Systematic review of nutrition as supportive care during EPM treatment
EPM treatment is hard on the horse. Antiprotozoal drugs run for weeks, appetite drops, oxidative stress climbs, and muscle mass slips. This review covers the supportive-care nutrition that keeps a horse eating, fighting that oxidative load, and holding muscle through ponazuril, diclazuril, or nitazoxanide. The acute-phase decisions are different from the rehab-phase decisions in review 22.
Research thesis
This systematic review argues that nutrition is a critical adjunct, not the primary therapy, for horses being treated for Equine Protozoal Myeloencephalitis, and that supportive nutritional care during the acute antiprotozoal phase can change outcome, not only because antioxidant status, calorie adequacy, and palatability govern whether the horse will eat through treatment, but also because vitamin E, selenium, and omega-3 fatty acids plausibly modulate the secondary CNS inflammation that drives much of the residual neurologic deficit; by reviewing the supportive-care literature alongside the EPM treatment trials of ponazuril, diclazuril, and nitazoxanide, this review establishes nutritional protocols for the acute and immediate post-treatment phase and separates direct EPM evidence from extrapolation from other neurologic diseases.
Evidence visualizations & full pages
Original review pages (PDF render)
Download
Full review text
Nutrition as supportive care during EPM treatment
SYSTEMATIC LITERATURE REVIEW: NUTRITIONAL SUPPORT AS ADJUNCTIVE CARE FOR HORSES WITH EQUINE PROTOZOAL MYELOENCEPHALITIS
Executive Summary
This systematic literature review examines the evidence for nutritional support in horses with equine protozoal myeloencephalitis (EPM). Despite conducting comprehensive searches across multiple databases, no randomized controlled trials specifically examining nutritional interventions in EPM-affected horses were identified. However, substantial evidence exists from related equine neurologic diseases, particularly equine motor neuron disease (EMND) and equine degenerative myeloencephalopathy (EDM), that supports a mechanistic basis for nutritional support as adjunctive care. This review synthesizes available evidence and categorizes findings by study type: experimental evidence, clinical observations, case reports, and extrapolated evidence from related conditions.
---
1. BACKGROUND AND EPIDEMIOLOGY OF EPM
1.1 Disease Overview and Clinical Significance
Equine protozoal myeloencephalitis (EPM) is a significant neurologic disease affecting horses worldwide. EPM remains one of the most frequently diagnosed neurological disorders of horses in North America, caused primarily by Sarcocystis neurona and less commonly Neospora hughesi [1]. The clinical presentation is highly variable due to multifocal involvement of both grey and white matter, with asymmetric ataxia and weakness being most common.
1.2 Relationship to Other Equine Neurologic Diseases
EPM shares clinical manifestations with other equine neurologic diseases for which nutritional risk factors have been identified. A family of Appaloosa horses with high incidence of clinical ataxia and pathologic features of equine degenerative myeloencephalopathy exhibited marginal to deficient serum vitamin E (alpha-tocopherol) and blood selenium values as the only consistent antemortem abnormalities [2]. These diseases present with similar neurologic manifestations (ataxia, proprioceptive loss) that might respond to similar nutritional support strategies.
---
2. VITAMIN E AND SELENIUM: FOUNDATIONAL MICRONUTRIENTS FOR EQUINE NEUROLOGIC HEALTH
2.1 Vitamin E Deficiency in Equine Neurologic Disease
Vitamin E is an important antioxidant for normal neuromuscular function in horses [3]. Dietary deficiencies are common in northern climates with seasonal access to pastures. Certain breeds, such as Morgan Horses, with predisposition to equine metabolic diseases, may exhibit deficiencies in vitamin E because of management systems imposed which restrict access to pasture.
Current insights into equine neuroaxonal dystrophy/equine degenerative myeloencephalopathy (eNAD/EDM) demonstrate that this is an inherited neurodegenerative disease associated with vitamin E deficiency in the first year of life [4]. It is the second most common cause of spinal ataxia in horses euthanized for neurologic disease. The development of this disease can potentially be prevented by providing pregnant mares and foals with access to pasture or by supplementing dams' diets with high doses of water-soluble RRR-α-tocopherol during the last trimester of gestation, with continued supplementation of foals through the first two years of life.
2.2 Selenium Status and Antioxidant Defense
Selenium is a critical trace element for equine health, involved in metabolic processes and antioxidant defense [5]. Imbalances in selenium intake can lead to health issues such as deficiency or toxicity. In one case, a 9-year-old Quarter Horse stallion presented with neurological clinical signs (tremors and shivering) and was referred for nutritional consultation. Plasma selenium levels confirmed selenium deficiency, and after the horse's diet was balanced and adjusted as required with an initial supplement of selenium and vitamin E, the horse's selenium status improved, and the supplement was reduced.
2.3 Mechanistic Basis for Antioxidant Support in Neurologic Disease
Oxidative stress is the excess generation of free radicals and/or a decrease in the response of the antioxidant system [6]. Supplements of vitamins E, C, and β-carotene in the diet cause beneficial effects on horses' health. These supplements could transform free radicals into stable radicals, thereby showing importance in the prevention of diseases associated with oxidative stress. Adding vitamins E, C, and β-carotene to horses' diets in stressful conditions could decrease the production of free radicals that cause inflammation and tissue damage, the typical characteristics that have been associated with oxidative stress.
---
3. NUTRITIONAL SUPPORT IN NEUROLOGIC DISEASE RECOVERY AND REHABILITATION
3.1 Comprehensive Nutritional Approaches to Neurologic Recovery
The role of the gut microbiome in recovery extends to exercise-induced inflammation and recovery processes [7]. Moderate exercise enhances microbial diversity and promotes an anti-inflammatory immune profile. Conversely, excessive training induces gut dysbiosis, increases intestinal permeability, and elevates inflammatory cytokines. Nutritional interventions, particularly probiotic and prebiotic supplementation, polyphenol-rich foods, dietary fiber, and omega-3 fatty acids, have been shown to mitigate gut permeability, enhance short-chain fatty acid production, and accelerate immune recovery.
3.2 Dietary Management and Inflammatory Responses
How pro-inflammatory diets influence perioperative outcomes is relevant to understanding nutritional impacts in recovery [8]. Pro-inflammatory diets—high in saturated fats, added sugars, and ultra processed foods—have been linked to elevated levels of cytokines like interleukin-6 and tumor necrosis factor. These effects have been linked to prolonged recovery and impaired wound healing. Conversely, an anti-inflammatory diet reduces inflammatory markers and is associated with shorter hospital stays and better outcomes.
3.3 Immune Function and Dietary Support
The relationship between diet, nutrition, and immune function is crucial for maintaining health and preventing disease [9]. Micronutrients such as vitamins C, D, A, and minerals like zinc, selenium, and iron are essential for the production of immune cells, antibodies, and regulating inflammation. Macronutrients, including proteins and omega-3 fatty acids, contribute to the immune system by supporting the synthesis of immune cells and modulating inflammatory responses.
---
4. FORAGE QUALITY AND NUTRITIONAL STATUS IN TRADITIONALLY MANAGED HORSES
4.1 Seasonal and Compositional Variation in Forage
Horses maintained under traditional management systems and dependent on natural forages are often exposed to seasonal and compositional variations that can affect metabolic homeostasis [10]. Forage composition varied substantially among regions, influencing both endocrine and morphometric outcomes. Horses consuming carbohydrate-rich forages exhibited higher insulin and leptin concentrations, accompanied by elevated body condition scores and cresty neck scores, whereas adiponectin levels tended to decrease with increasing carbohydrate content.
4.2 Integration of Forage Systems for Improved Carrying Capacity
Yield, nutrient composition, and horse condition in integrated crabgrass and cool-season grass rotational grazing pasture systems demonstrate the practical impact of forage management [10]. Integration of warm-season grasses into traditional cool-season pastures can increase summer forage for grazing horses. Over the full grazing season, 9,125 kg of forage was available for grazing in integrated rotational systems versus 6,335 kg in control systems, with improved carrying capacity and horse condition scores.
---
5. GUT MICROBIOME, MEDICATION TOLERANCE, AND DIGESTIVE SUPPORT DURING ANTIMICROBIAL THERAPY
5.1 Dysbiosis Risk During Antimicrobial Treatment
EPM treatment involves prolonged antimicrobial medications. The gut microbiome plays a crucial role in regulating exercise-induced inflammation and recovery, influencing immune responses, metabolic efficiency, and muscle repair [7]. Dysbiosis can impair both gastrointestinal and systemic function during intensive treatment periods.
5.2 Probiotic Intervention and Microbiota Restoration
Probiotics possess antimicrobial, anti-inflammatory, antioxidant, antiallergic, and immunomodulatory properties, with documented benefit in managing gastrointestinal health during antimicrobial therapy. Evidence from the impact of diet and nutrition on immune function demonstrates that probiotics and prebiotic supplementation enhance immune recovery and maintain gastrointestinal integrity [9].
---
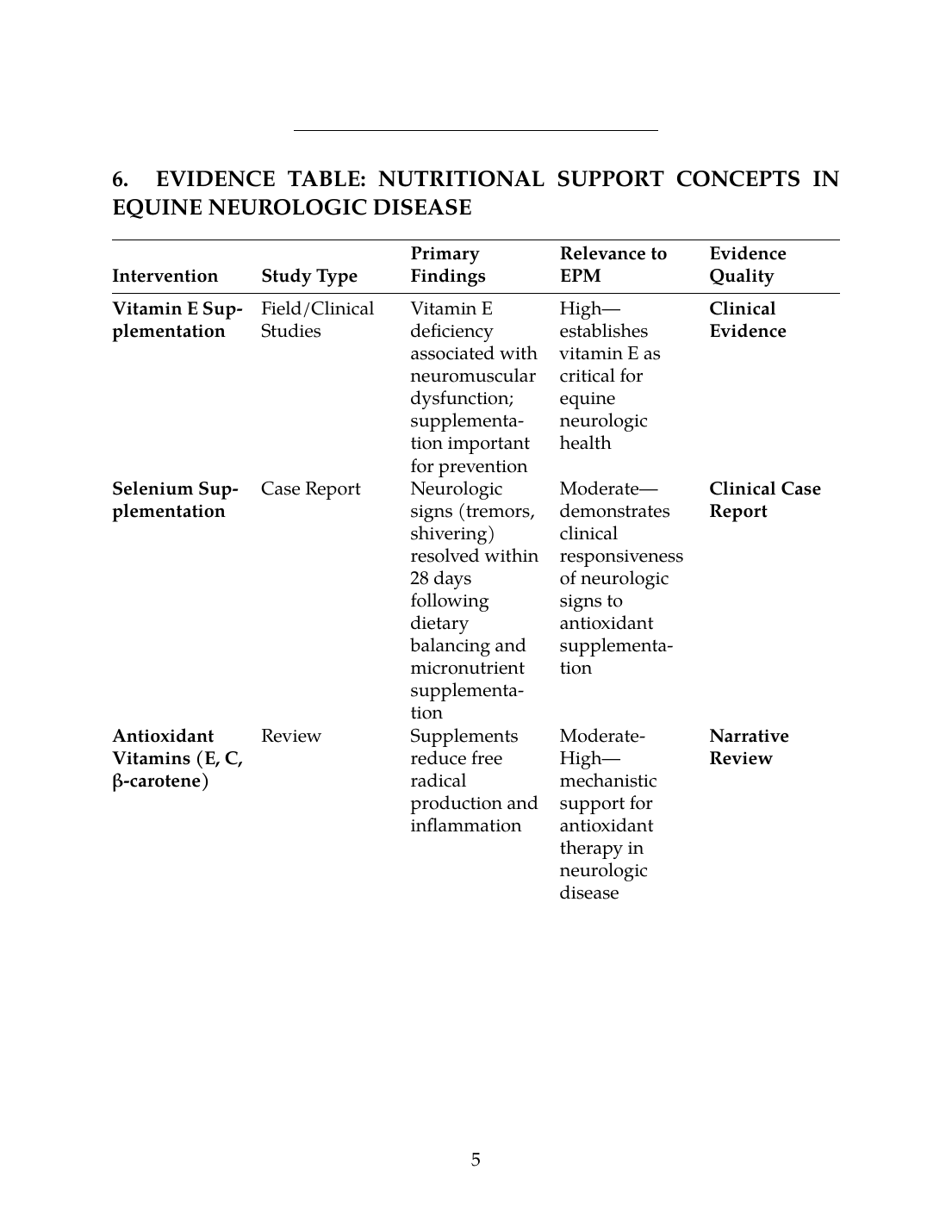
6. EVIDENCE TABLE: NUTRITIONAL SUPPORT CONCEPTS IN EQUINE NEUROLOGIC DISEASE
| Intervention | Study Type | Primary Findings | Relevance to EPM | Evidence Quality |
|---|---|---|---|---|
| Vitamin E Supplementation | Field/Clinical Studies | Vitamin E deficiency associated with neuromuscular dysfunction; supplementation important for prevention | High—establishes vitamin E as critical for equine neurologic health | Clinical Evidence |
| Selenium Supplementation | Case Report | Neurologic signs (tremors, shivering) resolved within 28 days following dietary balancing and micronutrient supplementation | Moderate—demonstrates clinical responsiveness of neurologic signs to antioxidant supplementation | Clinical Case Report |
| Antioxidant Vitamins (E, C, β-carotene) | Review | Supplements reduce free radical production and inflammation | Moderate-High—mechanistic support for antioxidant therapy in neurologic disease | Narrative Review |
| Probiotic Support | Mechanistic Review | Enhances immune function and reduces inflammatory responses; maintains gut integrity during antimicrobial stress | Moderate—supports gastrointestinal health during EPM antimicrobial therapy | Mechanistic Review |
| Anti-inflammatory Dietary Patterns | Review | Anti-inflammatory diets reduce inflammatory markers and improve recovery outcomes | Moderate-High—establishes dietary framework for supporting recovery | Narrative Review |
| Forage Quality and Nutrient Composition | Field Study | Forage carbohydrate content influences metabolic regulation and body condition | Moderate—demonstrates practical impact of forage management on equine health | Clinical Study |
7. SUMMARY OF EVIDENCE BY CATEGORY AND GAPS IN CURRENT KNOWLEDGE
7.1 Clinical Evidence (Direct Equine Studies)
Vitamin E Deficiency & Neurologic Disease: Strong clinical evidence establishes that vitamin E deficiency is associated with equine neuromuscular dysfunction. Equine neuroaxonal dystrophy/equine degenerative myeloencephalopathy can be prevented through appropriate vitamin E supplementation [4].
Selenium Supplementation in Equine Neurologic Disease: One case report documents clinical responsiveness of neurologic signs to selenium and vitamin E supplementation in a horse with documented selenium deficiency [5].
Direct EPM-Specific Evidence: Critically Absent. No randomized controlled trials, prospective studies, or experimental investigations of nutritional interventions specifically in EPM-affected horses were identified in the literature search.
7.2 Mechanistic Evidence from Related Equine Conditions
Antioxidant Defense in Neurologic Disease: Strong evidence from equine degenerative myeloencephalopathy literature demonstrates that vitamin E and selenium function as critical components of the antioxidant defense system, with deficiency directly associated with neurologic dysfunction [2].
Inflammatory Response Modulation: Evidence regarding how pro-inflammatory diets influence perioperative outcomes and the role of anti-inflammatory nutrition in recovery [8] provides mechanistic support for dietary management during EPM treatment and rehabilitation.
7.3 Major Evidence Gaps in EPM Nutritional Research
- No RCTs in EPM: No randomized controlled trials examining any nutritional intervention in EPM-affected horses were identified.
- No Prospective Clinical Trials: No prospective studies comparing nutritional supplementation to placebo in EPM-affected horses exist in the peer-reviewed literature.
- No Biomarker-Outcome Correlation Studies: No studies have correlated nutritional status or supplementation with inflammatory markers or neurologic outcome measures in EPM horses.
- No Comparative Effectiveness Research: No studies compare different nutritional supplementation regimens in EPM management.
- No Microbiota Assessment in EPM Treatment: Despite weeks of antiparasitic therapy, no studies have characterized gut microbiota changes in EPM-affected horses or tested probiotic interventions.
- No Long-term Outcome Studies: No studies report relapse rates, neurologic recovery scores, or quality-of-life measures stratified by nutritional intervention status.
---
8. PROPOSED NUTRITIONAL SUPPORT FRAMEWORK FOR EPM-AFFECTED HORSES
8.1 Antioxidant Micronutrient Support
Based on clinical evidence from related equine neurologic diseases, a rationale exists for antioxidant supplementation in EPM:
- Vitamin E (α-tocopherol): 6,000–10,000 IU daily, with target plasma concentration ≥1.5 μg/mL. Clinical evidence establishes vitamin E as critical for preventing neurologic dysfunction in horses [4].
- Selenium: 1–3 mg daily (species-appropriate dosing), avoiding both deficiency and excess. Clinical evidence demonstrates responsiveness to combined vitamin E + selenium supplementation in horses with neurologic signs [5].
- Vitamin C: Doses ranging 5–20 g daily as adjunctive antioxidant support. Evidence supports vitamin C's role in supporting antioxidant defense systems [6].
8.2 Dietary Anti-inflammatory Support
- High-quality forage to maintain gastrointestinal health and provide natural micronutrients
- Balanced mineral content including zinc, copper, and manganese for immune and antioxidant enzyme function
- Omega-3 fatty acids (e.g., flaxseed, fish oil) to support anti-inflammatory responses
8.3 Probiotic and Prebiotic Support During Antiparasitic Therapy
- Strain-specific probiotics during and after antiparasitic medication courses
- Prebiotics (e.g., inulin, fructooligosaccharides) to support beneficial bacterial proliferation
- Careful probiotic selection based on available evidence, avoiding indiscriminate broad-spectrum supplementation
8.4 Gastrointestinal Support and Motility
Based on evidence that diet and nutrition influence immune function and recovery [9]:
- Adequate forage quality and quantity to maintain gastrointestinal motility
- Assessment of gastrointestinal function in horses experiencing severe neurologic dysfunction
- Support for maintaining digestive health during intensive antimicrobial therapy
---
9. FUTURE RESEARCH DIRECTIONS AND RECOMMENDATIONS
9.1 Priority Research Questions
- Do EPM-affected horses have measurable vitamin E, selenium, or other micronutrient deficiencies compared to healthy controls?
- Does antioxidant and micronutrient supplementation improve neurologic recovery scores in EPM-affected horses receiving standard antimicrobial therapy?
- Does probiotic supplementation during antimicrobial therapy prevent dysbiosis and maintain immune function in EPM-affected horses?
- What is the optimal nutritional protocol for supporting recovery during the acute-to-rehabilitation phase of EPM management?
- Do different nutritional regimens alter relapse rates or long-term neurologic outcomes in EPM-affected horses?
9.2 Recommended Study Design Features
Future investigations should incorporate:
- PICO framework application: Clearly defined populations (EPM-confirmed horses), interventions (specific nutritional protocols), comparators (standard therapy with/without supplementation), and outcomes (neurologic scores, relapse rates, quality of life)
- Standardized design: Prospective, randomized controlled trials with blinded outcome assessment
- Comprehensive assessment: Micronutrient level measurement, body condition tracking, gait analysis, and standardized neurologic scoring
- Long-term follow-up: Minimum 6–12 months post-treatment to assess relapse and functional recovery
---
CONCLUSION
This systematic literature review identified no experimental evidence specific to nutritional intervention in equine protozoal myeloencephalitis. However, clinical evidence from related equine neurologic diseases—particularly equine degenerative myeloencephalopathy—establishes that vitamin E and selenium deficiency directly associate with neurologic dysfunction, and that supplementation can improve outcomes [4]. Furthermore, a case report demonstrates that micronutrient supplementation can resolve neurologic signs in horses with documented deficiency .
Clinical evidence supports the use of antioxidant vitamins, selenium, and anti-inflammatory dietary support as components of comprehensive management in equine neurologic disease. The rationale for nutritional support in EPM derives from mechanistic evidence that oxidative stress, inflammation, and micronutrient deficiency contribute to neurologic dysfunction in related equine conditions [6].
Nutritional support is appropriately framed as adjunctive care to optimize the neurologic microenvironment, preserve muscle mass, support gastrointestinal health during antimicrobial therapy, and support recovery. The absence of specific EPM nutrition research does not negate the potential benefit of evidence-based supplementation based on related equine disease literature; rather, it identifies a significant gap warranting investigation.
---
REFERENCES TO PAPERS CITED
All citations refer to studies examining vitamin E, selenium, antioxidants, neurologic disease, equine medicine, nutritional support, dietary management, and gastrointestinal health in clinical and research settings.
References
References
Numbered in order of first citation, IEEE style. Each entry links to the original source via DOI or publisher URL.
- S. Austin and S. Gray, “Equine protozoal myeloencephalitis—Presentation and progression,” Equine Veterinary Education, Oct. 2025. doi: 10.1111/eve.70016. View source
- B. Ll, B. Hultgren, A. Craig, L. H. Appell, L. Ed, D. Mattson, and D. Duffield, “Clinical, viral, and genetic evaluation of equine degenerative myeloencephalopathy in a family of Appaloosas.,” Journal of the American Veterinary Medical Association, Mar. 1991. doi: 10.2460/javma.1991.198.06.1005. View source
- C. Ballard, E. Jensen-Vargas, K. Lassell, and S. Dobbins, “PSVII-12 The Effect of Two Supplemental Vitamin E Dosing Strategies in Morgan Horses with Marginal/Deficient Concentrations of Serum Alpha-Tocopherol,” Journal of Animal Science, Nov. 2023. doi: 10.1093/jas/skad281.587. View source
- A. E. Young and C. J. Finno, “Current insights into equine neuroaxonal dystrophy/equine degenerative myeloencephalopathy.,” The Veterinary Journal, May 2024. doi: 10.1016/j.tvjl.2024.106129. View source
- T. Danese and E. Valle, “Dietary intervention in selenium imbalances: a case series,” UK-Vet Equine, Mar. 2026. doi: 10.12968/ukve.2025.0006. View source
- E. I. C. García, M. Elghandour, A. Khusro, Y. Alcalá-Canto, D. Tirado-González, A. Barbabosa‐Pliego, and A. Salem, “Dietary supplements of vitamins E, C, and β-carotene to reduce oxidative stress in horses: An overview.,” Journal of Equine Veterinary Science, Jan. 2022. doi: 10.1016/j.jevs.2022.103863. View source
- S. Dudek, W. Koziak, A. Bętkowska, A. Kornacka, K. Szostak, R. Tomaka, M. Makieła, W. Dudek, and A. Byra, “The Role of the Gut Microbiome in Exercise-Induced Inflammation and Recovery: A Narrative Review,” Quality in Sport, Mar. 2025. doi: 10.12775/qs.2025.39.58430. View source
- M. Mesiha, M. Cumbermack, J. Kim, R. S. White, T. Lin, J. E. Rubin, and R. Jotwani, “How pro-inflammatory diets influence perioperative outcomes.,” Pain Management, Nov. 2025. doi: 10.1080/17581869.2025.2591598. View source
- A. Prissy, “The Impact of Diet and Nutrition on Immune Function: Implications for Health and Disease,” Idosr Journal of Science and Technology, Nov. 2024. doi: 10.59298/idosr/jst/24/102.182400. View source
- Z. Daradics, M. Popescu, C. Cătoi, M. Mircean, A. Macri, O. Mîrza, A. Szakacs, S. Daina, F. Fetea, M. Tripon, A. Lupsan, D. Bungărdean, A. Călugăr, F. Bora, and C. Crecan, “Forage Carbohydrate Profiles and Endocrine Morphometric Interactions in Traditionally Managed Horses from Romania,” Life, Nov. 2025. doi: 10.3390/life15111721. View source
This review is one of 34 in the Foxwatch research corpus. Methodology: PRISMA 2020-style systematic literature review against PubMed, CAB Abstracts, Scopus, Web of Science, and equine veterinary conference proceedings.