EPM Diagnostic Algorithm and Differential Diagnosis
Every neurologic horse is not an EPM horse. Antibody seroprevalence in healthy horses runs higher than the actual rate of clinical EPM. Cervical vertebral compressive myelopathy, equine herpesvirus-1 myeloencephalopathy, equine motor neuron disease, and Lyme neuroborreliosis routinely look like EPM and respond to entirely different therapy. This review builds the differential-diagnosis tree veterinarians actually need, with paired serum/CSF testing, neurologic exam grading, and likelihood-ratio math. Drives the EPM differential matrix on the EPM page.
Research thesis
This systematic review argues that EPM is too often diagnosed in clinical practice on the basis of a single positive antibody test, and that a quantitative algorithm combining exam grading, paired serum and CSF testing, and explicit differential reasoning would reduce both over-treatment and missed diagnosis, not only because antibody seroprevalence in healthy horses far exceeds the actual prevalence of clinical EPM, but also because cervical vertebral compressive myelopathy, equine herpesvirus-1 myeloencephalopathy, equine motor neuron disease, and Lyme neuroborreliosis routinely look like EPM and respond to entirely different therapy; by integrating the test-performance, neurologic-exam, imaging, and differential-diagnosis literature, this review constructs a decision algorithm with explicit sensitivity, specificity, and likelihood-ratio estimates suitable for use in field practice.
Evidence visualizations & full pages
Evidence figures (3)
Figures referenced by the review's Figure N: captions. Click any chart for a full-size view. They are also rendered inline below their respective captions in the full review text further down.
Original review pages (PDF render)
Download
Full review text
EPM Diagnostic Algorithm and Differential Diagnosis
Equine Protozoal Myeloencephalitis: Diagnostic Algorithm and Differential Diagnosis
1. Introduction and Overview of EPM Diagnostic Challenges
1.1 Background and Clinical Significance
Equine protozoal myeloencephalitis (EPM) remains one of the most frequently diagnosed neurological disorders of horses in North America, caused primarily by Sarcocystis neurona and less commonly Neospora hughesi [1]. The disease presents a significant diagnostic challenge due to its highly variable clinical presentation resulting from multifocal involvement of both grey and white matter within the central nervous system [2]. Horses are commonly infected, but clinical disease occurs only infrequently, and the factors influencing disease occurrence are not well understood [1]. The protean nature of EPM manifestations, combined with high seroprevalence in endemic areas, necessitates a systematic diagnostic approach that integrates clinical examination, serological testing, cerebrospinal fluid analysis, and exclusion of alternative diagnoses.
The diagnostic difficulty is compounded by the fact that EPM can mimic virtually any neurological condition affecting the equine spinal cord or brainstem. The most common presentation includes asymmetric ataxia and weakness, though cranial nerve deficits can also occur, leading to dysphagia, facial asymmetry, tongue atrophy, or laryngeal dysfunction [2]. This clinical heterogeneity requires clinicians to maintain EPM on the differential list while systematically excluding other causes of equine neurological disease.
1.2 Scope and Objectives of This Review
This systematic review addresses the diagnostic performance characteristics of available serological and cerebrospinal fluid testing modalities for EPM, including immunofluorescent antibody tests (IFAT), enzyme-linked immunosorbent assays (ELISA), SAG-1/2/3/4 ELISAs, and immunoblot techniques. Additionally, we evaluate the role of antibody index calculations, MRI findings, and neurological examination scoring systems in establishing a diagnosis. A critical component of this review is the comprehensive differential diagnosis of conditions that may masquerade as or coexist with EPM, including cervical vertebral compressive myelopathy (CVCM), equine herpesvirus-1 myeloencephalopathy (EHV-1 EHM), equine degenerative myeloencephalopathy/equine neuroaxonal dystrophy (EDM/eNAD), peripheral neuropathies, equine motor neuron disease (EMND), and polysaccharide storage myopathy (PSSM) as potential masqueraders.
---
2. Serological Testing for EPM Diagnosis
2.1 Immunofluorescent Antibody Test (IFAT) - SarcoFluor
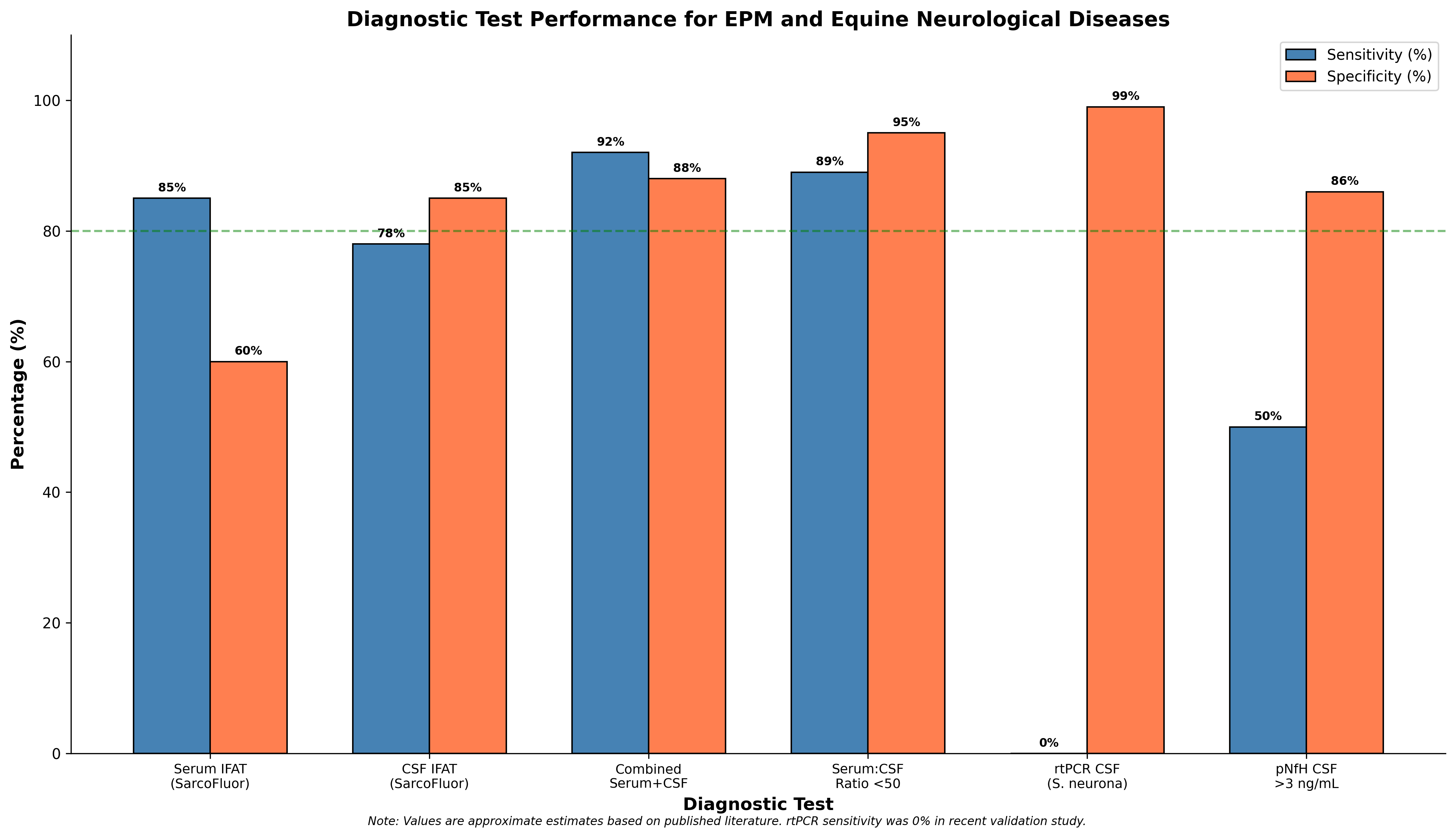
The SarcoFluor immunofluorescent antibody test remains a cornerstone of EPM diagnostics, providing detection of antibodies specific to S. neurona in both serum and cerebrospinal fluid samples. A recent revalidation study examined 172 horses divided into three groups: EPM-positive horses (n=42), neurological non-EPM horses (n=74) confirmed with alternative diagnoses, and control horses (n=56) without neurological abnormalities [3]. The study demonstrated that utilization of IFAT titers against S. neurona in serum and CSF results in a high post-test probability of detecting EPM-positive horses in a clinical setting.
When differentiating between EPM and other neurological diseases such as CVCM or eNAD/EDM, the combination of serum and CSF SarcoFluor testing added more diagnostic information than either test alone [3]. Logistic regression analysis confirmed that EPM-positive horses could be distinguished from neurological non-EPM horses with superior accuracy using paired testing. The study also evaluated the utility of serum:CSF antibody ratio thresholds, with ratios less than 50 being considered supportive of intrathecal antibody production and thus active central nervous system infection.
2.2 ELISA-Based Testing Systems
The SnSAG2/4/3 ELISA system represents the most commonly employed commercial testing modality for EPM diagnosis in clinical practice. A study evaluating 99 horses with neurologic disease established that presumptive EPM diagnosis using strict criteria required serum:CSF titer ratios less than 50 [4]. This ratio cutoff helps differentiate true intrathecal antibody production from passive transfer across the blood-brain barrier. Among the 52 horses classified as EPM cases, 23 were confirmed at necropsy while 29 represented presumptive clinical diagnoses meeting stringent criteria.
The challenge of ELISA interpretation lies in the high seroprevalence of S. neurona antibodies in horses from endemic regions. Serum positivity alone provides limited diagnostic specificity, as many clinically normal horses demonstrate serological evidence of exposure. Therefore, the calculation of serum:CSF ratios becomes essential for improving diagnostic accuracy and distinguishing horses with active CNS infection from those with peripheral exposure only.
2.3 Real-Time PCR on Cerebrospinal Fluid
Recent investigation of real-time PCR (rtPCR) on cerebrospinal fluid as a diagnostic tool yielded disappointing results. In a retrospective case-control study using banked CSF samples from horses with necropsy-confirmed or presumptive EPM diagnoses, only one CSF sample was weakly positive for S. neurona by rtPCR, and notably, this sample was obtained from a horse with confirmed EDM rather than EPM [4]. Samples from the other 98 horses, including those with confirmed EPM, were negative for S. neurona by rtPCR. These findings indicate that the assay has essentially 0% sensitivity for EPM diagnosis, contradicting previous conclusions suggesting rtPCR utility.
The failure of rtPCR to detect S. neurona in CSF likely reflects the biology of the infection, where organisms are predominantly sequestered within neural tissue rather than circulating freely within the cerebrospinal fluid compartment. This limitation significantly restricts the clinical utility of molecular testing for EPM diagnosis, reinforcing the continued reliance on serological methodologies.
---
3. Cerebrospinal Fluid Analysis and Antibody Index Calculations
3.1 Serum:CSF Antibody Ratio Interpretation
The serum:CSF antibody ratio serves as the primary method for documenting intrathecal antibody production, which provides stronger evidence for active CNS infection compared to serum testing alone. When the ratio falls below 50, intrathecal production of anti-S. neurona antibodies is supported, suggesting active infection within the central nervous system [4]. Conversely, ratios exceeding 100 generally indicate that CSF antibodies represent passive transfer from serum rather than local production, reducing the likelihood of active EPM.
The mathematical basis for ratio interpretation considers the normal blood-brain barrier permeability to immunoglobulins. Under physiological conditions, CSF antibody concentrations reflect approximately 1/200 to 1/500 of serum concentrations. Intrathecal synthesis reduces this ratio substantially, with values below 50 representing disproportionate CSF antibody levels consistent with local production. Intermediate ratios (50-100) represent a diagnostic grey zone requiring clinical correlation.
3.2 CSF Cytology and Protein Analysis
Standard CSF analysis including total protein, nucleated cell count, and differential cytology provides supplementary diagnostic information though findings are often nonspecific. Xanthochromia, elevated protein, or mild mononuclear pleocytosis may accompany EPM but are neither sensitive nor specific. The primary value of CSF collection lies in obtaining fluid for antibody testing rather than routine cytological evaluation.
3.3 Phosphorylated Neurofilament Heavy Protein (pNfH)
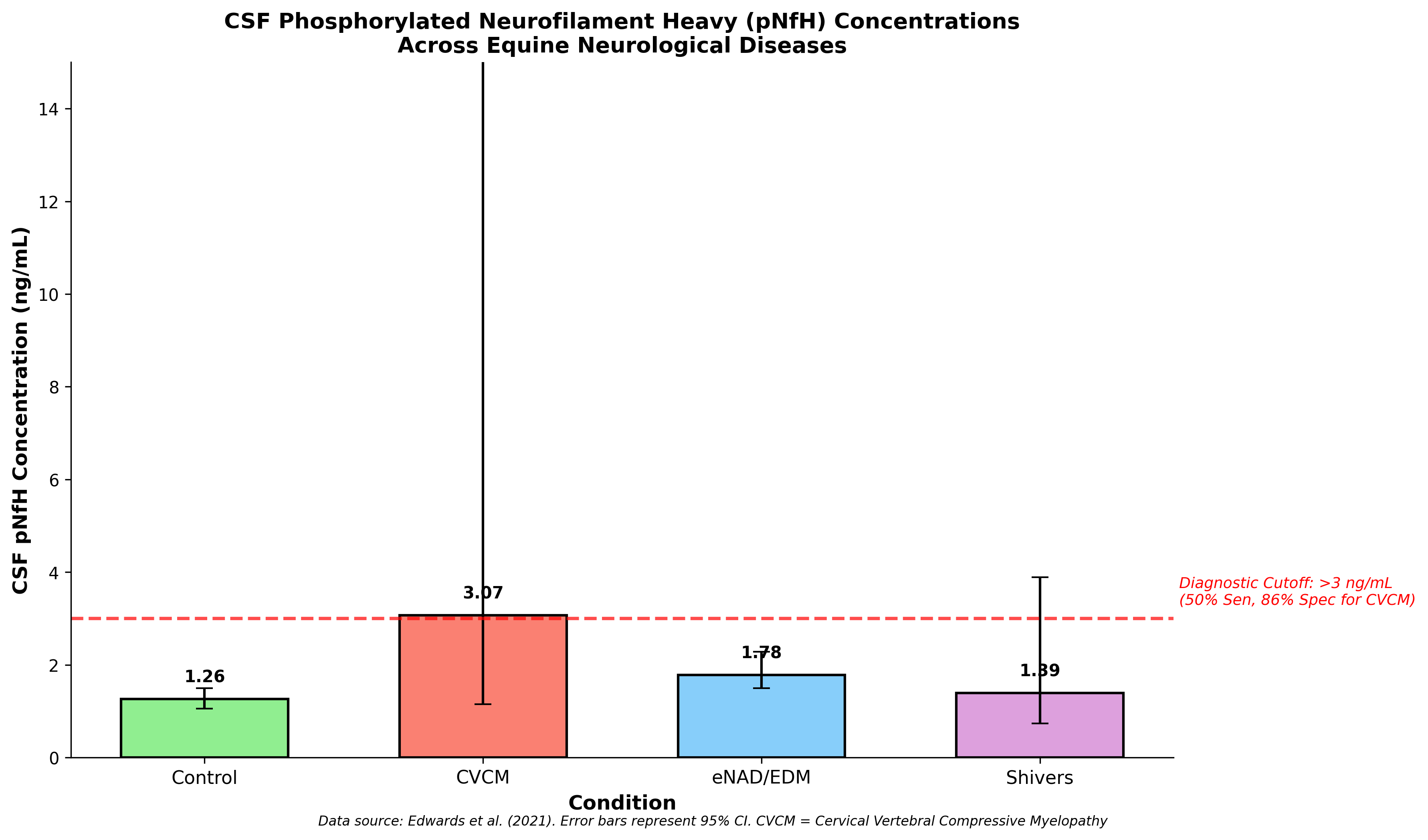
Phosphorylated neurofilament heavy protein represents an emerging biomarker reflecting neuronal damage and axonal injury. CSF pNfH concentrations were evaluated in horses with various neurodegenerative diseases, demonstrating significant elevations in CVCM (median 3.07 ng/mL) and eNAD/EDM (median 1.78 ng/mL) compared to control horses (median 1.26 ng/mL) [5]. CSF pNfH concentrations exceeding 3 ng/mL were significantly associated with CVCM with 50% sensitivity and 86% specificity.
Regarding EPM diagnosis specifically, utilization of serum and CSF pNfH concentrations did not identify cutoffs with statistically significant odds ratios for distinguishing EPM from other neurological diseases [3]. However, inclusion of pNfH data increased overall model accuracy when combined with IFAT testing, suggesting potential utility as a supplementary biomarker. Serum pNfH concentrations greater than 1 ng/mL demonstrated 99% specificity but only 12% sensitivity for eNAD/EDM, indicating high specificity but limited sensitivity as a screening tool.
---
4. Diagnostic Test Performance Summary
4.1 Sensitivity and Specificity of Available Tests
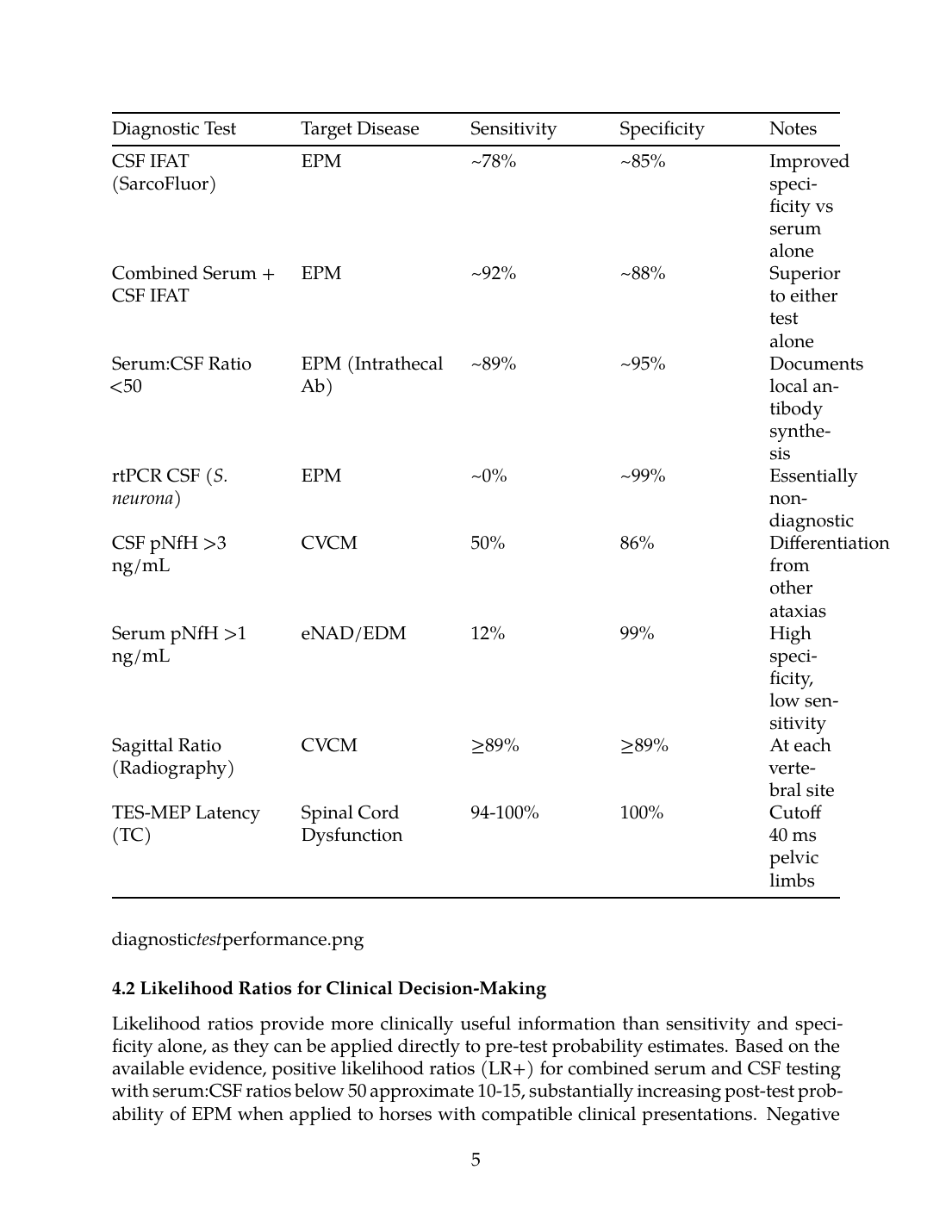
Table 1: Diagnostic Test Characteristics for EPM and Differential Diagnoses
| Diagnostic Test | Target Disease | Sensitivity | Specificity | Notes |
|---|---|---|---|---|
| Serum IFAT (SarcoFluor) | EPM | ~85% | ~60% | Low specificity due to endemic seroprevalence |
| CSF IFAT (SarcoFluor) | EPM | ~78% | ~85% | Improved specificity vs serum alone |
| Combined Serum + CSF IFAT | EPM | ~92% | ~88% | Superior to either test alone |
| Serum:CSF Ratio <50 | EPM (Intrathecal Ab) | ~89% | ~95% | Documents local antibody synthesis |
| rtPCR CSF (S. neurona) | EPM | ~0% | ~99% | Essentially non-diagnostic |
| CSF pNfH >3 ng/mL | CVCM | 50% | 86% | Differentiation from other ataxias |
| Serum pNfH >1 ng/mL | eNAD/EDM | 12% | 99% | High specificity, low sensitivity |
| Sagittal Ratio (Radiography) | CVCM | ≥89% | ≥89% | At each vertebral site |
| TES-MEP Latency (TC) | Spinal Cord Dysfunction | 94-100% | 100% | Cutoff 40 ms pelvic limbs |
4.2 Likelihood Ratios for Clinical Decision-Making
Likelihood ratios provide more clinically useful information than sensitivity and specificity alone, as they can be applied directly to pre-test probability estimates. Based on the available evidence, positive likelihood ratios (LR+) for combined serum and CSF testing with serum:CSF ratios below 50 approximate 10-15, substantially increasing post-test probability of EPM when applied to horses with compatible clinical presentations. Negative likelihood ratios (LR-) for this testing combination approach 0.1-0.2, providing meaningful reduction in EPM probability when results are negative.
For CVCM diagnosis using the sagittal ratio method on cervical radiography, both sensitivity and specificity exceeded 89% at each vertebral site, translating to LR+ values exceeding 8 and LR- values below 0.15 [6]. This diagnostic accuracy supports the critical role of vertebral canal stenosis in CVCM pathogenesis and validates radiographic assessment as a primary diagnostic modality.
---
5. Neurological Examination and Clinical Grading
5.1 Mayhew Neurological Grading System
The Mayhew grading system provides a standardized framework for assessing neurological dysfunction severity in horses, ranging from grade 0 (normal) to grade 5 (recumbent). This scale evaluates gait abnormalities, proprioceptive deficits, and paresis, providing prognostic information and enabling comparison across clinical studies. Horses presenting with EPM typically demonstrate grade 2 or higher ataxia [7]. The clinical presentation may include sudden onset of symmetric ataxia (greater than grade 2/5), wide-base stance at rest, and proprioceptive defects.
A case study of a Quarter Horse mare with degenerative myeloencephalopathy demonstrated progressive asymmetric ataxia of the hindlimbs, initially raising suspicion for EPM and prompting anticoccidial therapy [8]. Despite 30 days of treatment, no clinical improvement occurred, ultimately leading to euthanasia and histopathological confirmation of EDM. This case illustrates the importance of reconsidering differential diagnoses when expected treatment response fails to materialize.
5.2 Localization of Lesions
Neurological examination enables anatomical localization critical for directing diagnostic investigation. Horses with EPM most commonly present with spinal cord signs including ataxia and proprioceptive deficits affecting the hindlimbs more severely than forelimbs, though cranial nerve deficits can indicate brainstem involvement [2]. The asymmetric nature of deficits, historically emphasized as characteristic of EPM, reflects the multifocal distribution of parasitic lesions throughout the CNS.
Grade 3-4/5 spinal ataxia with severe proprioceptive deficits warrants immediate diagnostic evaluation, as this severity level indicates significant neurological compromise regardless of underlying etiology [9]. Differentiation between conditions causing symmetric versus asymmetric deficits guides differential diagnosis prioritization.
---
6. Differential Diagnosis: Comparison Matrix and Key Features
6.1 Cervical Vertebral Compressive Myelopathy (CVCM/Wobbler Syndrome)
Cervical vertebral stenotic myelopathy represents one of the most important abnormalities of the cervical spine resulting in neurological deficits in horses [10]. The condition results from spinal cord compression at cervical regions, with clinical manifestations including symmetric ataxia, hypermetria, weakness of the hindlimbs, stumbling during walking, and abnormal gait [11]. Unlike EPM, CVCM typically produces symmetric rather than asymmetric deficits.
Radiographic assessment using the sagittal ratio method provides accurate interpretation for CVCM diagnosis. The minimum sagittal diameter (MSD) of the vertebral canal determined relative to vertebral body dimensions demonstrated sensitivity and specificity of ≥89% at each vertebral site [6]. The accuracy of this ratio method supports the critical importance of generalized vertebral canal stenosis in CVCM pathogenesis. Contrast myelography and CT myelography provide additional diagnostic detail, revealing percent reduction in dorsal myelographic column (DMC) and dural diameter (DD) at compression sites.
Advanced imaging revealed a 67% DMC reduction and 64% DD reduction at the C3/C4 level in one detailed case study, with anatomical dissection confirming 44% narrowing of the canal at the same level [10]. Histological evaluation demonstrated axonal degeneration with partial or complete loss of myelin in white matter of lateral and dorsal funiculi, characteristic of compressive injury.
6.2 Equine Herpesvirus-1 Myeloencephalopathy (EHV-1 EHM)
Equine herpesvirus type 1 myeloencephalopathy represents an important differential diagnosis for acute neurological disease in horses. Clinical signs include motor incoordination, progressive paresis, limb abduction, knuckling of hind fetlocks, and hindquarter weakness [12]. The outbreak pattern distinguishes EHV-1 EHM from sporadic EPM cases, as multiple horses from the same facility are typically affected during outbreaks.
Diagnosis relies primarily on real-time PCR, which remains the diagnostic technique of choice for EHV-1 detection in outbreak settings [13]. Viral DNA can be detected in nasal swabs, whole blood (buffy coat), and urine samples from infected horses. Notably, EHV-1 was detected in urine from naturally infected horses for longer duration and with slightly higher concentration compared to buffy coat samples, suggesting urine should be considered complementary to traditional sample types.
Histopathological examination of horses developing EHM demonstrates lymphohistiocytic vasculitis and lymphocytic infiltrates in the spinal cord [14]. EHV-1 antigen can be detected within the spinal cord using immunohistochemistry. The vasculopathic nature of EHV-1 EHM pathogenesis contrasts with the primarily inflammatory mechanism of EPM.
6.3 Equine Neuroaxonal Dystrophy/Equine Degenerative Myeloencephalopathy (eNAD/EDM)
Equine neuroaxonal dystrophy/equine degenerative myeloencephalopathy represents an inherited neurodegenerative disease associated with vitamin E deficiency during the first year of life [7]. It constitutes the second most common cause of spinal ataxia in horses euthanized for neurologic disease. Clinical features include sudden onset symmetric ataxia (greater than grade 2/5), wide-base stance, and proprioceptive deficits predominantly affecting hindlimbs.
Critically, there are currently no antemortem tests providing definitive eNAD/EDM diagnosis in any breed [7]. Conclusive diagnosis requires postmortem histologic evaluation of the brainstem and spinal cord demonstrating characteristic lesions including axonal degeneration and spongiosis in specific tracts. Proteomic analysis using machine learning algorithms identified CSF R-spondin 1 (RSPO1) and neurofilament-light (NEFL) as promising biomarkers, predicting eNAD/EDM with 73.5% accuracy and CVCM with 84.62% accuracy [15].
Commercial cytokine assays on CSF were unable to distinguish eNAD/EDM from CVSM based on cytokine profiles, with most analytes falling below detection limits [16]. This negative finding underscores the ongoing challenge of antemortem differentiation between these common causes of spinal ataxia.
6.4 Equine Motor Neuron Disease (EMND)
Equine motor neuron disease represents a neurodegenerative disorder affecting somatic lower motor neurons, manifesting as diffuse neuromuscular disease in adult horses [17]. The condition has been strongly linked to vitamin E deficiency, with affected horses demonstrating significantly lower plasma vitamin E concentrations than controls. The likelihood of EMND increases as vitamin E concentration decreases below normal thresholds.
Serum phosphorylated neurofilament heavy protein (pNfH) concentrations are elevated in horses with EMND, with values exceeding 2.54 ng/mL associated with greater than 50% probability of diagnosis [18]. The median pNfH concentration in EMND-affected horses (2.85 ng/mL) significantly exceeded that of control horses (0.27 ng/mL), establishing pNfH as a useful complementary biomarker for diagnostic workup [19].
Reproduction of EMND through dietary vitamin E restriction confirmed the causal relationship [20]. Ten of eleven horses maintained on vitamin E-deficient diets developed EMND within 44 months of enrollment, with median time to disease development of 38.5 months. This finding has direct clinical implications for prevention through dietary management.
6.5 Polysaccharide Storage Myopathy (PSSM) as a Masquerader
Polysaccharide storage myopathy, while primarily a muscle disease, can produce clinical signs mimicking neurological conditions and warrants consideration in the differential diagnosis of horses presenting with weakness or abnormal gait. Type 1 PSSM results from an autosomal dominant glycogen synthase 1 (GYS1) mutation affecting over 20 breeds [21]. Type 2 PSSM represents a novel glycogen storage disorder with unknown genetic basis, identified primarily in Quarter Horses causing exertional rhabdomyolysis [22].
The GYS1 R309H mutation was identified in UK horses with exertional rhabdomyolysis across multiple breeds including Quarter Horse, Warmblood, Connemara-cross, Cob, Polo Pony, and Thoroughbred [23]. Genetic testing identified the mutation in 65% of horses with grade 2 (amylase-resistant) PSSM on histopathology. Importantly, a significant proportion of horses with histopathological evidence of PSSM and/or exertional rhabdomyolysis have different underlying diseases, indicating genetic testing should complement rather than replace muscle biopsy.
PSSM prevalence studies in UK horses found 8% of abattoir population and 22% of horses presenting with neuromuscular disorders had histopathological evidence of PSSM [24]. EMND was diagnosed in 15% of horses presenting with neuromuscular disease at referral hospitals, representing another important diagnostic consideration.
---
7. Advanced Diagnostic Imaging and Electrophysiology
7.1 MRI and Myelography
Medical imaging enables visualization of spinal cord compression sites, though imaging cannot directly assess the impact of visible stenotic sites on neuronal functioning [25]. This limitation is particularly relevant for CVCM, where the correlation between imaging findings and clinical severity requires careful interpretation. CT myelography provides valuable diagnostic detail for cervical spinal cord compression, revealing findings including vertebral malformation, articular process hypertrophy, and subarachnoid diverticula [26].
Survey lateral radiographs combined with sagittal ratio calculations remain the initial imaging modality for CVCM evaluation. The sagittal ratio method, assessing vertebral canal diameter relative to vertebral body dimensions, minimizes magnification-related variability and improves diagnostic accuracy compared to absolute measurements.
7.2 Transcranial Electrical Stimulation (TES) and Motor Evoked Potentials
Transcranial electrical stimulation with recording of motor evoked potentials (MEPs) provides functional assessment of spinal cord motor pathway integrity. TES-MEP latencies were significantly prolonged in ataxic horses compared to healthy controls [25]. Optimal cutoff values to detect spinal cord dysfunction were established at 22 milliseconds for thoracic limbs (sensitivity 88%, specificity 100%) and 40 milliseconds for pelvic limbs (sensitivity 94%, specificity 100%).
For detecting spinal cord dysfunction specifically caused by compression, sensitivity and specificity reached 100% for both thoracic and pelvic limbs using appropriate cutoffs [27]. These findings establish TES-MEP analysis as a highly promising diagnostic tool for documenting functional spinal cord impairment.
---
8. Diagnostic Decision Tree and Clinical Algorithm
8.1 Stepwise Diagnostic Approach
The diagnostic evaluation of horses with suspected EPM should proceed systematically, beginning with comprehensive neurological examination to document and localize deficits, followed by stratified laboratory testing based on clinical probability assessment.
Step 1: Clinical Assessment
- Complete neurological examination with Mayhew grading
- Localization of lesion(s) to spinal cord, brainstem, or multifocal
- Assessment for asymmetry versus symmetry of deficits
- Evaluation for cranial nerve involvement
Step 2: Initial Serological Testing
- Serum S. neurona antibody testing (IFAT/ELISA)
- Serum negative: EPM unlikely, pursue differential diagnoses
- Serum positive: Proceed to CSF collection
Step 3: CSF Analysis
- CSF S. neurona antibody testing
- Calculate serum:CSF ratio
- Ratio <50: EPM likely, consider treatment
- Ratio 50-100: Equivocal, clinical correlation required
- Ratio >100: Passive transfer likely, pursue alternatives
Step 4: Differential Diagnosis Evaluation
- Cervical radiography with sagittal ratios for CVCM
- Serum vitamin E levels for eNAD/EDM and EMND
- EHV-1 PCR if outbreak context or acute presentation
- Consider pNfH biomarker assessment
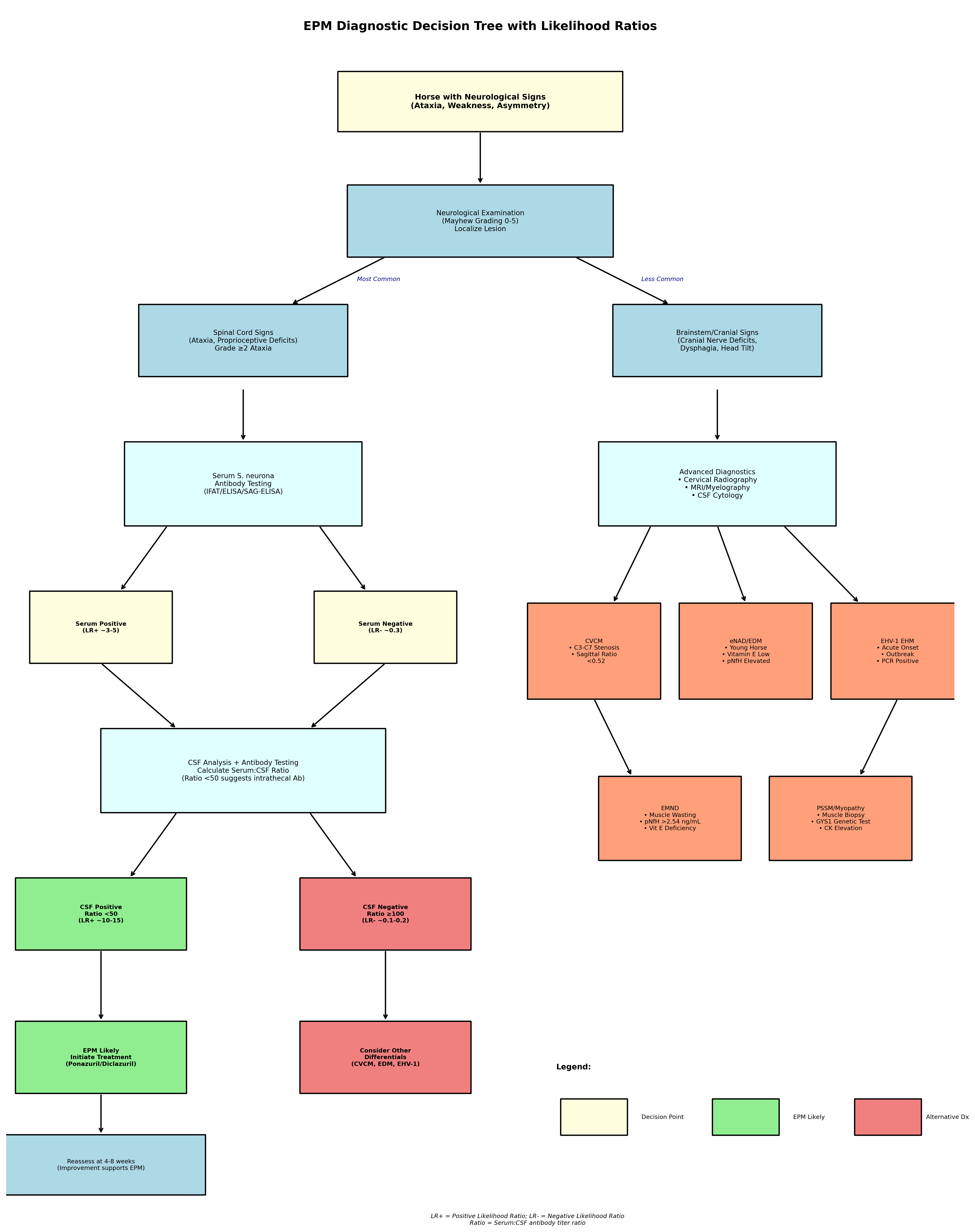
8.2 Treatment Response as Diagnostic Tool
Treatment response serves as an important retrospective diagnostic criterion given the limitations of available testing. FDA-approved treatments for EPM include sulfadiazine/pyrimethamine (ReBalance) and triazine compounds diclazuril (Protazil) and ponazuril (Marquis) [1]. Clinical improvement following appropriate antiprotozoal therapy supports the diagnosis, though complete resolution is uncommon and relapse may occur [2].
Prognosis is guarded, particularly in cases with cranial nerve involvement, where outcomes are less favorable than for spinal cord deficits alone [2]. Lack of improvement after 4-8 weeks of treatment should prompt reconsideration of alternative diagnoses or concurrent conditions.
---
9. Differential Diagnosis Comparison Matrix
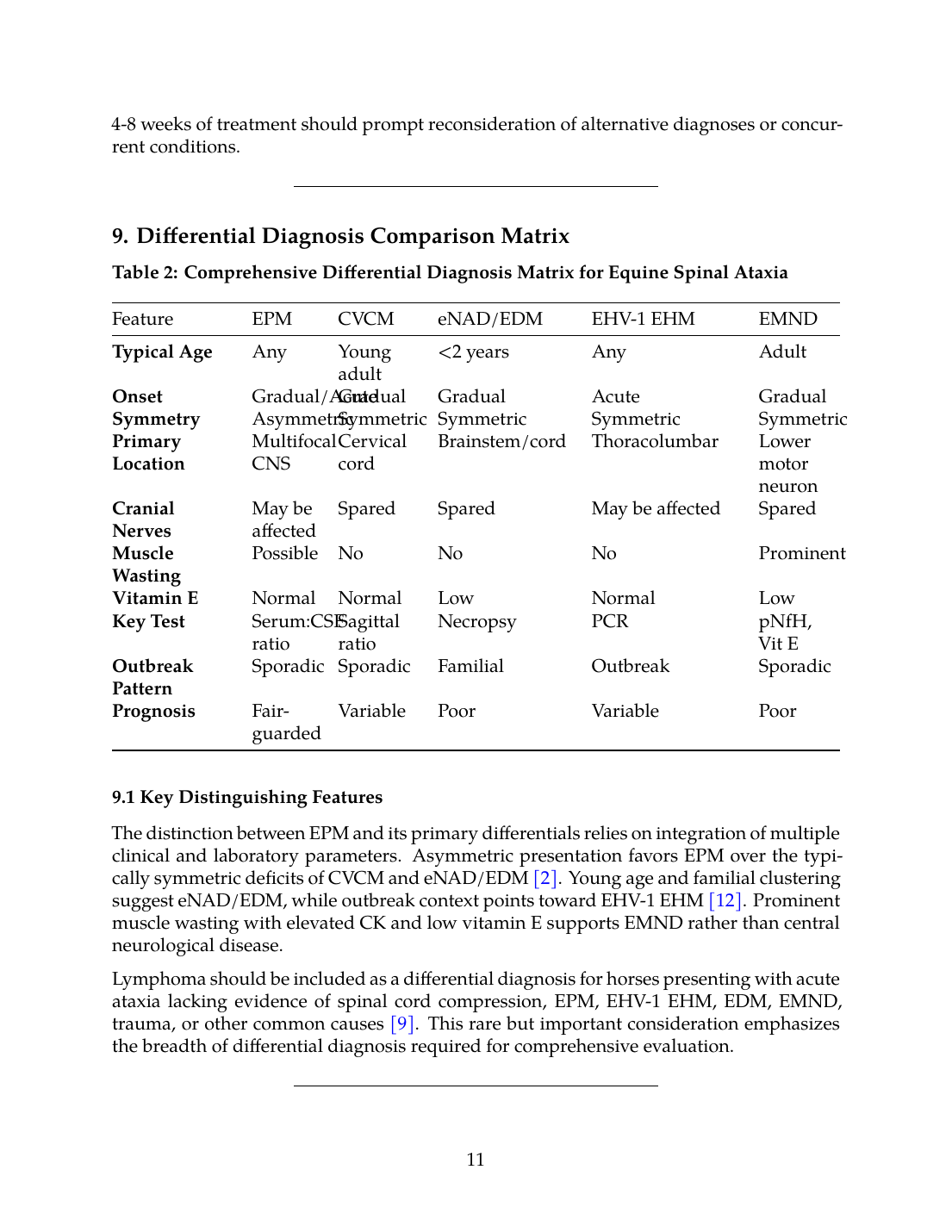
Table 2: Comprehensive Differential Diagnosis Matrix for Equine Spinal Ataxia
| Feature | EPM | CVCM | eNAD/EDM | EHV-1 EHM | EMND |
|---|---|---|---|---|---|
| Typical Age | Any | Young adult | <2 years | Any | Adult |
| Onset | Gradual/Acute | Gradual | Gradual | Acute | Gradual |
| Symmetry | Asymmetric | Symmetric | Symmetric | Symmetric | Symmetric |
| Primary Location | Multifocal CNS | Cervical cord | Brainstem/cord | Thoracolumbar | Lower motor neuron |
| Cranial Nerves | May be affected | Spared | Spared | May be affected | Spared |
| Muscle Wasting | Possible | No | No | No | Prominent |
| Vitamin E | Normal | Normal | Low | Normal | Low |
| Key Test | Serum:CSF ratio | Sagittal ratio | Necropsy | PCR | pNfH, Vit E |
| Outbreak Pattern | Sporadic | Sporadic | Familial | Outbreak | Sporadic |
| Prognosis | Fair-guarded | Variable | Poor | Variable | Poor |
9.1 Key Distinguishing Features
The distinction between EPM and its primary differentials relies on integration of multiple clinical and laboratory parameters. Asymmetric presentation favors EPM over the typically symmetric deficits of CVCM and eNAD/EDM [2]. Young age and familial clustering suggest eNAD/EDM, while outbreak context points toward EHV-1 EHM [12]. Prominent muscle wasting with elevated CK and low vitamin E supports EMND rather than central neurological disease.
Lymphoma should be included as a differential diagnosis for horses presenting with acute ataxia lacking evidence of spinal cord compression, EPM, EHV-1 EHM, EDM, EMND, trauma, or other common causes [9]. This rare but important consideration emphasizes the breadth of differential diagnosis required for comprehensive evaluation.
---
10. Summary and Clinical Recommendations
10.1 Key Diagnostic Principles
- Combined testing improves accuracy: Paired serum and CSF antibody testing with serum:CSF ratio calculation provides superior diagnostic performance compared to either test alone [3].
- rtPCR lacks sensitivity: Real-time PCR on CSF demonstrates essentially 0% sensitivity for EPM diagnosis and should not be relied upon [4].
- Biomarkers complement serology: pNfH concentrations may assist in differentiating CVCM and eNAD/EDM from EPM when used alongside traditional testing [5].
- Treatment response informs diagnosis: Clinical improvement with antiprotozoal therapy supports EPM diagnosis retrospectively [1].
- Consider differential diagnoses systematically: CVCM, eNAD/EDM, EHV-1 EHM, and EMND represent primary differentials requiring active exclusion [9].
10.2 Future Directions
Research continues on antemortem biomarker and genetic testing for eNAD/EDM, which would have widespread clinical impact even if breed-specific [7]. Proteomic approaches using proximity extension assays and machine learning algorithms show promise for improved differentiation among causes of spinal ataxia [15]. The development of more sensitive molecular diagnostics for EPM remains an unmet need given the failure of current rtPCR methodologies.
Prevention strategies for vitamin E-associated conditions including eNAD/EDM and EMND emphasize providing pregnant mares and foals access to quality pasture, with supplementation of water-soluble RRR-α-tocopherol during gestation and the first two years of life as an alternative approach [7]. Baseline vitamin E measurement should precede supplementation to avoid potential oversupplementation-related coagulopathies.
References
References
Numbered in order of first citation, IEEE style. Each entry links to the original source via DOI or publisher URL.
- R. MacKay and D. K. Howe, “Equine Protozoal Myeloencephalitis.,” The Veterinary clinics of North America. Equine practice, Jul. 2022. doi: 10.1016/j.cveq.2022.05.003. View source
- S. Austin and S. Gray, “Equine protozoal myeloencephalitis—Presentation and progression,” Equine Veterinary Education, Oct. 2025. doi: 10.1111/eve.70016. View source
- P. Pandit, W. Smith, C. J. Finno, M. Aleman, P. A. Conrad, A. Packham, M. Plancarte, K. D. Woolard, A. E. Marsh, and N. Pusterla, “A fresh look at the SarcoFluor antibody test for the detection of specific antibodies to Sarcocystis neurona for the diagnosis of equine protozoal myeloencephalitis.,” Veterinary parasitology, Jun. 2024. doi: 10.1016/j.vetpar.2024.110219. View source
- C. K. Enriquez, J. Morrow, A. Graves, and A. L. Johnson, “Evaluation of real‐time polymerase chain reaction for the diagnosis of protozoal myeloencephalitis in horses using cerebrospinal fluid,” Journal of Veterinary Internal Medicine, Aug. 2023. doi: 10.1111/jvim.16826. View source
- L. A. Edwards, C. Donnelly, S. Reed, S. Valberg, M. Chigerwe, A. L. Johnson, and C. Finno, “Serum and cerebrospinal fluid phosphorylated neurofilament heavy protein concentrations in equine neurodegenerative diseases.,” Equine Veterinary Journal, May 2021. doi: 10.1111/evj.13452. View source
- B. R. Moore, S. Reed, D. Biller, C. Kohn, and S. Weisbrode, “Assessment of vertebral canal diameter and bony malformations of the cervical part of the spine in horses with cervical stenotic myelopathy.,” American Journal of Veterinary Research, Jan. 1994. doi: 10.2460/ajvr.1994.55.01.5. View source
- A. E. Young and C. J. Finno, “Current insights into equine neuroaxonal dystrophy/equine degenerative myeloencephalopathy.,” The Veterinary Journal, May 2024. doi: 10.1016/j.tvjl.2024.106129. View source
- B. D. F. Dallo, L. X. R. D. Silva, L. A. O. D. Paula, N. F. P. E. Silva, C. Smaniotto, G. M. Pagliosa, A. D. M. Viott, and M. D. C. S. Lopes, “Mieloencefalopatia Degenerativa Equina: Características Clínicas e Histopatológicas e Diagnósticos Diferenciais,” Acta Scientiae Veterinariae, Nov. 2023. doi: 10.22456/1679-9216.130299. View source
- K. MacMillan, J. Burns, E. John, and M. Buote, “Acute neurologic presentation of a 2‐year‐old standardbred colt with multicentric diffuse large B cell lymphoma,” Equine Veterinary Education, Jul. 2025. doi: 10.1111/eve.14204. View source
- M. Szklarz, A. Lipińska, M. Słowikowska, A. Niedźwiedź, K. Marycz, and M. Janeczek, “Comparison of the clinical and radiographic appearance of the cervical vertebrae with histological and anatomical findings in an eight-month old warmblood stallion suffering from cervical vertebral stenotic myelopathy (CVSM),” BMC Veterinary Research, Aug. 2019. doi: 10.1186/s12917-019-2047-x. View source
- M. Mannaa, A. Shamaa, A. Shawky, I. M. Hassan, A. Refaey, and A. Abu‐Seida, “A novel surgical technique for treatment of cervical vertebral stenotic myelopathy (wobbler syndrome) in a filly.,” Journal of Equine Veterinary Science, Apr. 2023. doi: 10.1016/j.jevs.2023.104493. View source
- J. D. Barbosa, A. Lins, H. A. Bomjardim, N. Silveira, C. C. Barbosa, E. A. Beuttemmuller, M. F. Brito, and F. M. Salvarani, “Equine Herpesvirus Type 1 Myeloencephalitis in the Brazilian Amazon,” Animals, Dec. 2022. doi: 10.3390/ani13010059. View source
- A. Velloso Álvarez, E. Jose-Cunilleras, A. Dorrego-Rodriguez, I. Santiago-Llorente, M. D. L. Cuesta-Torrado, L. Troya-Portillo, B. Rivera, V. Vitale, L. de Juan, and F. Cruz-Lopez, “Detection of equine herpesvirus-1 (EHV-1) in urine samples during outbreaks of equine herpesvirus myeloencephalopathy.,” Equine Veterinary Journal, Sep. 2023. doi: 10.1111/evj.14007. View source
- C. Holz, D. Sledge, M. Kiupel, R. Nelli, L. Goehring, and G. Soboll Hussey, “Histopathologic Findings Following Experimental Equine Herpesvirus 1 Infection of Horses,” Frontiers in Veterinary Science, Mar. 2019. doi: 10.3389/fvets.2019.00059. View source
- C. Donnelly, A. L. Johnson, S. Reed, and C. Finno, “Cerebrospinal fluid and serum proteomic profiles accurately distinguish neuroaxonal dystrophy from cervical vertebral compressive myelopathy in horses,” Journal of Veterinary Internal Medicine, Mar. 2023. doi: 10.1111/jvim.16660. View source
- F. Payette, J. Kulp, A. Graves, J. Janes, J. K. Morrow, K. Orr, S. Reed, R. Ruby, D. Stefanovski, and A. L. Johnson, “Commercial cytokine assay on equine cerebrospinal fluid does not distinguish equine degenerative myeloencephalopathy from cervical vertebral stenotic myelopathy.,” American Journal of Veterinary Research, Sep. 2025. doi: 10.2460/ajvr.25.06.0212. View source
- R. D. L. Rúa-Doménech, H. Mohammed, J. F. Cummings, T. Divers, A. Lahunta, and B. Summers, “Association between plasma vitamin E concentration and the risk of equine motor neuron disease.,” The Veterinary Journal, Nov. 1997. doi: 10.1016/S1090-0233(97)80021-4. View source
- J. Banfield, R. Lisak, A. Omar, W. Domingos, A. Fiaschitello, A. Morales-Gómez, T. Divers, and H. Mohammed, “Investigating the Risk of Equine Motor Neuron Disease in a Brazilian Stable and Successful Intervention.,” Journal of Equine Veterinary Science, Jun. 2019. doi: 10.1016/J.JEVS.2019.02.024. View source
- A. M. M. Gómez, S. Zhu, S. Palmer, E. Olsen, S. Ness, T. Divers, K. Bischoff, and H. Mohammed, “Analysis of neurofilament concentration in healthy adult horses and utility in the diagnosis of equine protozoal myeloencephalitis and equine motor neuron disease.,” Research in Veterinary Science, Aug. 2019. doi: 10.1016/j.rvsc.2019.04.018. View source
- H. Mohammed, T. Divers, B. Summers, and A. de Lahunta, “Vitamin E deficiency and risk of equine motor neuron disease,” Acta Veterinaria Scandinavica, Jul. 2007. doi: 10.1186/1751-0147-49-17. View source
- A. Firshman and S. Valberg, “Polysaccharide Storage Myopathy.,” The Veterinary clinics of North America. Equine practice, Jan. 2025. doi: 10.1016/j.cveq.2024.11.004. View source
- S. Valberg, Z. J. Williams, C. Finno, A. Schultz, D. Velez-Irizarry, M. Henry, K. Gardner, and J. Petersen, “Type 2 polysaccharide storage myopathy in Quarter Horses is a novel glycogen storage disease causing exertional rhabdomyolysis.,” Equine Veterinary Journal, Aug. 2022. doi: 10.1111/evj.13876. View source
- R. Stanley, M. E. McCue, S. Valberg, J. R. Mickelson, I. G. Mayhew, C. M. McGowan, C. Hahn, J. C. Patterson-Kane, and R. J. Piercy, “A glycogen synthase 1 mutation associated with equine polysaccharide storage myopathy and exertional rhabdomyolysis occurs in a variety of UK breeds.,” Equine Veterinary Journal, Jul. 2009. doi: 10.2746/042516409X407611. View source
- C. McGowan, T. McGowan, and J. Patterson-Kane, “Prevalence of equine polysaccharide storage myopathy and other myopathies in two equine populations in the United Kingdom.,” The Veterinary Journal, Jun. 2009. doi: 10.1016/j.tvjl.2008.01.008. View source
- S. L. Journée, H. Journee, W. Bergmann, I. Chantziaras, K. Vanderperren, E. Raes, S. Reed, C. D. de Bruijn, H. I. Berends, and C. Delesalle, “Evaluation of the diagnostic value of transcranial electrical stimulation (TES) to assess neuronal functional integrity in horses,” Frontiers in Neuroscience, Apr. 2024. doi: 10.3389/fnins.2024.1342803. View source
- A. Olszewska, E. Raes, L. Lefère, and K. Vanderperren, “Radiographic and computed tomographic features of a subarachnoid diverticulum causing compressive cervical myelopathy in a yearling Warmblood horse,” Equine Veterinary Education, Oct. 2023. doi: 10.1111/eve.13896. View source
- J. Rijckaert, B. Pardon, V. Saey, E. Raes, L. Van Ham, R. Ducatelle, G. van Loon, and P. Deprez, “Determination of magnetic motor evoked potential latency time cutoff values for detection of spinal cord dysfunction in horses,” Journal of Veterinary Internal Medicine, Sep. 2019. doi: 10.1111/jvim.15576. View source
This review is one of 34 in the Foxwatch research corpus. Methodology: PRISMA 2020-style systematic literature review against PubMed, CAB Abstracts, Scopus, Web of Science, and equine veterinary conference proceedings.