulin secretion by decreasing overall carbohydrate intake, and may improve fat oxidation capacity through substrate availability effects [36]. L-carnitine supplementation supports fatty acid transport into mitochondria and may enhance both glucose and fatty acid oxidation pathways.
9.4 Forage Testing and Quality Assessment
Laboratory testing of hay for NSC content is essential because visible characteristics do not reliably predict sugar and starch levels [37]. Mature hay is generally preferred over energy-dense immature hay, though all forage should be free of mold, dust, and foreign material [37]. Pasture grasses can also be analyzed when grazing is part of the management program.
Well-maintained pastures for PSSM horses should contain low-sugar grasses with few legumes [38]. Lush pastures with high sugar content can trigger inappropriate insulin responses and should be avoided. Low-yield acreage with mature grasses may be suitable, though grazing muzzles may be necessary during periods of high pasture sugar content.
---
10. Exercise Management: Mechanistic Rationale
10.1 Daily Exercise Requirements
Regular daily exercise is critical for managing PSSM horses because it enhances glucose utilization and improves energy metabolism in skeletal muscle [13]. Exercise training increases mitochondrial capacity to oxidize fats, providing metabolic flexibility that partially compensates for the glycogen synthesis defect [28]. The recommendation for daily exercise, even as brief as 10 minutes, reflects the rapid loss of conditioning that occurs with inactivity.
Prolonged rest after an episode appears counterproductive, predisposing PSSM horses to further episodes [31]. This is mechanistically consistent with the energy deficit hypothesis: inactive muscle has lower oxidative capacity and relies more heavily on glycogenolysis when exercise resumes. Early return to controlled exercise following symptomatic treatment is advised [35].
10.2 Exercise Prescription Principles
Exercise reintroduction for PSSM horses should follow specific guidelines: providing adequate time for dietary adaptation prior to commencing exercise, restricting duration more than intensity initially, ensuring gradual introduction with consistent performance, and minimizing days without any form of exercise [31]. Duration of exercise is more important to restrict than intensity because total metabolic demand accumulates with time.
For horses with PSSM2, recommendations include prolonged warm-up periods and stretching, with opportunities for rest and stretching during exercise sessions [39]. These modifications may address muscle stiffness and optimize metabolic transitions during exercise onset.
---
Mechanistic Pathway Diagram
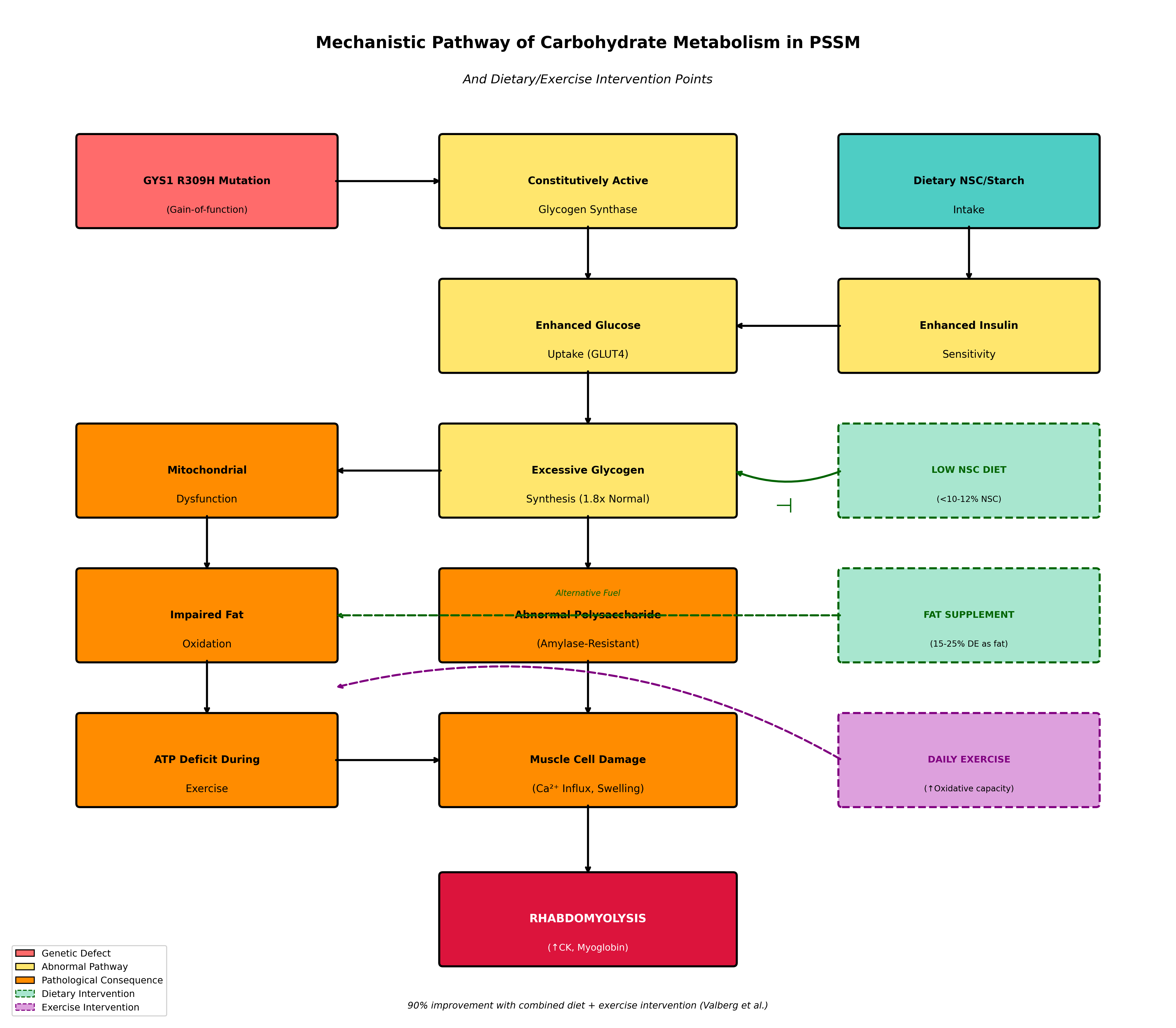
Figure 1. Mechanistic pathway of carbohydrate metabolism dysfunction in PSSM and intervention points. The diagram illustrates how the GYS1 R309H mutation causes constitutive activation of glycogen synthase, leading through enhanced insulin sensitivity and glucose uptake to excessive glycogen synthesis. Pathological consequences include abnormal polysaccharide accumulation, mitochondrial dysfunction, impaired fat oxidation, ATP deficit, and ultimately rhabdomyolysis. Dietary interventions (low NSC diet, fat supplementation) and exercise intervention target specific pathway nodes to interrupt the pathogenic cascade. Data synthesized from McCue et al. (2008), Maile et al. (2017), Valberg et al. (1999), and Williams et al. (2018).
---
Evidence Summary Table
| Mechanistic Component | Key Finding | Dietary/Exercise Implication | Evidence Quality |
|---|---|---|---|
| GYS1 R309H mutation | Constitutive GS activation, gain-of-function | Rationale for reducing substrate (glucose) availability | High (genomic, functional) |
| Glycogen accumulation | 1.8-fold higher than normal | Low NSC reduces synthesis substrate | High (clinical, biochemical) |
| Enhanced insulin sensitivity | Higher glucose infusion rate in clamp studies | Avoid insulin-stimulating feeds | Moderate (limited studies) |
| Postprandial insulin | Elevated with >12% NSC hay | Target <10-12% NSC in hay | Moderate (feeding trials) |
| Abnormal polysaccharide | Amylase-resistant, less-branched | Marker for diagnosis, not directly targeted | High (histopathology) |
| Mitochondrial dysfunction | Decreased OXPHOS with Complex I substrates | Exercise increases mitochondrial capacity | Moderate (respirometry) |
| Impaired fat oxidation | Malonyl-CoA inhibition of CPT with high carbohydrate | Fat supplementation bypasses pathway | Moderate (metabolic studies) |
| Fat supplementation benefit | Decreased exercise intolerance, increased NEFA | 15-25% DE as fat | High (randomized trials) |
| Exercise adaptation | Increased CS activity, mitochondrial density | Daily exercise essential | Moderate (training studies) |
| Rhabdomyolysis prevention | 50% improve with diet; 90% with diet + exercise | Combined approach optimal | High (retrospective) |
| Fiber type changes | More type 2A, fewer type 2X in affected muscle | May reflect adaptive response | Low (limited data) |
| ATP deficit during exercise | Increased IMP, adenine nucleotide degradation | Energy deficit underlies damage | Moderate (exercise testing) |
Unresolved Mechanistic Questions
Questions Regarding Molecular Pathophysiology
- Why does the R309H mutation cause constitutive activation? While structural homology modeling suggests conformational changes, the precise atomic-level mechanism by which this substitution alters enzyme kinetics remains incompletely characterized.
- What is the relationship between glycogen accumulation and mitochondrial dysfunction? Whether mitochondrial impairment is a primary defect contributing to pathology or a secondary consequence of polysaccharide accumulation displacing organelles requires clarification.
- Why doesn't heightened insulin sensitivity explain rhabdomyolysis directly? Dexamethasone treatment can normalize insulin sensitivity without preventing rhabdomyolysis, suggesting separate but potentially linked defects [24].
Questions Regarding PSSM2
- What is the molecular basis of PSSM2? Without elevated glycogen concentrations in many cases, the rationale for identical dietary recommendations is unclear [10].
- Are current dietary recommendations optimal for PSSM2? The absence of excessive glycogen storage suggests alternative pathogenic mechanisms that may require different interventions.
Questions Regarding Therapeutic Mechanisms
- How does fat supplementation improve metabolic flux? The observation that fat-supplemented horses show improved glycolytic pathway function seems paradoxical and warrants mechanistic investigation.
- What is the minimum effective exercise dose? While 10 minutes daily shows benefit, dose-response relationships for exercise duration and intensity remain undefined.
- Does chromium supplementation provide mechanistic benefit? Reports of improved insulin sensitivity and lower exercise lactate with chromium remain preliminary [12].
Questions Regarding Disease Variability
- Why do some homozygous horses remain subclinical? Penetrance is incomplete, suggesting genetic modifiers or environmental factors that buffer against disease expression.
- What explains breed-specific differences in clinical presentation? The distinct phenotypes—tying up in Quarter Horses, weakness in drafts, back pain in Warmbloods—suggest breed-specific modifier effects [40].
- Does concurrent RYR1 mutation explain severe phenotypes? Horses with both GYS1 and RYR1 mutations show more severe disease [41], but the mechanistic interaction requires further study.
- What is the role of oxidative stress? Vitamin E and selenium status may modulate severity, suggesting potential oxidative injury pathways deserving investigation [42].
---
Conclusions
The mechanistic understanding of PSSM has advanced substantially since the identification of the GYS1 R309H mutation, revealing a complex interplay between genetic defects in glycogen synthesis regulation, altered insulin signaling, mitochondrial dysfunction, and impaired alternative fuel utilization. The gain-of-function nature of the mutation explains the persistent drive toward glycogen synthesis that underlies the disease phenotype. Dietary management strategies—including low-NSC forage, grain elimination, and fat supplementation—directly target the identified mechanistic nodes by reducing substrate availability for the overactive enzyme and providing alternative fuel sources that bypass the dysregulated glycogen synthesis pathway.
Exercise represents an equally critical intervention, increasing mitochondrial capacity and oxidative metabolism while promoting glycogen utilization. The dramatic improvement seen with combined dietary and exercise management (90% efficacy versus 50% with diet alone) underscores the importance of addressing multiple mechanistic targets simultaneously [13]. Future research should address the substantial knowledge gaps regarding PSSM2 pathophysiology, optimal intervention parameters, and the molecular basis for clinical variability among affected horses.
References
References
Numbered in order of first citation, IEEE style. Each entry links to the original source via DOI or publisher URL.
- “Glycogen synthase (GYS1) mutation causes a novel skeletal muscle ..,” internet, Unknown Year. Available online
- “Glycogen synthase (GYS1) mutation causes a novel skeletal muscle glycogenosis - Experts@Minnesota,” internet, Unknown Year. Available online
- P. Szauter, PhD, C. S. Officer, and EquiSeq, “Polysaccharide Storage Myopathy type 1 (PSSM1) – EquiSeq,” internet, Jan. 2026. Available online
- “A highly prevalent equine glycogen storage disease is explained by constitutive activation of a mutant glycogen synthase - University of Copenhagen Research Portal,” internet, Unknown Year. Available online
- D. P. Darani, “How to Feed a Horse with Polysaccharide Storage Myopathy (PSSM),” internet, Aug. 2021. Available online
- “Polysaccharide Storage Myopathy (PSSM) – Equiimed,” internet, Unknown Year. Available online
- “Polysaccharide Storage Myopathy, Type 1 | College of Veterinary Medicine,” internet, Unknown Year. Available online
- “Evidence of positive selection for a glycogen synthase (GYS1) mutation in domestic horse populations. - PDF Download Free,” internet, Unknown Year. Available online
- Z. J. Williams, M. Bertels, and S. J. Valberg, “Muscle glycogen concentrations and response to diet and exercise regimes in Warmblood horses with type 2 Polysaccharide Storage Myopathy,” internet, Aug. 2018. Available online
- “Muscle glycogen concentrations and response to diet and exercise regimes in Warmblood horses with type 2 Polysaccharide Storage Myopathy,” internet, Sep. 2018. Available online
- “Insulin sensitivity and skeletal muscle glucose transport in horses with equine polysaccharide storage myopathy - PubMed,” internet, Unknown Year. Available online
- P. H. Nutrition, “POLYSACCHARIDE STORAGE MYOPATHY (PSSM) – Performance Horse Nutrition,” internet, Unknown Year. Available online
- “Polysaccharide Storage Myopathy Type 1,” internet, Jan. 2025. Available online
- “Effect of Diet on Haflinger Horses With GYS1 Mutation (Polysaccharide Storage Myopathy Type 1) | SLU publication database (SLUpub),” internet, Unknown Year. Available online
- 108.179.40.139, “Equine polysaccharide storage myopathy,” internet, Unknown Year. Available online
- “Polysaccharide Storage Myopathy Type 1,” internet, Jan. 2025. Available online
- “Skeletal muscle metabolic response to exercise in horses with ''tying ..,” internet, Unknown Year. Available online
- V. SJ, “Glycogen Synthase 1 ( GYS1 ) Mutation in Diverse Breeds with Polysaccharide Storage Myopathy,” internet, Sep. 2008. Available online
- “https://link.springer.com/content/pdf/10.1186/1746-6148-5-29.pdf?error=cookies_not_supported&code=8d6434be-1111-485e-a6ce-5d819945ee7f,” internet, Sep. 2015. Available online
- C. Wood, T. Ross, J. Armstrong, and D. Hall, “Skeletal Muscle Fiber Type Composition and Citrate Synthase Activity in Fit and Unfit Warmbloods and Quarter Horses,” internet, Nov. 2022. Available online
- “Altered mitochondrial oxidative phosphorylation capacity in horses suffering from polysaccharide storage myopathy. | Research Bank,” internet, Sep. 2023. Available online
- “Altered mitochondrial oxidative phosphorylation capacity in horses suffering from polysaccharide storage myopathy,” internet, Aug. 2018. Available online
- “Effect of dietary fats with odd or even numbers of carbon atoms on ..,” internet, Mar. 2010. Available online
- “Polysaccharide Storage Myopathy | IVIS,” internet, Dec. 2006. Available online
- “Polysaccharide Storage Myopathy: One Important Cause of Exertional Rha – KER Targeted Nutrition,” internet, Aug. 2015. Available online
- “Submaximal exercise training improves mitochondrial efficiency in the gluteus medius but not in the triceps brachii of young equine athletes | Scientific Reports,” internet, Oct. 2017. Available online
- “Physical Fitness and Mitochondrial Respiratory Capacity in Horse Skeletal Muscle | PLOS One,” internet, Unknown Year. Available online
- F. Watkins, “A nutritional review of polysaccharide storage myopathy,” internet, Jul. 2023. Available online
- M. V, N. A. V, G. S. E, R. Z, S. A, and T. I., “Rhabdomyolysis - StatPearls - NCBI Bookshelf - NIH,” internet, Jul. 2025. Available online
- “Exertional Myopathies in Horses - Musculoskeletal System - MSD Veterinary Manual,” internet, May 2022. Available online
- E. E. Ony, “Diet Adjustments Provide Relief for PSSM Horses,” internet, Feb. 2016. Available online
- H. Scofield, “Type 1 PSSM in Horses: Genetic Cause, Symptoms, & Management,” internet, Aug. 2023. Available online
- F. Watkins, “UK Vet Equine - A nutritional review of polysaccharide storage myopathy,” internet, Jul. 2023. Available online
- N. Dewsbury, “Polysaccharide Storage Myopathy in Horses: Causes and Management - All About Horses,” internet, Mar. 2025. Available online
- “PSSM IN HORSES: COMMON BUT UNDER-DIAGNOSED CONDITION?,” internet, Unknown Year. Available online
- D. P. Darani, “How to Feed a Horse with Polysaccharide Storage Myopathy (PSSM),” internet, Aug. 2021. Available online
- F. S. -. 0. Health, “Diet Adjustments Provide Relief for Horses with Polysaccharide Storage Myopathy,” internet, Dec. 2016. Available online
- E. E. Ony, “Diet Adjustments Provide Relief for PSSM Horses,” internet, Feb. 2016. Available online
- “Polysaccharide Storage Myopathy (PSSM) in Horses | PetMD,” internet, Oct. 2022. Available online
- H. Scofield, “Type 1 PSSM in Horses: Genetic Cause, Symptoms, & Management,” internet, Aug. 2023. Available online
- 108.179.40.139, “Equine polysaccharide storage myopathy,” internet, Unknown Year. Available online
- “https://vanners.org/wp-content/uploads/2023/11/PSSM1-article.pdf,” internet, Feb. 2005. Available online
This review is one of 34 in the Foxwatch research corpus. Methodology: PRISMA 2020-style systematic literature review against PubMed, CAB Abstracts, Scopus, Web of Science, and equine veterinary conference proceedings.















