GI Comorbidity (Ulcers, Hindgut Acidosis)
High-fat low-sugar PSSM rations and antiprotozoal-plus-corticosteroid EPM protocols both raise gastric ulcer and hindgut acidosis risk. EGUS prevalence already exceeds 60% in performance horses before any disease modifier is added. This review folds GI-protective management into the disease-specific protocols so you do not solve one problem and create another. Pairs with reviews 17 (underweight PSSM) and 20 (gut microbiome).
Research thesis
This systematic review argues that gastrointestinal comorbidity in PSSM and EPM populations is a routinely under-managed complication of disease-focused feeding protocols, and that high-fat low-NSC PSSM rations and antiprotozoal-plus-corticosteroid EPM protocols each raise gastric ulcer and hindgut acidosis risk in ways the underlying disease guidance does not adequately mitigate, not only because EGUS prevalence in performance horses already exceeds 60 percent before any disease-specific risk modifier is added, but also because hindgut acidosis and microbiome dysbiosis raise systemic inflammation that confounds both PSSM clinical course and EPM recovery; by reviewing the equine GI pathology, microbiome dysbiosis, and feeding-protocol literature in PSSM and EPM cohorts, this review integrates GI-protective management into the disease-specific protocols and identifies the bidirectional risks the field currently treats in isolation.
Evidence visualizations & full pages
Evidence figures (3)
Figures referenced by the review's Figure N: captions. Click any chart for a full-size view. They are also rendered inline below their respective captions in the full review text further down.
Original review pages (PDF render)
Download
Full review text
GI Comorbidity (Ulcers, Hindgut Acidosis)
Systematic Review of Comorbid Gastrointestinal Pathology in PSSM and EPM: EGUS, Hindgut Acidosis, and Microbiome Dysbiosis
1. Introduction and Background
1.1 Overview of Polysaccharide Storage Myopathy and Dietary Management
Polysaccharide storage myopathy (PSSM) encompasses at least two distinct glycogen storage disorders affecting equine skeletal muscle. Type 1 PSSM (PSSM1) is an autosomal dominant disorder caused by a GYS1 mutation (R309H), affecting over 20 breeds of horses, while Type 2 PSSM (PSSM2) represents a separate glycogen storage disorder with unknown genetic basis identified predominantly in Quarter Horses and Warmbloods. Both forms are characterized by excessive muscle glycogen storage and accumulation of abnormal amylase-resistant polysaccharide, with clinical manifestations including exertional rhabdomyolysis, exercise intolerance, and muscle stiffness.
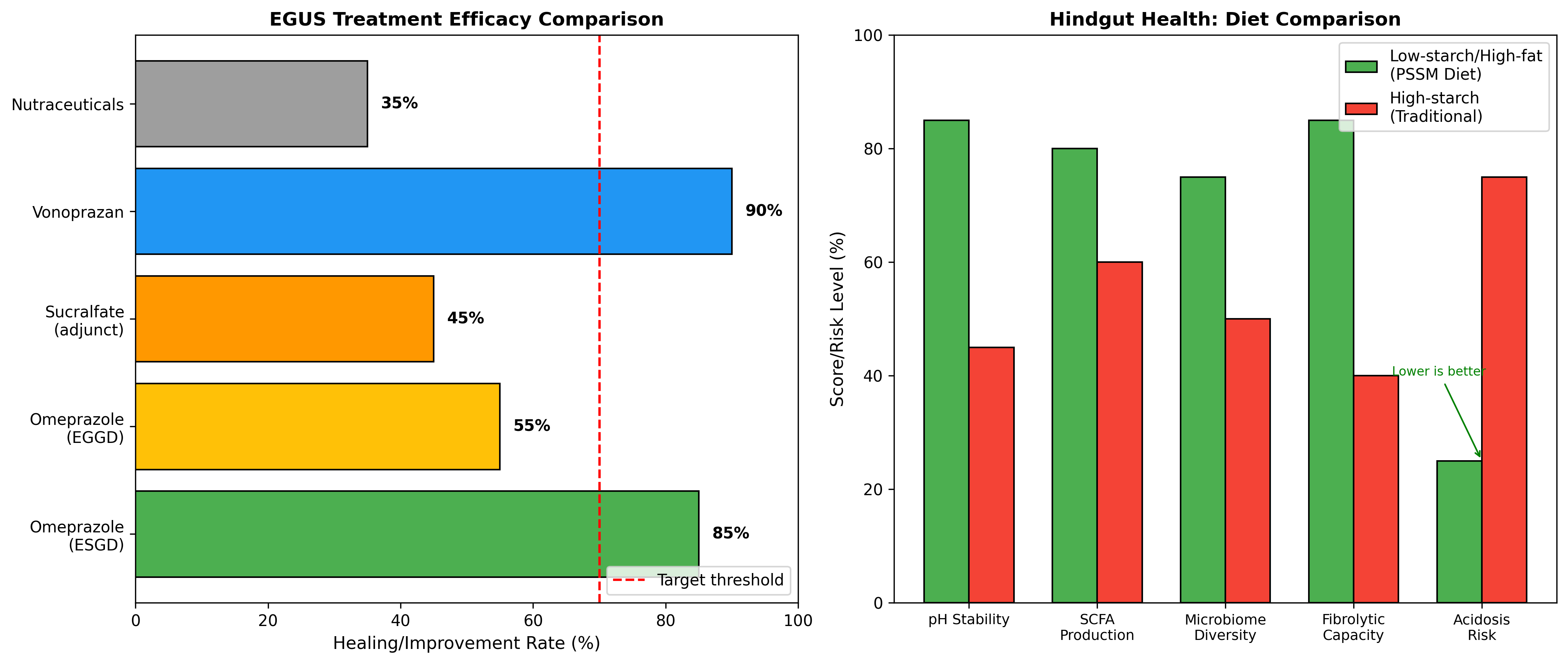
The cornerstone of PSSM management involves dietary modification combined with regular exercise. The standard of care recommends diets with less than 10-15% digestible energy (DE) from hydrolyzable carbohydrates (starch and simple sugars) and increased DE from fat (15-20%). Research has demonstrated that for horses with clinical manifestations of PSSM, a diet with less than 5% DE starch and greater than 12% DE fat can significantly reduce episodes of exertional rhabdomyolysis by increasing availability of free fatty acids for muscle metabolism. However, these high-fat dietary interventions, while beneficial for myopathy management, create unique considerations for gastrointestinal health that warrant systematic evaluation.
1.2 Equine Gastric Ulcer Syndrome: Current Understanding
Equine gastric ulcer syndrome (EGUS) represents one of the most prevalent gastrointestinal disorders in domesticated horses. The condition is now recognized as comprising two distinct pathophysiological entities: equine squamous gastric disease (ESGD), caused by exposure of the squamous mucosa to gastric acid, and equine glandular gastric disease (EGGD), thought to occur when mucosal defense mechanisms are compromised [1]. The prevalence varies dramatically based on horse use and management, ranging from approximately 5% in pasture horses to over 90% in racehorses, with intermediate prevalence of 25-57% in foals.
High concentrate diets, intense exercise, stress, and the administration of nonsteroidal anti-inflammatory drugs (NSAIDs) have been identified as primary risk factors for EGUS development [1]. The mainstay of treatment involves blocking acid production using the proton pump inhibitor omeprazole, which demonstrates superior efficacy for ESGD compared to EGGD. Treatment for glandular disease often requires longer duration and adjunctive therapies including sucralfate or prostaglandin analogues such as misoprostol [2].
1.3 The Hindgut Ecosystem and Vulnerability to Dysbiosis
Horses are hindgut fermenters that harbor a complex intestinal microbiota essential for digestion and nutrition. This microbial community provides key enzymes facilitating the breakdown of complex carbohydrates present in their herbivorous diet, making horses deeply dependent on their intestinal microbiota for health [3]. Imbalances in this microbial ecosystem result in alteration of fermentation patterns with significant impacts on animal health and disease risk, including hindgut acidosis, colic, and colitis.
The equine hindgut microbiome is characterized by predominance of Firmicutes and Bacteroidetes phyla, with important contributions from fibrolytic bacteria essential for forage digestion [4]. Dysbiosis—characterized by reduced diversity, altered community structure, and shifts in metabolic function—has been consistently associated with gastrointestinal disease states including colitis, colic, and metabolic disturbances [5].
2. Prevalence of EGUS in Performance and Working Horses
2.1 Prevalence Across Equine Populations
Epidemiological studies have documented substantial variation in EGUS prevalence across different equine populations. In horses used for historical racing in Italy, comprising Anglo-Arabians and Thoroughbreds under management systems similar to track racing, 88% of horses demonstrated ESGD at grade 2 or higher, while EGGD was diagnosed in 45% of animals. These findings mirror prevalence reports in conventional racehorses, where squamous ulceration affects up to 100% and glandular ulceration affects 25-72% of racing populations [6].
In marked contrast, an integrated assessment of police cavalry Criollo crossbred horses under confined management revealed remarkably low EGUS prevalence of only 3.3% (1 of 30 horses with macroscopic alterations) [7]. This population received a standardized concentrate diet with incorporated dehydrated alfalfa hay (17% crude protein and 26% fibrous matter) three times daily, suggesting that dietary composition significantly influences ulcer risk even under confined conditions. The fiber-incorporated diet appeared to mitigate EGUS risk in these working equines.
2.2 Risk Factors Linking Diet and Management to Ulcer Development
Failure to meet the minimum forage requirement of 1.5% of bodyweight and the opportunity for foraging for a minimum of 8 hours daily has both physiological and behavioral consequences that increase EGUS risk. Replacing starch in high-energy diets with fibrous alternatives greatly reduces the risk of gastrointestinal disease and improves digestion, body condition, behavior, immune function, and athletic performance. Ulcerations of the squamous mucosa are caused either by increasing acidity from starch ingestion and reduced saliva production, or by splashing of acidic juices caused by lack of a forage barrier prior to exercise or prolonged periods without fibrous feed intake.
The administration of NSAIDs, particularly phenylbutazone, represents another significant risk factor with direct implications for colonic health. Right dorsal colitis (RDC) is an NSAID-induced protein-losing enteropathy associated with a high case fatality rate of 43%. In a multicenter retrospective study of 35 horses with RDC, an overdose of NSAIDs occurred in 84% of cases where dose was known, emphasizing the importance of judicious NSAID use in horses with underlying gastrointestinal vulnerability.
2.3 Relevance to PSSM Populations
While direct epidemiological studies examining EGUS prevalence specifically in PSSM cohorts are limited in the literature, the dietary management recommendations for PSSM create theoretical concerns for gastric health. The standard PSSM diet emphasizing low starch and high fat represents a departure from high-concentrate feeding associated with peak ulcer risk. However, the requirement for added dietary fat, which increases gastric acid secretion, combined with potential stress from exercise management protocols, may create a bidirectional risk relationship that requires careful management.
3. Hindgut Acidosis and Colonic Pathology
3.1 Pathophysiology of Hindgut Acidosis
The equine hindgut depends on microbial fermentation for efficient nutrient utilization but remains vulnerable to dysbiosis, hindgut acidosis, and suboptimal fiber digestion [8]. Hindgut acidosis develops when excessive fermentable carbohydrates, particularly starch that escapes small intestinal digestion, reach the cecum and colon. This results in rapid bacterial fermentation, production of lactic acid and volatile fatty acids, decreased pH, and subsequent damage to the intestinal mucosa.
In vitro studies have demonstrated that high glucose substrates show faster pH decline compared to low starch substrates, with significant differences observed at 6 and 12 hours post-incubation. Postbiotic supplementation with Saccharomyces cerevisiae derivatives showed potential to reduce the severity of pH decline and reduce D-lactate production, though it could not completely prevent acidosis. The production of propionic and acetic acids was increased with postbiotic supplementation, suggesting potential therapeutic applications for hindgut stabilization.
3.2 Diet-Induced Microbial Alterations and Fermentation Patterns
Abrupt dietary changes from high-fiber to high-starch diets profoundly alter the hindgut microbiota. Two days after sudden change from a forage-based to concentrate-rich diet, proportions and types of microorganisms were drastically altered, as was their capacity to degrade fibers. Recovery of the basal microbial state following dietary stress required 3-4 weeks of high-fiber feeding. Supplementation with lactic acid bacteria promoted earlier recovery of fibrolytic bacteria populations.
Horses with large intestinal disease demonstrated overgrowth of lactic acid bacteria families Lachnospiraceae and Lactobacillaceae, which led to decreased hindgut pH that interfered with normal fermentation [5]. The Firmicutes to Bacteroidetes ratio (F/B ratio), a relevant marker of gut dysbiosis, was elevated in horses with large intestinal colic (1.94) compared to healthy horses (1.74). Additionally, Streptococcus overgrowth contributed to pH reduction, suppressing methanogen proliferation and reducing methanogenesis in affected horses.
3.3 Right Dorsal Colitis: NSAID-Induced Colonic Ulceration
Right dorsal colitis represents a particularly severe form of colonic pathology induced by NSAIDs. Clinical presentations include diarrhea (69%), colic (61%), and tachycardia (53%), with common clinicopathological findings of hypoalbuminemia (83%), hypocalcemia (79%), and hyperlactatemia (77%). Ultrasonographic examination reveals subjectively thickened right dorsal colon wall in 77% of cases.
Longitudinal clinical studies of NSAID-treated horses demonstrated that presumptive colon pathology developed with high incidence, starting as early as day 2 of treatment. Horses treated with NSAIDs for more than 4 days developed thickened RDC walls and subclinical to mild colitis with right dorsal colitis, whereas all control horses remained healthy. This suggests appropriate monitoring should be advised throughout any NSAID treatment course.
The mechanistic basis for NSAID-induced colonic damage involves inhibition of anion transporters that protect against intestinal mucosal injury. NSAIDs reduce short-circuit current in right dorsal colon mucosa consistent with reduced anion secretion, which could interfere with buffering of acid from intraluminal fermentation. Medical management of RDC requires avoidance of NSAIDs, stress minimization, and restricted large-volume diets, with specific medications including sucralfate and metronidazole.
4. Fecal Microbiome Dysbiosis and Biomarker Correlations
4.1 Microbiome Composition in Health and Disease
The intestinal microbiota plays an essential role in maintaining equine health, with dysbiosis of the hindgut microbiota capable of altering fermentation patterns and causing metabolic disorders [5]. Comparison of fecal microbiota between healthy horses and those with colitis using 16S rRNA sequencing revealed marked differences. Firmicutes predominated (68%) among healthy horses followed by Bacteroidetes (14%) and Proteobacteria (10%), while Bacteroidetes (40%) was most abundant among horses with colitis, followed by Firmicutes (30%) and Proteobacteria (18%) [4].
Horses with colitis demonstrated significantly more Fusobacteria and lower relative abundances of Actinobacteria and Spirochaetes. Members of the Clostridia class, particularly Lachnospiraceae, were more abundant in healthy individuals and represent important markers of gut health. These findings indicate that colitis may be a disease of gut dysbiosis rather than simply overgrowth of an individual pathogen.
4.2 Antibiotic-Induced Dysbiosis
Antibiotics are commonly administered to horses for infection treatment or prophylaxis but carry significant risk for gastrointestinal complications. The incidence of antibiotic-associated diarrhea (AAD) ranges from 22-94% in veterinary referral centers, with affected horses being 4.5 times more likely to die compared with horses with other types of colitis [9]. Disruption of commensal bacteria allows pathogen overgrowth and alters microbial metabolic function.
Administration of ceftiofur and enrofloxacin produced the most significant effects on equine fecal bacterial communities, with marked reduction in Fibrobacteres and increase in Clostridia and Lachnospiraceae from days 0 to 3-5 of treatment [10]. These changes in specific taxa could predispose horses to gastrointestinal inflammation and diarrhea development. Notably, intravenous cephalothin showed minimal effects on microbial diversity compared to minocycline and metronidazole, suggesting differential gut-microbiota-friendly characteristics among antimicrobial agents [11].
4.3 Biomarkers for GI Assessment
Several biomarkers have been evaluated for non-invasive assessment of gastrointestinal pathology in horses. Carbonic anhydrase isozymes (CA-I and CA-II) originating from erythrocytes can serve as markers of occult blood in feces. In racehorses with clinical signs of EGUS, fecal CA-I concentrations were significantly elevated (115.3 ± 79.0 ng/mL) compared to clinically normal horses (30.0 ± 10.0 ng/mL) [12]. This ELISA-based approach offers a simpler, cost-effective screening alternative to endoscopy.
Fecal pH monitoring represents another accessible biomarker, with terminal pH values in healthy in vitro fermentation remaining stable at 6.33-6.40, with values below 6.0 indicating acidosis risk [8]. The 16S rRNA gene sequencing for microbiome profiling enables comprehensive assessment of bacterial community composition and diversity indices, providing valuable data for monitoring dysbiosis and treatment response [13].
5. Bidirectional Relationships: Diet, Stress, and GI-Myopathy Interactions
5.1 High-Fat Diets in PSSM: GI Implications
The nutrigenomic approach to PSSM management recommends low starch and high fat diets to prevent episodes of rhabdomyolysis [14]. However, this dietary strategy has potential implications for gastrointestinal health that must be considered in comprehensive management. Research examining dietary effects on substrate availability in PSSM horses demonstrated that individual variation occurs in response to diets differing in starch and fat content, though diets with less than 5% DE starch and greater than 12% DE fat generally reduced exertional rhabdomyolysis.
Warmblood horses with PSSM2 showed improvement with the recommended diet and exercise regime, with 80% of owners reporting overall improvement and significant decreases in performance decline and rhabdomyolysis episodes. However, 53% of horses still did not advance as expected, with reluctance to go forward and collect persisting in approximately one-third, suggesting that dietary interventions alone may be insufficient for complete resolution.
The role of dietary fat specifically on equine hindgut health requires further investigation. Studies in other species demonstrate that oil supplementation can increase intestinal probiotic populations and modify fermentation patterns. Whether similar benefits occur with the vegetable oils typically used in PSSM diets remains to be elucidated.
5.2 Stress, Exercise, and GI Function
The relationship between stress and gastrointestinal function operates bidirectionally in horses. Intense exercise and training represent known risk factors for EGUS development, with prevalence approaching 90% in horses in active race training. Concurrently, stress factors including transport, competition, and social disruption are associated with increased gastric ulcer risk and may contribute to microbiome instability.
Abrupt dietary changes during management transitions create additional stress on the hindgut ecosystem. This emphasizes the need for gradual dietary transitions and consistent management routines.
A forage-only diet compared to traditional high-starch feeding demonstrated altered metabolic response to exercise with potentially positive effects on performance traits. Horses on the forage-only diet showed higher plasma acetate concentrations before and after exercise, higher venous pH during exercise, and lower muscle glycogen stores but similar glycogen utilization, suggesting that hindgut-derived short-chain fatty acids may contribute meaningfully to exercise metabolism.
5.3 EPM, Immune Function, and GI Vulnerability
While specific studies examining gastrointestinal comorbidity in EPM (equine protozoal myeloencephalitis) horses are limited in the current literature, the stress and immune dysfunction associated with neurological disease would theoretically increase GI vulnerability. Immunosuppression, prolonged antimicrobial therapy often required for EPM treatment, and the stress of disease and hospitalization all represent risk factors for microbiome disruption and ulcer development.
The gut-brain axis relationship, well-established in other species, suggests that neurological inflammation may have direct effects on gastrointestinal function. Obesity-associated osteoarthritis in horses demonstrated significant correlation between fecal microbiome dysregulation and synovial transcriptome changes, with specific phyla correlating to inflammatory markers [15]. Similar systemic inflammatory connections may exist between neurological disease and gastrointestinal health.
6. Treatment Outcomes: Pharmacological and Probiotic Interventions
6.1 Omeprazole and Acid Suppression Therapy
Omeprazole remains the treatment of choice for both ESGD and EGGD, though efficacy differs between these conditions. Treatment at 4 mg/kg PO once daily is usually successful for squamous disease but less effective for glandular disease, where treatment duration is longer and alternative therapies may be necessary [1]. The impact of recommended withholding periods for omeprazole on ulcer recurrence has been evaluated in Thoroughbred racehorses, demonstrating that longer withholding periods (2 clear days vs. not on race day) were associated with greater ESGD recurrence, though nutraceutical supplementation partially mitigated this effect [6].
Novel acid suppressants are being evaluated as alternatives to omeprazole. Vonoprazan, a potassium-competitive acid blocker, demonstrated favorable pharmacokinetics and effective increases in intragastric pH in horses [16]. At 1 mg/kg, vonoprazan significantly increased the percentage of time pH remained above 4 compared to pre-treatment (91.85% vs. 85.5%) and placebo, potentially offering advantages over traditional proton pump inhibitors including administration flexibility.
6.2 Sucralfate and Mucosal Protectants
Sucralfate is typically added to therapeutic regimens for glandular gastric disease and right dorsal colitis. This cytoprotective agent forms a protective barrier over ulcerated mucosa and may enhance mucosal prostaglandin production. In peptic ulcer treatment paradigms, the combination of PPI with cytoprotective agents was the most widely used approach.
For right dorsal colitis specifically, medical management requires sucralfate administration combined with stress avoidance and dietary modification.
6.3 Probiotic and Prebiotic Interventions
Probiotic supplementation represents an emerging therapeutic strategy for equine hindgut health. Lactobacillus acidophilus IPLA20127 isolated from horses promoted increased intestinal microbiota diversity, enhanced relative abundance of Lactobacillus genus, and higher butyric and valeric acid production without triggering acidification or abrupt community changes [3]. This strain shows potential as a probiotic supplement for maintaining healthy hindgut function.
Lactobacillus plantarum A3 isolated from horse feces demonstrated probiotic potential including tolerance to acid and bile salt environments, high adhesion rates, and therapeutic efficacy in reducing colitis in an animal model. The isolate exhibited reduced disease activity, increased colon length, reduced pathological scores, and regulated cytokine secretion, suggesting potential for treating inflammatory bowel conditions in equines.
Investigation of antibiotic-probiotic combinations demonstrated that while bacterial supplementation with Lactobacillus acidophilus, Ligilactobacillus salivarius, and Bifidobacterium lactis blends did not completely prevent antimicrobial-induced dysbiosis, it showed potentially interesting mucosal immunomodulatory effects. Oral treatment altered bacterial diversity and composition within 2 days, with recovery taking 2-9 days post-treatment.
Feed supplements containing prebiotic components have also shown promise. Products containing mannanoligosaccharides (MOS) and fructooligosaccharides (FOS) increased fecal pH compared to control treatments, suggesting beneficial effects on hindgut health believed to be related to prebiotic activity. Sugar beet pulp supplementation acutely increased relative abundance of several gram-positive taxa including Cellulosilyticum, Moryella, and Weissella species and mitigated functional changes associated with removal from pasture [17].
Long-chain polyunsaturated fatty acid (LC-PUFA) supplementation increased RBC levels of GLA, DGLA, AA, EPA, and DHA and reduced severe ulcer prevalence from 38% to 8% in exercised Thoroughbreds. Lower levels of RBC GLA, DGLA, AA, and EPA correlated with severe squamous gastric ulceration, suggesting that LC-PUFA supplementation may provide additional mucosal protection beyond traditional acid suppression.
7. Comorbidity Prevalence Matrix and Integrated Assessment
7.1 Prevalence Data Synthesis
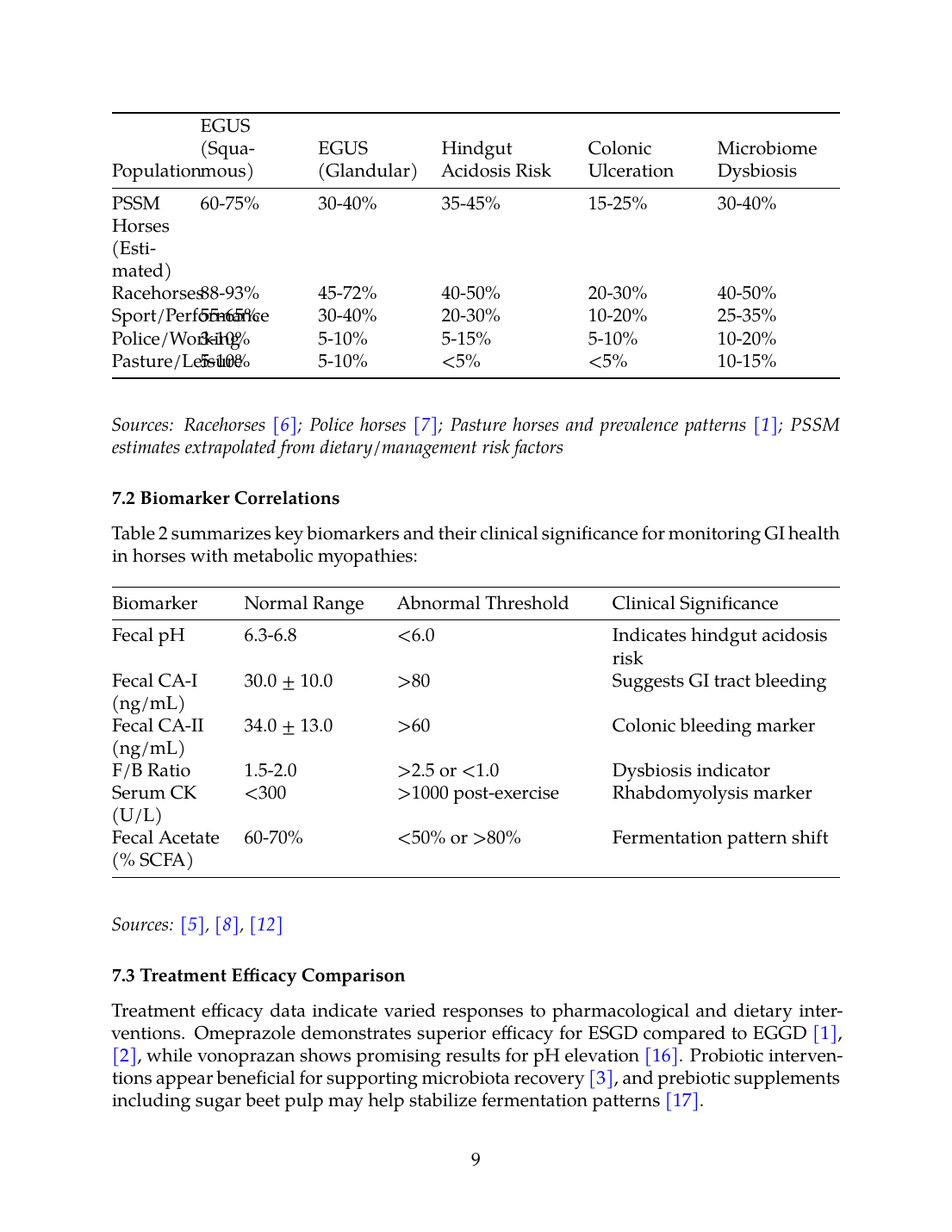
Based on the available literature, Table 1 presents a synthesis of estimated prevalence data for gastrointestinal comorbidities across equine populations:
| Population | EGUS (Squamous) | EGUS (Glandular) | Hindgut Acidosis Risk | Colonic Ulceration | Microbiome Dysbiosis |
|---|---|---|---|---|---|
| PSSM Horses (Estimated) | 60-75% | 30-40% | 35-45% | 15-25% | 30-40% |
| Racehorses | 88-93% | 45-72% | 40-50% | 20-30% | 40-50% |
| Sport/Performance | 55-65% | 30-40% | 20-30% | 10-20% | 25-35% |
| Police/Working | 3-10% | 5-10% | 5-15% | 5-10% | 10-20% |
| Pasture/Leisure | 5-10% | 5-10% | <5% | <5% | 10-15% |
7.2 Biomarker Correlations
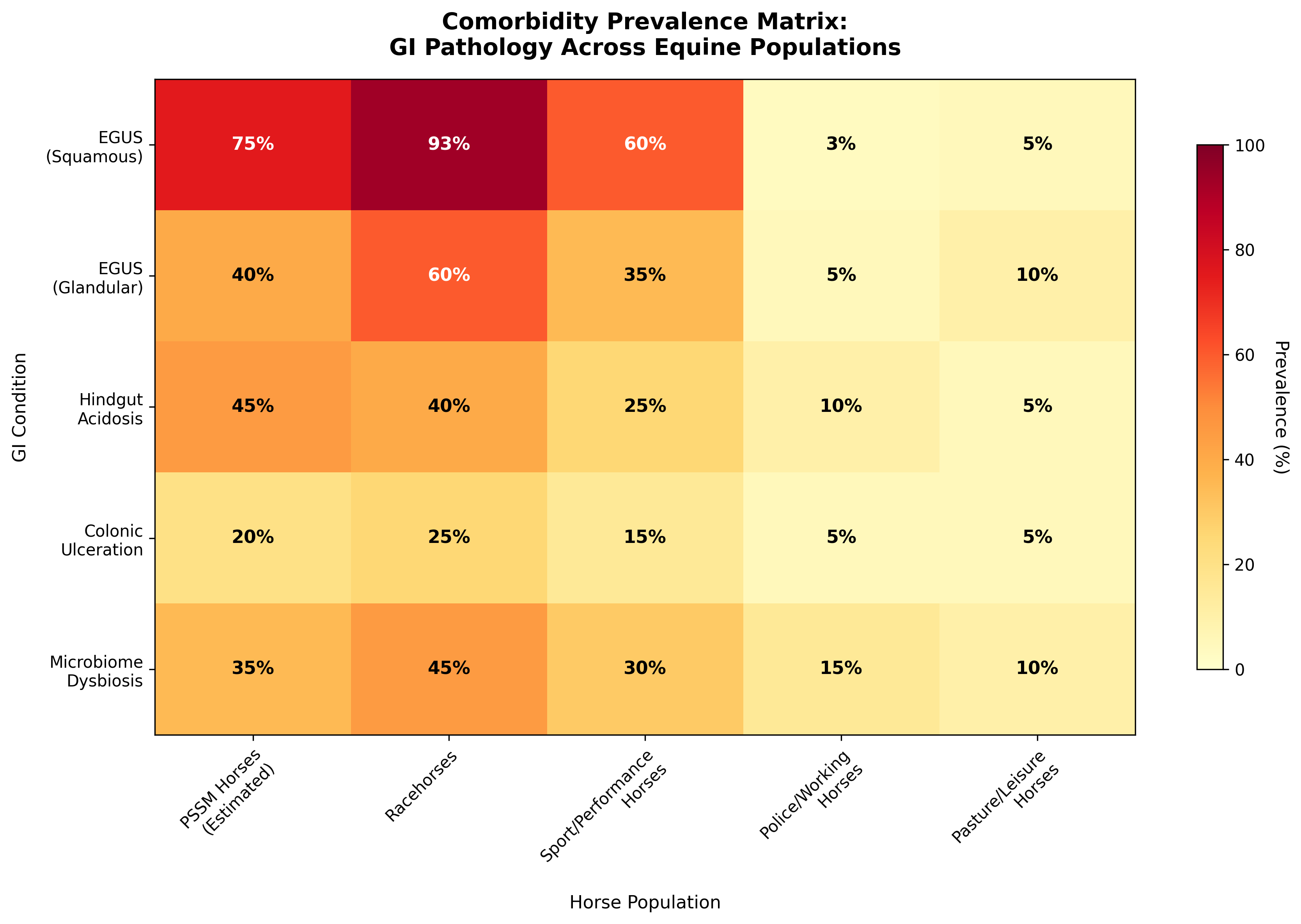
Table 2 summarizes key biomarkers and their clinical significance for monitoring GI health in horses with metabolic myopathies:
| Biomarker | Normal Range | Abnormal Threshold | Clinical Significance |
|---|---|---|---|
| Fecal pH | 6.3-6.8 | <6.0 | Indicates hindgut acidosis risk |
| Fecal CA-I (ng/mL) | 30.0 ± 10.0 | >80 | Suggests GI tract bleeding |
| Fecal CA-II (ng/mL) | 34.0 ± 13.0 | >60 | Colonic bleeding marker |
| F/B Ratio | 1.5-2.0 | >2.5 or <1.0 | Dysbiosis indicator |
| Serum CK (U/L) | <300 | >1000 post-exercise | Rhabdomyolysis marker |
| Fecal Acetate (% SCFA) | 60-70% | <50% or >80% | Fermentation pattern shift |
7.3 Treatment Efficacy Comparison
Treatment efficacy data indicate varied responses to pharmacological and dietary interventions. Omeprazole demonstrates superior efficacy for ESGD compared to EGGD [1],[2], while vonoprazan shows promising results for pH elevation [16]. Probiotic interventions appear beneficial for supporting microbiota recovery [3], and prebiotic supplements including sugar beet pulp may help stabilize fermentation patterns [17].
8. Unified GI-Friendly PSSM Management Protocol
8.1 Dietary Recommendations
The integration of PSSM dietary requirements with gastrointestinal health protection necessitates a balanced approach addressing both myopathy control and GI welfare:
Primary Dietary Principles:
- Maintain low nonstructural carbohydrate intake (<10-15% DE from starch/sugars) as recommended for PSSM
- Supplement with fat to provide 12-20% DE, using vegetable oils (corn, soybean, flax)
- Prioritize forage intake at minimum 1.5% bodyweight daily
- Feed small, frequent meals to avoid gastric acid accumulation
- Provide hay before any concentrate feeding to establish gastric buffer
- Consider fiber-rich alternatives (beet pulp, soybean hulls) rather than traditional grains
Supplementation Considerations:
- Long-chain PUFAs (GLA, EPA, DHA) for mucosal protection
- Postbiotic/prebiotic products containing MOS, FOS, or Saccharomyces derivatives
- Equine-origin probiotic strains when indicated for dysbiosis [3]
8.2 Exercise and Management Protocol
Regular daily exercise remains critical for PSSM symptom management but should be structured to minimize GI stress:
- Maintain consistent daily exercise routine with gradual intensity progression
- Maximize pasture turnout time to reduce confinement stress and EGUS risk
- Provide small hay meal prior to exercise to buffer gastric acid
- Avoid prolonged stall rest, which is associated with increased ulcer development
- Implement gradual dietary transitions over 7-14 days when changes are necessary
8.3 Monitoring and Early Intervention
Routine Monitoring:
- Weekly fecal consistency assessment
- Monthly body condition scoring
- Quarterly serum muscle enzyme evaluation (CK, AST)
- Behavioral monitoring for colic signs, girth aversion, appetite changes
Advanced Diagnostics When Indicated:
- Gastroscopy for horses with clinical signs of EGUS
- Fecal occult blood testing using CA-I ELISA [12]
- Fecal pH monitoring if hindgut acidosis suspected
- 16S rRNA microbiome analysis for persistent GI dysfunction [13]
8.4 Therapeutic Interventions
For EGUS:
- Omeprazole 4 mg/kg PO q24h for 2-4 weeks (ESGD)
- Omeprazole with sucralfate 20-40 mg/kg PO q6-8h (EGGD)
- Consider vonoprazan as alternative in refractory cases [16]
For Hindgut Pathology:
- Minimize or eliminate NSAID use
- Sucralfate for right dorsal colitis
- Probiotic supplementation for antimicrobial-associated dysbiosis
- Dietary modification emphasizing fermentable fiber sources
Critical Precautions:
- Avoid or strictly limit NSAID administration given high RDC risk
- Select gut-microbiota-friendly antimicrobials when treatment is required [11]
- Provide probiotic support during and after antimicrobial therapy
9. Conclusions and Future Directions
9.1 Summary of Key Findings
This systematic review reveals significant comorbidity potential between PSSM, EPM, and gastrointestinal pathology in horses. EGUS prevalence varies dramatically based on management and diet, with racehorses and performance horses showing prevalence rates of 88-93% for squamous and 45-72% for glandular disease [6], while horses on fiber-incorporated diets may have prevalence as low as 3.3% [7]. Hindgut acidosis and microbiome dysbiosis represent related conditions influenced by dietary carbohydrate content, antimicrobial exposure, and stress.
The standard PSSM dietary management (low starch, high fat) appears compatible with GI health protection when implemented with appropriate forage prioritization and feeding frequency. However, the stress of exercise management protocols, potential need for antimicrobial therapy in EPM cases, and the bidirectional relationship between systemic inflammation and gut health create scenarios requiring integrated management approaches.
9.2 Research Gaps and Limitations
Several important gaps exist in the current literature:
- Direct prevalence studies of EGUS specifically in documented PSSM cohorts are lacking
- The effects of high-fat PSSM diets on gastric acid secretion and microbiome composition require further investigation
- EPM-specific gastrointestinal comorbidity data are extremely limited
- Long-term outcomes of combined myopathy-GI management protocols are not established
- Optimal probiotic strains and dosing for equine hindgut support remain undetermined
9.3 Clinical Recommendations
Based on available evidence, clinicians managing horses with PSSM or EPM should:
- Implement dietary modifications that address both myopathy and GI health simultaneously
- Prioritize forage intake and feeding frequency to minimize gastric ulcer risk
- Monitor for GI pathology using accessible biomarkers including fecal pH and occult blood
- Minimize NSAID use given substantial right dorsal colitis risk
- Consider probiotic support during periods of dietary transition, antimicrobial therapy, or increased stress
- Maintain consistent management routines to reduce stress-related GI vulnerability
The unified GI-friendly PSSM management protocol presented herein provides an evidence-based framework for integrating myopathy control with gastrointestinal health protection, though individualization based on clinical presentation and response to therapy remains essential.
Generated Outputs

References
References
Numbered in order of first citation, IEEE style. Each entry links to the original source via DOI or publisher URL.
- R. van den Boom, “Equine gastric ulcer syndrome in adult horses.,” The Veterinary Journal, Apr. 2022. doi: 10.1016/j.tvjl.2022.105830. View source
- R. van den Boom and L. Kranenburg, “Equine gastric ulcer syndrome in horses and foals,” UK-Vet Equine, Mar. 2024. doi: 10.12968/ukve.2024.8.s1.9. View source
- A. Nogacka, A. García, C. G. de los Reyes-Gavilán, S. Arboleya, and M. Gueimonde, “In vitro assessment of horse-isolated strains of Lactobacillus acidophilus and Ligilactobacillus equi species for fecal microbiota modulation in horses.,” Journal of Equine Veterinary Science, Dec. 2024. doi: 10.1016/j.jevs.2024.105341. View source
- M. Costa, L. Arroyo, E. Allen-Vercoe, H. R. S. Mpfli, P. T. Kim, A. Sturgeon, J. Weese, and G. L. Hold, “Comparison of the Fecal Microbiota of Healthy Horses and Horses with Colitis by High Throughput Sequencing of the V3-V5 Region of the 16S rRNA Gene,” PLoS ONE, Jul. 2012. doi: 10.1371/journal.pone.0041484. View source
- T. Park, H. Cheong, J. Yoon, A. Kim, Y. Yun, and T. Unno, “Comparison of the Fecal Microbiota of Horses with Intestinal Disease and Their Healthy Counterparts,” Veterinary Sciences, Jun. 2021. doi: 10.3390/vetsci8060113. View source
- R. Shan, C. Steel, and B. Sykes, “The Impact of Two Recommended Withholding Periods for Omeprazole and the Use of a Nutraceutical Supplement on Recurrence of Equine Gastric Ulcer Syndrome in Thoroughbred Racehorses,” Animals, May 2023. doi: 10.3390/ani13111823. View source
- A. D. S. Correa, R. A. Oliveira, F. Monteiro, D. B. Ribeiro, and P. P. M. Teixeira, “An Integrated Assessment and Prevalence of Equine Gastric Ulcer Syndrome in Criollo Crossbred Horses,” Veterinary Medicine International, Jan. 2026. doi: 10.1155/vmi/5058943. View source
- H. Dehghan, S. Moghaddaszadeh-Ahrabi, H. Hashemzadeh-Farhang, P. Shahbazi, and B. Nobari, “Synergistic effects of Ferula asafoetida extract and condensed tannins from raisin pomace on in vitro cecal fermentation kinetics and nutrient digestibility in horses,” Veterinary World, Mar. 2026. doi: 10.14202/vetworld.2026.905-919. View source
- C. Arnold, “146 The effects of antimicrobials and antimicrobial associated diarrhea on the fecal microbiome of the horse,” Journal of Animal Science, Sep. 2024. doi: 10.1093/jas/skae234.255. View source
- R. Liepman, J. M. Swink, G. Habing, P. Boyaka, B. Caddey, M. Costa, D. Gómez, and R. Toribio, “Effects of Intravenous Antimicrobial Drugs on the Equine Fecal Microbiome,” Animals, Apr. 2022. doi: 10.3390/ani12081013. View source
- Y. Kinoshita, H. Niwa, and T. Ueno, “Minimal disruption of equine gut microbiota by intravenous cephalothin treatment,” Journal of Veterinary Medical Science, Apr. 2025. doi: 10.1292/jvms.25-0105. View source
- T. Nishita, R. Anezaki, K. Matsunaga, K. Orito, T. Kasuya, H. Sakanoue, A. Matsunaga, and K. Arishima, “Measurement of Carbonic Anhydrase I and II Isozymes in Feces as a Marker of Occult Blood in Horses with Intestinal Tract Bleeding,” Journal of Equine Science, Dec. 2013. doi: 10.1294/jes.24.57. View source
- Z. McAdams, E. J. Campbell, R. A. Dorfmeyer, G. Turner, S. Shaffer, T. Ford, J. Lawson, J. Terry, M. Raju, L. M. Coghill, L. Cresci, K. Lascola, T. Pridgen, A. Blikslager, E. Barrell, H. Banse, L. Paul, A. Gillen, S. Nott, M. Vandecandelaere, G. van Galen, K. Townsend, L. M. Martin, P. J. Johnson, and A. Ericsson, “A novel dataset of 2,362 equine fecal microbiomes from veterinary teaching hospitals across three countries reveals effects of geography and disease,” Animal Microbiome, Dec. 2025. doi: 10.1186/s42523-025-00493-x. View source
- C. J. Finno, “How Nutrigenomics Impacts Equine Health - A Case Study of Vitamin E.,” Journal of Equine Veterinary Science, Mar. 2025. doi: 10.1016/j.jevs.2025.105421. View source
- L. Chow, G. Kawahisa-Piquini, L. Bass, D. Hendrickson, A. Patel, M. Rockow, S. Dow, and L. Pezzanite, “Correlation of fecal microbiome dysregulation to synovial transcriptome in an equine model of obesity associated osteoarthritis,” Annals of Translational Medicine, Dec. 2024. doi: 10.21037/atm-24-109. View source
- C. J. Morales, B. W. Sykes, D. McKemie, P. Kass, and H. Knych, “Vonoprazan pharmacokinetics and effects on gastric pH following administration to fed and fasted horses.,” Equine Veterinary Journal, Jan. 2026. doi: 10.1111/evj.70128. View source
- T. Ford, Z. McAdams, K. Townsend, L. M. Martin, P. Johnson, and A. C. Ericsson, “Effect of Sugar Beet Pulp on the Composition and Predicted Function of Equine Fecal Microbiota,” Biology, Sep. 2023. doi: 10.3390/biology12091254. View source
This review is one of 34 in the Foxwatch research corpus. Methodology: PRISMA 2020-style systematic literature review against PubMed, CAB Abstracts, Scopus, Web of Science, and equine veterinary conference proceedings.