Meal Timing, Feeding Frequency, Turnout
When you feed matters almost as much as what you feed. Pasture grass sugar content rises through the day and falls overnight; a grain meal spikes glucose differently if the horse worked first or rested first; insulin response shifts with feeding interval. This review covers the diurnal sugar curve, post-prandial dynamics, and pre-exercise meal composition in horses already biased toward storing too much glycogen. Powers the time-of-day pasture chart on the feeding-PSSM page. Read with review 02 (threshold) and 08 (pasture management).
Research thesis
This systematic review argues that meal timing, feeding frequency, and pasture-access scheduling are an undervalued lever in PSSM management, comparable in clinical effect to NSC restriction itself, not only because diurnal NSC variation in pasture grass and post-prandial glucose curves drive measurable swings in muscle glycogen flux, but also because feeding interval and pre-exercise meal composition shift insulin response in horses already biased toward excess glycogen storage; by examining grazing-pattern, glycemic-response, and feeding-frequency trials across PSSM1 and suspected PSSM2 populations, this review establishes meal-timing and turnout protocols supported by clinical-outcome data, not just by glucose-curve studies.
Evidence visualizations & full pages
Evidence figures (4)
Figures referenced by the review's Figure N: captions. Click any chart for a full-size view. They are also rendered inline below their respective captions in the full review text further down.
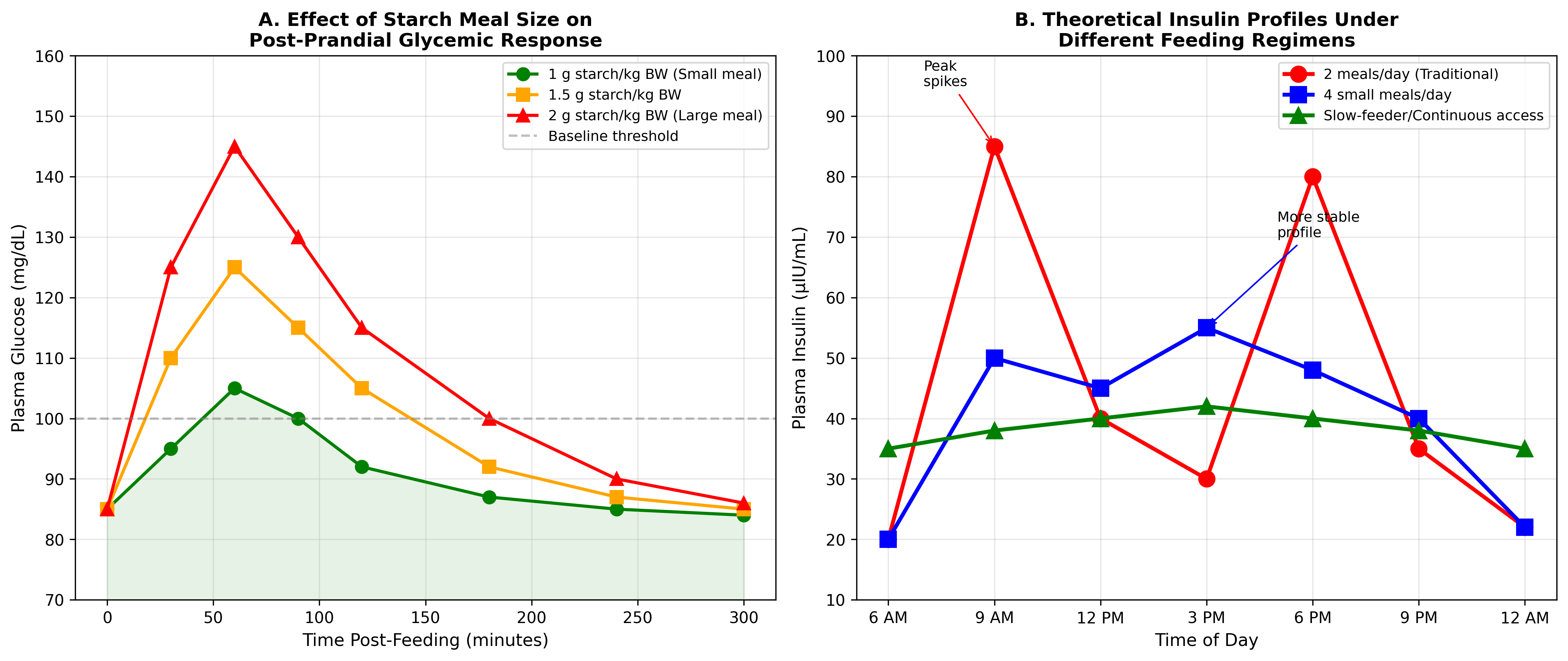 Figure 1. Effect of starch meal size on post-prandial glycemic response (Panel A) and theoretical insulin profiles under different feeding regimens (Panel B). Data adapted from Varasteh et al. (2023). Smaller,
Figure 1. Effect of starch meal size on post-prandial glycemic response (Panel A) and theoretical insulin profiles under different feeding regimens (Panel B). Data adapted from Varasteh et al. (2023). Smaller,
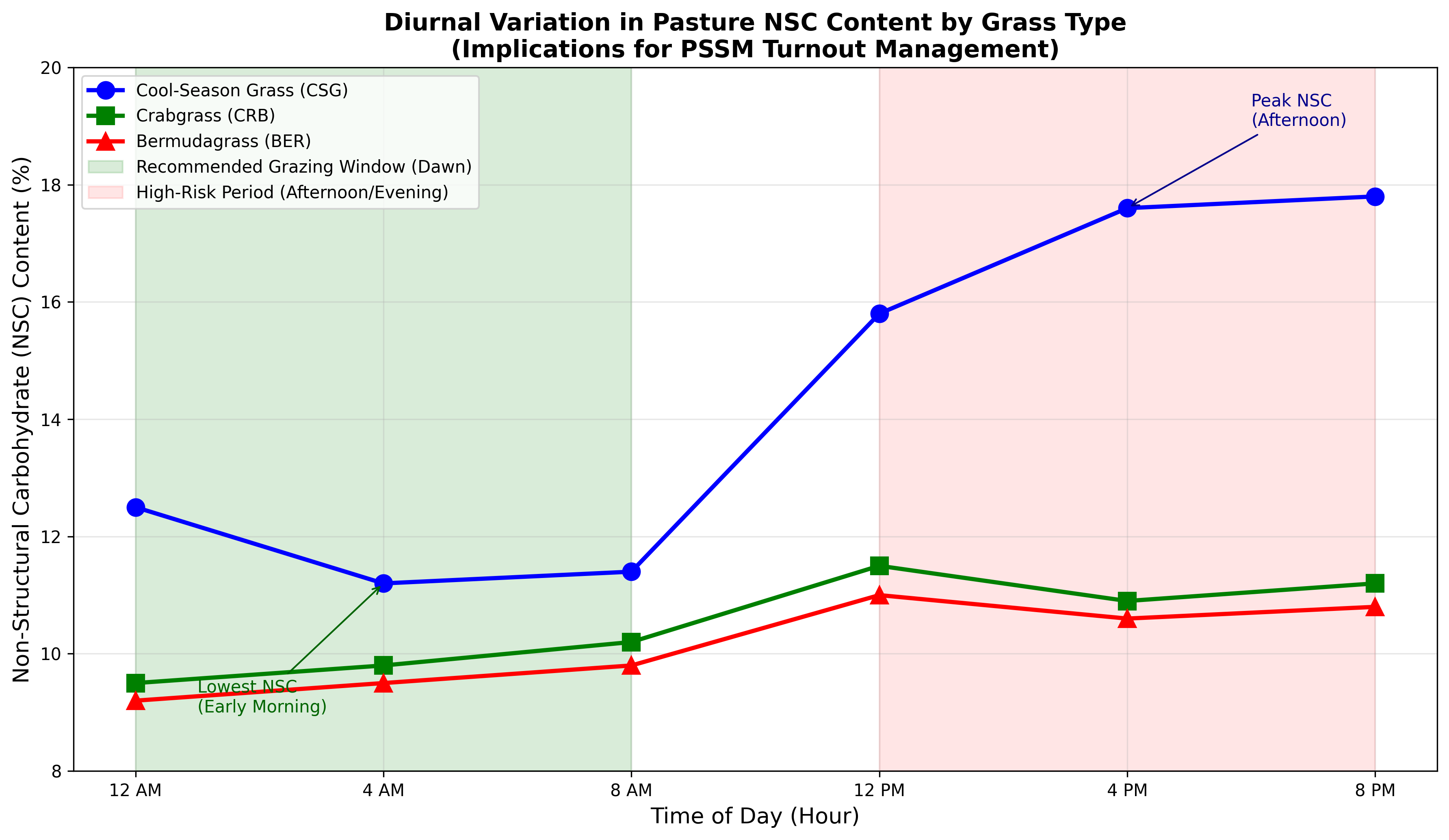 Figure 1. Effect of starch meal size on post-prandial glycemic response (Panel A) and theoretical insulin profiles under different feeding regimens (Panel B). Data adapted from Varasteh et al. (2023). Smaller,
Figure 1. Effect of starch meal size on post-prandial glycemic response (Panel A) and theoretical insulin profiles under different feeding regimens (Panel B). Data adapted from Varasteh et al. (2023). Smaller,
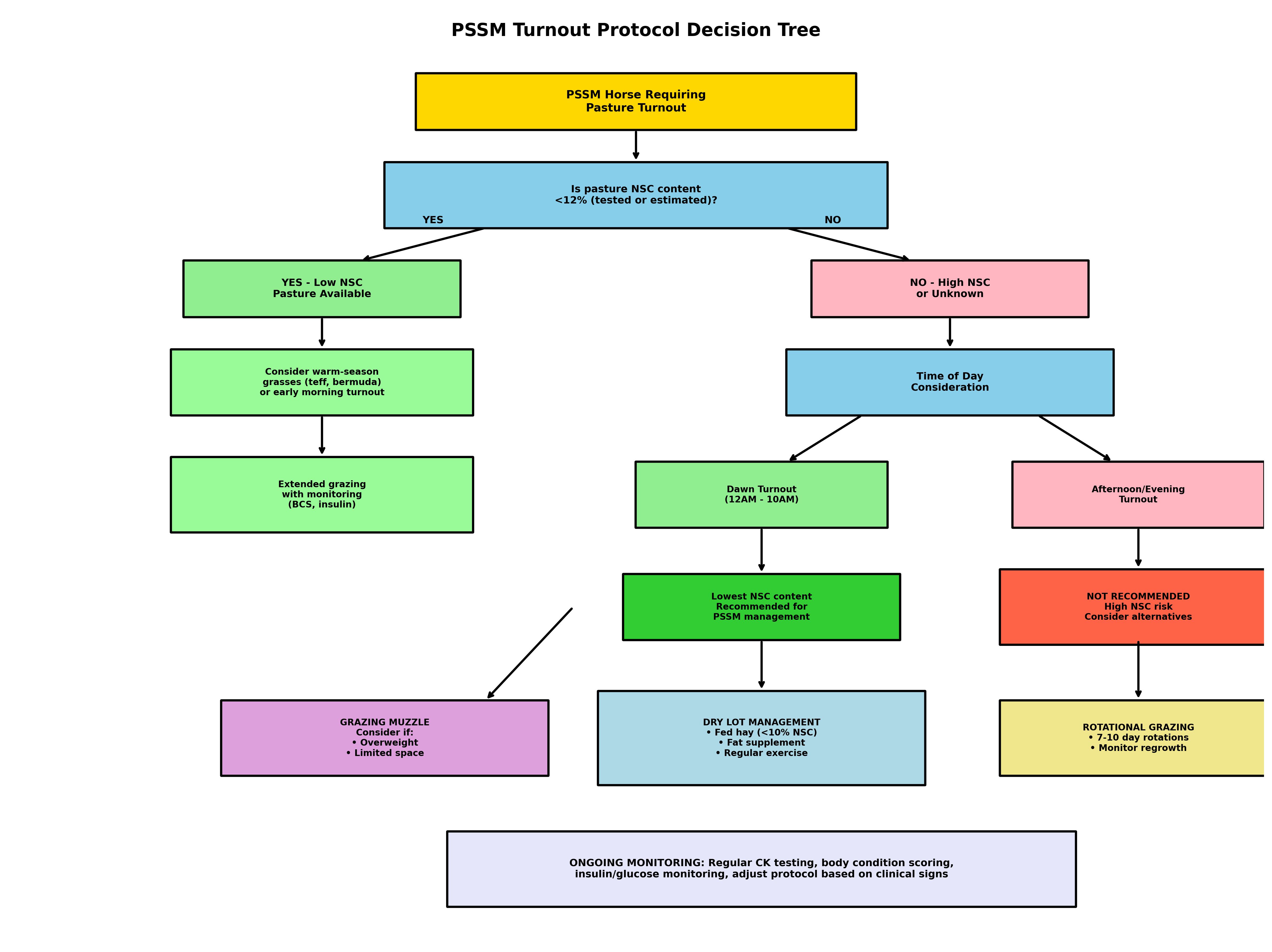 Figure 1. Effect of starch meal size on post-prandial glycemic response (Panel A) and theoretical insulin profiles under different feeding regimens (Panel B). Data adapted from Varasteh et al. (2023). Smaller,
Figure 1. Effect of starch meal size on post-prandial glycemic response (Panel A) and theoretical insulin profiles under different feeding regimens (Panel B). Data adapted from Varasteh et al. (2023). Smaller,
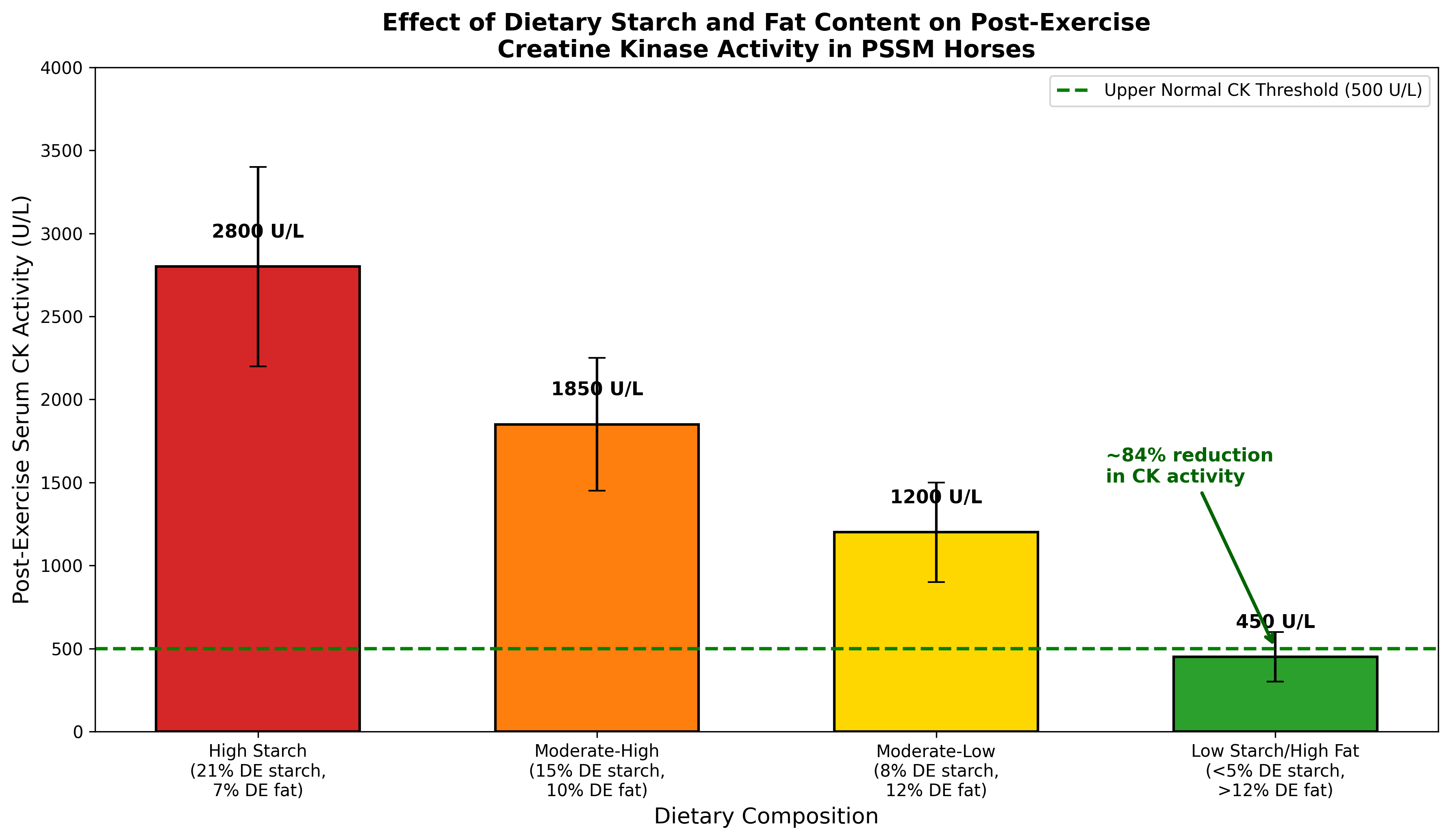 Figure 1. Effect of starch meal size on post-prandial glycemic response (Panel A) and theoretical insulin profiles under different feeding regimens (Panel B). Data adapted from Varasteh et al. (2023). Smaller,
Figure 1. Effect of starch meal size on post-prandial glycemic response (Panel A) and theoretical insulin profiles under different feeding regimens (Panel B). Data adapted from Varasteh et al. (2023). Smaller,
Original review pages (PDF render)
Download
Full review text
Meal Timing, Feeding Frequency, Turnout
Meal Timing, Feeding Frequency, and Pasture Turnout Management for Polysaccharide Storage Myopathy: A Systematic Review
1. Introduction
1.1 Background and Clinical Significance
Polysaccharide storage myopathy (PSSM) represents a significant clinical challenge in equine medicine, characterized by abnormal glycogen and amylase-resistant polysaccharide accumulation within skeletal muscle fibers [1]. Type 1 PSSM (PSSM1) is an autosomal dominant glycogen storage disorder caused by a gain-of-function mutation (R309H) in the glycogen synthase 1 gene (GYS1), affecting more than 20 breeds of horses and presenting with various clinical signs including exertional rhabdomyolysis [2]. Both PSSM1 and PSSM2 are characterized by elevated resting and post-exercise creatine kinase (CK) activities, with homozygous PSSM1 horses demonstrating median resting CK activities of 364 U/L compared to 260 U/L in controls, reflecting ongoing subclinical muscle damage [1].
The pathophysiology of PSSM involves impaired glycogen metabolism that results in excessive glycogen accumulation and exercise-induced muscle injury. Clinical manifestations typically include stiffness, reluctance to move, and episodes of rhabdomyolysis that can be triggered by inappropriate feeding practices and inadequate exercise management. Both PSSM1 and PSSM2-ER respond favorably to a low nonstructural carbohydrate (NSC), high-fat diet combined with regular exercise [2]. Understanding the nuanced interactions between meal timing, feeding frequency, and pasture turnout management is critical for optimizing outcomes in affected horses.
1.2 Scope of This Review
This systematic review examines the current evidence regarding nutritional timing strategies for PSSM management, including pre-exercise meal timing considerations for carbohydrate versus fat-based feeds, the comparison of small frequent feeding regimens versus traditional two-meal approaches, effects of overnight fasting on glycogen dynamics, diurnal cycling of NSC in pastures, grazing management tools including muzzles and dry lots, and rotational grazing systems. The review synthesizes evidence from equine-specific studies where available and extrapolates from relevant research in comparative species physiology and sports nutrition science.
---
2. Pre-Exercise Meal Timing: Carbohydrate Versus Fat-Based Approaches
2.1 Metabolic Considerations for Pre-Exercise Feeding
The timing and composition of pre-exercise meals significantly influence substrate availability and metabolic responses during subsequent exercise [3]. Carbohydrate feedings prior to endurance exercise commonly enhance performance by optimizing glycogen stores, although these meals simultaneously increase insulin levels and reduce fat oxidation. For PSSM horses, this metabolic profile presents a paradox: while carbohydrate availability supports immediate energy demands, the hyperinsulinemic response may exacerbate glycogen synthesis and storage in already glycogen-overloaded muscle fibers.
Research on pre-exercise carbohydrate quantity and quality demonstrates that high carbohydrate meals (1.5 g/kg body weight) preserve muscle function more effectively than low carbohydrate meals (0.8 g/kg body weight), with greater carbohydrate oxidation and insulin response attenuating central fatigue mechanisms [4]. High glycemic index carbohydrates demonstrate superior preservation of force production and carbohydrate oxidation compared to low glycemic index alternatives. However, these findings from healthy athletic subjects must be interpreted cautiously for PSSM horses, where minimizing insulin-driven glycogen synthesis is a primary therapeutic objective.
2.2 Fat-Based Pre-Exercise Feeding Strategies
High-fat meals demonstrate beneficial metabolic effects including increased fat oxidation and potential glycogen sparing, though these effects do not universally translate into enhanced performance [3]. For PSSM horses, the shift toward fat oxidation represents a desirable metabolic adaptation that reduces reliance on glycogen as a primary fuel substrate. The strategic use of fat-supplemented meals before exercise aims to increase availability of free fatty acids (FFA) for muscle metabolism, potentially reducing the glycogen mobilization that triggers rhabdomyolysis.
Research specifically evaluating PSSM horses found that dietary interventions with less than 5% digestible energy (DE) from starch and greater than 12% DE from fat significantly reduced post-exercise CK activity compared to high-starch diets [5]. The postprandial insulin response was substantially higher for high-starch diets versus low-starch/high-fat alternatives, while FFA availability increased markedly on fat-supplemented diets. These findings support fat-based feeding strategies before exercise, though the optimal timing interval between feeding and exercise remains to be precisely defined for PSSM horses. Adaptation to dietary changes required approximately 3 to 6 months, with 16 of 19 horses demonstrating no episodes of exertional rhabdomyolysis after sustained low-carbohydrate, high-fat diet implementation [6].
2.3 Timing Recommendations for Pre-Exercise Meals
Current sports nutrition guidelines recommend consuming carbohydrates throughout resistance exercise to promote euglycemia and maintain glycogen stores [7]. For PSSM horses, however, the goal is to minimize acute glycemic and insulinemic spikes while ensuring adequate energy substrate availability. The size and timing of pre-exercise meals significantly impact the extent to which post-exercise feeding interventions are required, suggesting that strategic meal timing can modulate overall metabolic responses.
---
3. Feeding Frequency: Small Frequent Meals Versus Traditional Two-Meal Regimens
3.1 Glycemic Response to Meal Size
The relationship between meal size and glycemic response has been systematically evaluated in horses, demonstrating that increasing starch levels per meal from 1 to 2 g/kg body weight results in significantly higher postprandial plasma glucose concentrations [8]. Micronized flaked corn demonstrated three times greater in vitro enzymatic dry matter disappearance compared to ground corn, and horses consuming processed grain exhibited higher post-prandial glucose concentrations. These findings underscore the importance of both meal size and feed processing in determining glycemic impact.
Processing methods significantly influence metabolic responses, with micronizing demonstrating superior pre-caecal starch digestibility compared to toasting [9]. The amount of starch escaping enzymatic digestion in the small intestine exceeded expectations, suggesting that significant amounts of fermentable substrate may reach the hindgut even with processed feeds. For PSSM horses, this highlights the importance of controlling total starch intake per meal rather than relying solely on processing methods to moderate glycemic responses.
3.2 Benefits of Frequent Small Meals
Dividing the daily ration into multiple smaller meals reduces peak glycemic and insulinemic excursions by distributing substrate delivery over extended periods. Research on nutrient timing in athletic contexts indicates that meeting total daily intake of protein through evenly spaced feedings (approximately every 3 hours) should be viewed as a primary emphasis for exercising individuals [7]. Although this research focused on protein, the principle of distributing nutrients to avoid large bolus effects applies equally to carbohydrate management in PSSM horses.
Horses maintained on low-starch diets (feeding less than 1 g NSC/kg body weight per meal) demonstrated protective effects against gastric ulceration, an important secondary consideration when designing feeding protocols [10]. Being fed complementary feed at these controlled NSC levels was protective for gastroscopically significant squamous ulceration, indicating that frequent small meals support both metabolic and gastrointestinal health.

Figure 1. Effect of starch meal size on post-prandial glycemic response (Panel A) and theoretical insulin profiles under different feeding regimens (Panel B). Data adapted from Varasteh et al. (2023). Smaller, more frequent meals produce lower peak glucose concentrations and more stable insulin profiles compared to traditional two-meal regimens.
3.3 Practical Implementation Considerations
Implementing frequent feeding requires balancing practical management constraints with metabolic optimization. Research on voluntary forage intake demonstrates that animal traits and forage quality significantly impact consumption patterns, with pastured animals consuming less than stalled animals potentially due to the time required for grazing behavior [11]. This finding suggests that slow-feeders and hay nets may serve dual purposes: extending eating duration to simulate grazing patterns while controlling total intake.
---
4. Overnight Fasting and Glycogen Dynamics
4.1 Metabolic Consequences of Extended Fasting
Extended overnight fasting significantly alters metabolic substrate availability and glycogenic responses to subsequent meals. Research demonstrates that extending the overnight fast (i.e., skipping breakfast) increases glycemic responses to the following meal, a phenomenon known as the second-meal effect [12]. When breakfast skipping is extended over 6 weeks, there is evidence of more stable interstitial glucose concentrations and altered gene expression in adipose tissue, though these adaptations may not be desirable for PSSM horses requiring consistent substrate availability.
Metabolic responses to food ingestion demonstrate time-of-day dependency, with glucose and triacylglycerol concentrations showing larger postprandial increases in the evening versus morning [12]. These diurnal rhythms are mediated by variations in β-cell function, insulin secretion, clearance, and sensitivity. For PSSM horses, these findings suggest that identical meals fed at different times may produce different metabolic responses, with potential implications for optimal feeding timing.
4.2 Glycogen Synthesis Regulation During Fasting and Refeeding
Skeletal muscle glycogen synthesis involves key signaling proteins including AMPK, Akt2, and their downstream targets TBC1D1/4, which facilitate glucose uptake via GLUT4 translocation [13]. Following exercise-induced glycogen depletion, both type I and type II muscle fibers demonstrate similar increases in insulin-mediated glucose uptake during recovery. The ultimate fate of glucose in the post-exercise state is predominantly glycogen resynthesis, making feeding timing relative to exercise particularly relevant for PSSM management.
Research examining mice with skeletal muscle-specific deficiency of long-chain acyl-CoA synthetase demonstrated that impaired fatty acid oxidation capacity increased glucose utilization and elevated respiratory exchange ratio during overnight fasting [14]. These animals showed severely compromised systemic glucose homeostasis during exercise, reaching hypoglycemia at exhaustion while controls maintained normal glucose levels. Although this represents an extreme phenotype, it illustrates how substrate availability constraints during fasting can profoundly affect exercise capacity.
4.3 Recommendations for Overnight Management
For PSSM horses, overnight access to low-NSC forage is generally recommended to prevent extended fasting periods that might trigger compensatory hyperinsulinemia upon refeeding. Delayed feeding of high-sucrose diets in rats increased body weight gain by affecting body temperature, insulin resistance, and circadian oscillation of lipid metabolism-related genes [15]. This suggests that irregular feeding timing may disrupt metabolic programming in ways that promote insulin resistance and altered energy partitioning.
---
5. Diurnal Cycling of Pasture Non-Structural Carbohydrates
5.1 NSC Variation Patterns in Different Grass Types
Pasture NSC content demonstrates significant diurnal variation that directly impacts PSSM management decisions. Research evaluating cool-season grasses (CSG), crabgrass (CRB), and bermudagrass (BER) demonstrated that NSC was greatest in the afternoon and evening (14.5-14.9%) and lowest in the early morning (11.2-11.4%) [16]. Critically, diurnal variation was most pronounced in cool-season grasses versus either warm-season alternative. Cool-season grasses demonstrated significantly higher NSC (17.6%) compared to bermudagrass (10.6%) or crabgrass (10.9%), supporting recommendations to consider warm-season grasses for metabolically compromised horses.

Figure 2. Diurnal variation in pasture NSC content by grass type. Data adapted from Weinert-Nelson et al. (2021). Cool-season grasses demonstrate pronounced diurnal variation with peak NSC in afternoon/evening hours, while warm-season grasses maintain lower, more stable NSC levels throughout the day. Green shading indicates recommended grazing window (dawn); red shading indicates high-risk periods.
5.2 Environmental Factors Affecting NSC Accumulation
NSC accumulation in forages results from photosynthetic activity during daylight hours, with sugars produced during the day being partially utilized for overnight respiration. Conditions that enhance photosynthesis (sunny weather) or reduce overnight respiration (cool nights) result in elevated morning NSC levels that continue to accumulate throughout the day. These results support recommendations for restricting grazing to early morning hours to limit NSC consumption, particularly in cool-season grass pastures [16].
Integrating warm-season grasses into cool-season equine rotational grazing systems can increase pasture availability during hot, dry summer months while providing metabolic benefits [17]. Distinct shifts in the equine fecal microbiota occur in response to different forages, with changes most influenced by forage NSC and crude protein rather than fiber content. Akkermansia and Clostridium butyricum were enriched in horses grazing warm-season pastures and negatively correlated with NSC and peak plasma glucose concentrations, suggesting potential microbiome-mediated metabolic benefits.
5.3 Seasonal Considerations
The insulin response to oral sugar testing does not demonstrate significant seasonal variation in healthy horses, though individual horses showed variable insulin dysregulation status throughout the year [18]. However, cresty neck score and lack of exercise were identified as risk factors for insulin dysregulation. Horses with insulin dysregulation had lower serum adiponectin concentrations, suggesting that metabolic status interacts with pasture management requirements.
Long-term high-energy diet feeding to Shetland pony mares led to significant hyperinsulinemia and obesity within 24 weeks, with mean body weight increasing by 27% [19]. Hyperinsulinemic status was reversed during 17 weeks of hay-only feeding regardless of body condition, but returned rapidly after restarting high-energy feeding. This demonstrates the metabolic plasticity of the insulin response and the importance of consistent dietary management.
---
6. Grazing Muzzle Effectiveness
6.1 Purpose and Mechanisms of Action
Grazing muzzles represent a practical tool for limiting pasture intake while maintaining social and behavioral benefits of turnout. Research on obesity management in horses indicates that weight-management typically focuses on restricting consumption through limiting pasture grazing, often facilitated through stabling, which can lead to social isolation and secondary welfare issues [20]. Exercise-related variables correlated with decreased reports of laminitis and Equine Metabolic Syndrome, supporting combined approaches that maintain mobility while controlling intake.
6.2 Evidence for Effectiveness
Studies evaluating space-restricted rotational grazing demonstrated significant effects on time budgets and grazing behavior [21]. Horses in space-restricted groups displayed different grazing patterns compared to continuous grazing controls, with proportion of grazing having an inverse relationship with standing time. Grazing was displayed more in restricted horses during initial weeks before patterns reversed, suggesting behavioral adaptation to limited forage availability.
Comparison of rotational versus continuous grazing systems over 27 months revealed that continuously grazed horses maintained higher body condition scores (6.3 ± 0.05) and body fat percentages (17.9 ± 0.15%) compared to rotationally grazed horses (BCS 5.9 ± 0.05; body fat 16.8 ± 0.15%) [22]. Rotationally grazed horses had pasture access for approximately half the total study duration while being fed moderate quality hay at 2% body weight when not grazing. These findings support structured turnout approaches that combine limited grazing with controlled hay supplementation.
---
7. Dry Lot Management Strategies
7.1 Indications for Dry Lot Housing
Management of equine metabolic syndrome, which shares pathophysiological features with PSSM through insulin dysregulation, centers on strict dietary NSC restriction, controlled forage intake, and elimination or careful management of pasture access [23]. Environmental and management factors, particularly high-NSC diets, unrestricted pasture access, and physical inactivity are primary drivers of metabolic dysfunction. Dry lot housing provides controlled conditions for implementing strict NSC restriction when pasture quality cannot be adequately managed.
Horses transitioning from pasture into training represent a high-risk period, with those coming off pasture into training for 4 weeks or less showing significantly higher risk for gastric ulceration compared to those adapted for 5 weeks or more [10]. However, being stabled while spending more than 2 hours per day in a paddock was protective for gastric health compared with either minimal paddock time or full-time turnout. This suggests that partial turnout with controlled grazing may optimize both metabolic and gastrointestinal health.
7.2 Feeding Protocols for Dry Lot Management
Research on PSSM2 Warmblood horses demonstrated that with recommended diet and exercise regimes, 80% of owners reported overall improvement with significant decreases in horses showing decline in performance and rhabdomyolysis [24]. However, 53% of PSSM2 horses remained unable to advance as expected, with reluctance to go forward and collect persisting in approximately one-third of horses. Horses with higher muscle glycogen concentrations were significantly more likely to show performance decline, indicating ongoing challenges in glycogen management even with appropriate feeding protocols.
---
8. Rotational Grazing Systems
8.1 Principles and Implementation
Rotational grazing involves systematically moving horses through pasture sections to allow vegetation recovery while controlling intake. A decade of research on horse pasture utilization demonstrated that horses allowed to rotationally graze using best management practices throughout growing seasons showed reduced glucose and insulin responses when grazing low-NSC forages such as teff compared to perennial cool-season grasses [25]. Strategic use of pasture forages can help manage common health concerns including obesity and Equine Metabolic Syndrome.
Comparison of 27 months of rotational versus continuous grazing demonstrated that rotational pastures maintained greater sward height (11.8 ± 0.1 cm versus 6.9 ± 0.1 cm) and herbage mass (1,513 ± 41 kg/ha versus 781 ± 35 kg/ha) while supporting lower horse body condition [22]. Rotational pastures had higher proportions of vegetative cover and planted grasses but lower proportions of undesirable grass weeds and bare ground. Average grazing bout length increased numerically each year from 7.88 days in 2014 to 10.9 days in 2016, indicating pasture improvement allowing extended grazing periods.
8.2 Effects on Metabolic Parameters and Pasture Quality
Research evaluating rotational grazing effects on soil and vegetation demonstrates that proper management increases pasture productivity and strengthens forage base sustainability [26]. The number of plant species and average vegetation height were significantly greater under rotational grazing compared to intensive unsystematic use. These improvements in pasture quality translate to enhanced nutritional control and more predictable NSC content for metabolically compromised horses.

Figure 3. Turnout protocol decision tree for PSSM horses. This algorithm guides pasture management decisions based on forage NSC content, time of day, and available management tools. Based on evidence from Weinert-Nelson et al. (2021), Williams et al. (2020), and Bertin (2026).
---
9. Creatine Kinase Biomarker Trajectories and Diet Response
9.1 CK as a Marker of Muscle Damage
Serum creatine kinase activity represents a primary biomarker for monitoring muscle damage and treatment response in PSSM horses. Resting plasma AST activity was significantly associated with severity of subsarcolemmal vacuolation and cytoplasmic inclusions in PSSM1 horses, indicating that plasma enzyme activity reflects underlying pathological severity [1]. The association between muscle enzyme levels and pathology severity suggests that physical disruption of myofibers may contribute directly to the myopathic phenotype.
McArdle disease, a related glycogenosis in humans, demonstrates persistently elevated baseline CK without objective weakness and dramatically fluctuating CK levels as important diagnostic clues [27]. Fifteen patients demonstrated median CK values ranging from 273 to 75,510 IU/L with significant fluctuations, emphasizing the importance of serial monitoring rather than single measurements.
9.2 Dietary Effects on CK Activity
The relationship between dietary composition and post-exercise CK activity has been systematically evaluated in PSSM horses. Four isocaloric diets ranging from 21.2% to 3.9% DE from starch demonstrated a clear dose-response relationship, with log CK activity significantly higher on high-starch diets compared to the lowest starch option [5]. Individual variation occurred in response to different diets, but horses with clinical manifestations consistently benefited from diets with less than 5% DE from starch and greater than 12% DE from fat.

Figure 4. Effect of dietary starch and fat content on post-exercise creatine kinase activity in PSSM horses. Data adapted from Ribeiro et al. (2004). Progressive reduction in starch content with corresponding increase in fat supplementation reduces post-exercise CK activity by approximately 84% compared to high-starch diets.
Sixteen of 19 horses with exertional rhabdomyolysis demonstrated no recurrence after 3 to 6 months of low-carbohydrate, high-fat diet implementation, with post-exercise CK and AST activities significantly reduced compared to initial values [6]. All horses showed evidence of chronic myopathic change and abnormal glycogen accumulation on biopsy, with 11 also demonstrating complex polysaccharide accumulation. Adaptation required approximately 3 to 6 months, emphasizing the need for sustained dietary modification rather than short-term interventions.
---
10. Weight Management and Body Condition Considerations
10.1 Obesity as a Complicating Factor
Obesity exacerbates metabolic dysfunction through suspected adipose-derived inflammation and is frequently observed in horses with insulin dysregulation [23]. Although obesity is a frequent finding in metabolically compromised horses, it can occur in lean animals and not all obese horses are affected, necessitating objective diagnostic testing rather than reliance on body condition alone. Regular exercise improves insulin sensitivity but must be adapted to current disease status.
Space-restricted rotational grazing significantly affected body weight and time budgets in horses, with controlled intake allowing weight management without completely eliminating pasture access [21]. Continuously grazed horses maintained higher body condition scores than rotationally managed horses despite similar hay supplementation costs, supporting structured turnout approaches for weight management.
10.2 Exercise Integration
Exercise represents a critical component of PSSM management that synergizes with dietary interventions. Exercise training combined with high-protein diet in late-onset Pompe disease (a human glycogenosis) resulted in improved peak aerobic power and significant reductions in both CK and LDH levels compared to exercise alone [28]. The combination of lifestyle interventions also improved pulmonary function and quality of life measures. Although this research focused on a different species and glycogenosis, the principles of combined exercise and nutritional optimization appear broadly applicable.
Research in Quarter Horses with PSSM2 demonstrated that exertional rhabdomyolysis improved with regular exercise and a low starch/fat-supplemented diet [29]. Horses were predominantly involved in barrel racing and working cow/roping performance activities, indicating that appropriate management enables athletic performance even in affected individuals.
---
11. Summary Tables
Table 1. Dietary Composition Recommendations for PSSM Horses
| Parameter | Recommended Level | Evidence Source |
|---|---|---|
| Starch (% DE) | <5% | Ribeiro et al. (2004) |
| Fat (% DE) | >12% | Ribeiro et al. (2004) |
| NSC per meal | <1 g/kg BW | Luthersson et al. (2023) |
| Adaptation period | 3-6 months | Valentine et al. (1998) |
| Exercise requirement | Regular, moderate | Firshman & Valberg (2025) |
Table 2. Pasture NSC Content by Grass Type and Time of Day
| Grass Type | Early Morning NSC (%) | Afternoon NSC (%) | Peak Variation |
|---|---|---|---|
| Cool-Season Grass | 11.2-11.4 | 14.5-14.9 | High |
| Crabgrass | 9.5-10.2 | 10.9-11.5 | Moderate |
| Bermudagrass | 9.2-9.8 | 10.6-11.0 | Low |
Table 3. Grazing Management System Comparison
| System | Body Condition Impact | Pasture Quality | NSC Control |
|---|---|---|---|
| Continuous Grazing | Higher BCS (6.3) | Lower sward height | Poor |
| Rotational Grazing | Lower BCS (5.9) | Higher sward height | Moderate |
| Dry Lot + Hay | Controllable | Not applicable | Excellent |
| Grazing Muzzle | Variable | Variable | Good |
---
12. Conclusions and Practical Recommendations
12.1 Key Findings
This systematic review identifies several evidence-based principles for meal timing, feeding frequency, and turnout management in PSSM horses:
- Pre-exercise feeding should emphasize fat-based energy sources to promote fatty acid oxidation while minimizing glycemic and insulinemic responses that exacerbate glycogen synthesis [5].
- Feeding frequency should favor multiple small meals delivering less than 1 g NSC/kg body weight per meal to maintain stable glycemic profiles and reduce peak insulin excursions [8].
- Overnight management should provide continuous access to low-NSC forage to prevent extended fasting that triggers compensatory metabolic responses upon refeeding [12].
- Pasture turnout timing should prioritize early morning hours (prior to 10 AM) when NSC content is lowest, particularly for cool-season grass pastures [16].
- Warm-season grasses such as teff and bermudagrass offer lower and more stable NSC profiles suitable for metabolically compromised horses [25].
- Rotational grazing systems support better weight management and pasture quality compared to continuous grazing while maintaining welfare benefits of turnout [22].
12.2 Clinical Implementation
Successful PSSM management requires integration of dietary, exercise, and environmental management strategies tailored to individual horse needs. Serial monitoring of CK activity provides objective assessment of treatment response, with adaptation to dietary changes requiring 3 to 6 months before maximum benefit [6]. Close collaboration between veterinarians, nutritionists, and owners is essential for sustained implementation of management recommendations, with favorable prognosis when interventions are recognized early and maintained rigorously [23].
References
References
Numbered in order of first citation, IEEE style. Each entry links to the original source via DOI or publisher URL.
- R. Naylor, L. Livesey, J. Schumacher, N. Henke, C. Massey, K. Brock, M. Fernández-Fuente, and R. Piercy, “Allele Copy Number and Underlying Pathology Are Associated with Subclinical Severity in Equine Type 1 Polysaccharide Storage Myopathy (PSSM1),” PLoS ONE, Jul. 2012. doi: 10.1371/journal.pone.0042317. View source
- A. Firshman and S. Valberg, “Polysaccharide Storage Myopathy.,” The Veterinary clinics of North America. Equine practice, Jan. 2025. doi: 10.1016/j.cveq.2024.11.004. View source
- M. Ormsbee, C. W. Bach, and D. A. Baur, “Pre-Exercise Nutrition: The Role of Macronutrients, Modified Starches and Supplements on Metabolism and Endurance Performance,” Nutrients, Apr. 2014. doi: 10.3390/nu6051782. View source
- T. Khong, V. Selvanayagam, S. Hamzah, and A. Yusof, “Effect of quantity and quality of pre-exercise carbohydrate meals on central fatigue.,” Journal of applied physiology, Oct. 2018. doi: 10.1152/japplphysiol.00221.2018. View source
- W. P. Ribeiro, S. Valberg, J. D. Pagan, and B. Gustavsson, “The effect of varying dietary starch and fat content on serum creatine kinase activity and substrate availability in equine polysaccharide storage myopathy.,” Journal of Veterinary Internal Medicine, Nov. 2004. doi: 10.1111/j.1939-1676.2004.tb02637.x. View source
- B. A. Valentine, H. F. Hintz, K. M. Freels, A. J. Reynolds, and K. N. Thompson, “Dietary control of exertional rhabdomyolysis in horses.,” Journal of the American Veterinary Medical Association, May 1998. doi: 10.2460/javma.1998.212.10.1588. View source
- C. Kerksick, S. Arent, B. Schoenfeld, J. R. Stout, B. Campbell, C. Wilborn, L. Taylor, D. Kalman, A. Smith‐Ryan, R. Kreider, D. Willoughby, P. Arciero, T. A. VanDusseldorp, M. Ormsbee, R. Wildman, M. Greenwood, T. Ziegenfuss, A. A. Aragon, and J. Antonio, “International society of sports nutrition position stand: nutrient timing,” Journal of the International Society of Sports Nutrition, Aug. 2017. doi: 10.1186/s12970-017-0189-4. View source
- F. Varasteh, S. H. Ebrahimi, A. Naserian, S. Zerehdaran, and V. Miri, “Effect of micronization and meal size of corn grain on glycemic response and in vitro hindgut acidosis potential in horses.,” Journal of Equine Veterinary Science, Dec. 2023. doi: 10.1016/j.jevs.2023.104982. View source
- N. W. Thorringer, M. R. Weisberg, and R. B. Jensen, “The effects of processing barley and maize on metabolic and digestive responses in Horses.,” Journal of Animal Science, Nov. 2020. doi: 10.1093/jas/skaa353. View source
- N. Luthersson, Ú. Ý. Þorgrímsdóttir, P. Harris, T. Parkin, C. Hopster-Iversen, and E. Bennet, “Risk Factors for Equine Gastric Ulcer Syndrome Incidence in Adult Icelandic Riding Horses,” Animals, Nov. 2023. doi: 10.3390/ani13223512. View source
- E. Leishman, M. Sahar, S. Cieslar, P. Darani, and J. L. Ellis, “What the hay: predicting equine voluntary forage intake using a meta-analysis approach.,” Animal, Jul. 2024. doi: 10.1016/j.animal.2024.101266. View source
- J. Gonzalez, “Novel dietary approaches to appetite regulation, health and performance,” Journal of Physiology, Mar. 2022. doi: 10.1113/JP282727. View source
- R. Mackenzie, “Skeletal muscle glycogen synthesis – it turns out we still know very little,” Journal of Physiology, Sep. 2020. doi: 10.1113/JP280759. View source
- L. O. Li, T. J. Grevengoed, D. S. Paul, O. Ilkayeva, T. Koves, F. Pascual, C. Newgard, D. Muoio, and R. Coleman, “Compartmentalized Acyl-CoA Metabolism in Skeletal Muscle Regulates Systemic Glucose Homeostasis,” Diabetes, Jul. 2014. doi: 10.2337/db13-1070. View source
- D. Kim, F. Hanzawa, H. Shimizu, S. Sun, M. Umeki, S. Ikeda, S. Mochizuki, and H. Oda, “Delayed feeding of a high-sucrose diet led to increased body weight by affecting the circadian rhythm of body temperature and hepatic lipid-metabolism genes in rats.,” Journal of Nutritional Biochemistry, Oct. 2022. doi: 10.1016/j.jnutbio.2022.109185. View source
- J. R. Weinert-Nelson, W. Meyer, and C. A. Williams, “Diurnal Variation in Forage Nutrient Composition of Mixed Cool-Season Grass, Crabgrass, and Bermudagrass Pastures.,” Journal of Equine Veterinary Science, Dec. 2021. doi: 10.1016/j.jevs.2021.103836. View source
- J. R. Weinert-Nelson, A. Biddle, H. Sampath, and C. A. Williams, “Fecal Microbiota, Forage Nutrients, and Metabolic Responses of Horses Grazing Warm- and Cool-Season Grass Pastures,” Animals, Feb. 2023. doi: 10.3390/ani13050790. View source
- N. Karikoski, J. R. Box, A. Mykkänen, V. V. Kotiranta, and M. Raekallio, “Variation in insulin response to oral sugar test in a cohort of horses throughout the year and evaluation of risk factors for insulin dysregulation,” Equine Veterinary Journal, May 2021. doi: 10.1111/evj.13529. View source
- N. M. M. D. Fonseca, C. Gibson, D. A. van Doorn, M. de Ruijter-Villani, T. Stout, and E. Roelfsema, “Effect of long‐term overfeeding of a high‐energy diet on glucose tolerance in Shetland pony mares,” Journal of Veterinary Internal Medicine, May 2020. doi: 10.1111/jvim.15788. View source
- C. J. Naydani and T. Coombs, “Exercise as a welfare strategy? Insights from horse (Equus caballus) owners in the UK,” Animal Welfare, Mar. 2025. doi: 10.1017/awf.2025.11. View source
- B. Gartland, W. Strunk, B. A. Schulte, F. J. DeGraves, and J. Koostra, “Time budgets differ in horses during continuous and space-restricted rotational grazing,” Veterinary and Animal Science, Jun. 2024. doi: 10.1016/j.vas.2024.100371. View source
- C. A. Williams, L. B. Kenny, J. R. Weinert, K. Sullivan, W. Meyer, and M. Robson, “Effects of 27 mo of rotational vs. continuous grazing on horse and pasture condition,” Translational Animal Science, Jun. 2020. doi: 10.1093/tas/txaa084. View source
- F. Bertin, “Diagnosis and management of equine metabolic syndrome,” Equine Veterinary Education, Mar. 2026. doi: 10.1111/eve.70094. View source
- Z. J. Williams, M. Bertels, and S. Valberg, “Muscle glycogen concentrations and response to diet and exercise regimes in Warmblood horses with type 2 Polysaccharide Storage Myopathy,” PLoS ONE, Sep. 2018. doi: 10.1371/journal.pone.0203467. View source
- K. Martinson, “58 Awardee Talk: A decade of discovery in horse pasture utilization,” Journal of Animal Science, Sep. 2024. doi: 10.1093/jas/skae234.249. View source
- А. Karynbayev, Z. Kuzembayuly, and А. Okshebaye, “PRODUCTIVITY OF PASTURES AND CHANGES IN VEGETATION QUALITY INDICATORS DEPENDING ON THEIR USE,” Ġylym ža̋ne bìlìm, Jun. 2025. doi: 10.52578/2305-9397-2025-2-3-99-108. View source
- S. Uysal, G. Li, and B. R. Claytor, “Searching for Clues in the Diagnosis of McArdle Disease,” Cureus, Jun. 2025. doi: 10.7759/cureus.86793. View source
- A. Sechi, L. Zuccarelli, B. Grassi, R. Frangiamore, R. S. D. Amicis, M. Marzorati, S. Porcelli, A. Tullio, A. Bacco, S. Bertoli, A. Dardis, L. Biasutti, M. Pasanisi, G. Devigili, and B. Bembi, “Exercise training alone or in combination with high-protein diet in patients with late onset Pompe disease: results of a cross over study,” Orphanet Journal of Rare Diseases, Apr. 2020. doi: 10.1186/s13023-020-01416-6. View source
- S. Valberg, Z. J. Williams, C. Finno, A. Schultz, D. Velez-Irizarry, M. Henry, K. Gardner, and J. Petersen, “Type 2 polysaccharide storage myopathy in Quarter Horses is a novel glycogen storage disease causing exertional rhabdomyolysis.,” Equine Veterinary Journal, Aug. 2022. doi: 10.1111/evj.13876. View source
This review is one of 34 in the Foxwatch research corpus. Methodology: PRISMA 2020-style systematic literature review against PubMed, CAB Abstracts, Scopus, Web of Science, and equine veterinary conference proceedings.













