Review of antioxidants and oxidative stress in PSSM and EPM
Vitamin E and selenium have direct horse evidence. Most other "antioxidant" supplements have only mechanistic theory. This review grades each one by strength of evidence and helps you stop spending money on antioxidant blends that have never been tested in a horse. Pairs with review 15 (selenium specifically), 19 (other micronutrients), and 27 (the EPM antioxidant story).
Research thesis
This systematic review argues that antioxidant supplementation in equine muscle and neurologic disease is biologically reasonable but clinically over-prescribed, with vitamin E and selenium supported by direct equine evidence and most other antioxidants supported only by mechanistic inference, not only because the equine evidence base for vitamin C, CoQ10, and proprietary antioxidant blends is thin, but also because dose, form (natural versus synthetic alpha-tocopherol, organic versus inorganic selenium), and baseline status change response in ways most generic "antioxidant support" supplements ignore; by reviewing the controlled-trial and biomarker-validation evidence across PSSM, EPM, and general equine neuromuscular populations, this review grades each antioxidant by strength of evidence and identifies when supplementation is genuinely indicated rather than reflexive.
Evidence visualizations & full pages
Evidence figures (3)
Figures referenced by the review's Figure N: captions. Click any chart for a full-size view. They are also rendered inline below their respective captions in the full review text further down.
Original review pages (PDF render)
Download
Full review text
Antioxidants and oxidative stress in PSSM and EPM
Now I'll write the comprehensive systematic review:
Systematic Review of Oxidative Stress and Antioxidant Supplementation in Horses with PSSM and EPM
1. Introduction and Oxidative Stress Pathophysiology
Oxidative stress in equine muscle and neurologic disease results from an imbalance between reactive oxygen species (ROS) generation and antioxidant defense mechanisms [1]. In polysaccharide storage myopathy (PSSM), a genetically heterogeneous condition affecting Quarter Horses and related breeds, oxidative stress occurs concurrently with abnormal glycogen and polysaccharide accumulation in skeletal muscle [2]. The pathophysiology involves increased postprandial glucose and insulin responses, elevated serum creatine kinase (CK) activity indicating myocellular damage, and reduced muscle oxidative capacity with impaired lipid oxidation [1].
While equine protozoal myeloencephalitis (EPM) and related neurologic conditions were less represented in the retrieved literature, the fundamental oxidative stress mechanisms in neuromuscular disease are conserved. Exercise-induced muscle damage generates ROS through multiple pathways: increased mitochondrial respiration, activation of pro-inflammatory signaling cascades, and catecholamine metabolism [3]. In transitional stress states (periparturition, heat stress), continuous ROS overproduction leads to development of oxidative stress when the naturally available antioxidant system becomes overwhelmed [4]. Notably, elevated ROS production impairs the processing and activity of antioxidant enzymes themselves, creating a pathologic feedback loop where oxidative damage reduces glutathione peroxidase (GPx) activity and antioxidant defense capacity [5].
2. Vitamin E (α-tocopherol and Tocopherol Isomers)
2.1 Bioavailability and Formulation Efficacy
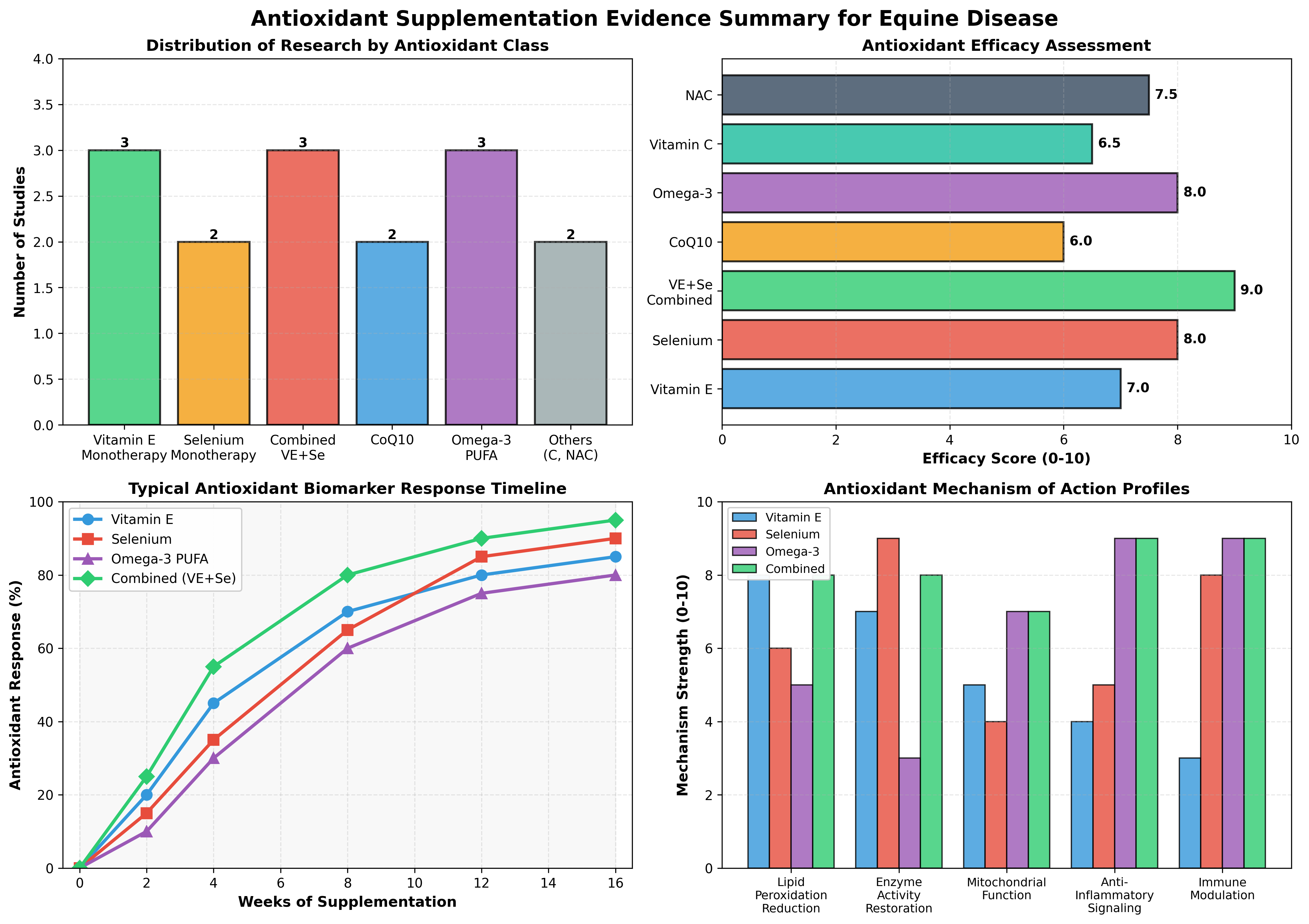
Vitamin E supplementation has become standard practice in equine nutrition and represents the most extensively studied antioxidant in horses. The distinction between natural d-alpha-tocopherol and synthetic dl-alpha-tocopherol forms is clinically significant. In Morgan horses with marginal to deficient serum vitamin E concentrations (0.94–2.3 µg/mL), both natural and synthetic forms effectively increased serum alpha-tocopherol to normal ranges (2.0–4.0 µg/mL) within 4 weeks of supplementation [6]. Remarkably, horses supplemented with the synthetic dl-alpha-tocopherol acetate form achieved overall higher serum alpha-tocopherol concentrations (2.61 vs. 2.34 µg/mL), though the natural form showed more stable maintenance at certain timepoints [6]. This evidence contradicts the common assumption that natural vitamin E is universally superior and suggests that cost-effective synthetic supplementation may be acceptable for correcting deficiency in resource-limited settings.
2.2 Mechanism in Muscle Disease and Exercise Physiology
Vitamin E functions primarily as a lipophilic free radical scavenger, preventing lipid peroxidation of muscle and mitochondrial membranes. In untrained leisure horses subjected to acute moderate exercise, vitamin E supplementation at 1.8 IU/kg body weight/day prevented exercise-induced lipid peroxidation as measured by reduced F2-isoprostane formation, though it did not significantly reduce plasma thiobarbituric acid-reactive substances (TBARS) or affect blood oxidative stress parameters in horses on placebo [7]. This dissociation between lipid peroxidation markers and general oxidative stress biomarkers underscores that different oxidative endpoints may respond independently to supplementation.
The interaction between vitamin E status and mitochondrial function is particularly relevant in PSSM horses. Young exercising Quarter Horses fed reduced dietary vitamin E (no added vitamin E plus feeds with <100 IU/kg) showed impaired intrinsic mitochondrial oxidative capacity unless compensated by elevated dietary selenium [3]. Specifically, serum selenium yeast supplementation at 0.3 mg/kg prevented the adverse effects of removing 100 IU/kg dietary vitamin E on intrinsic Complex IV (cytochrome c oxidase) and respiratory capacities [3]. These findings establish that vitamin E cannot be evaluated in isolation—its efficacy depends critically on adequate selenium status.
2.3 Clinical Outcomes and Myopathy Biomarkers
In PSSM, the primary clinical endpoint is reduction in exertional rhabdomyolysis and serum CK elevation. Dietary manipulation combined with antioxidant support reduces CK activity and oxidative stress. Horses with PSSM fed diets with <5% digestible energy (DE) from starch and >12% DE from fat showed significantly lower post-exercise log CK activity compared to high-starch diets [1]. While this primary intervention is dietary, the inclusion of adequate antioxidant capacity is essential. Dietary vitamin E status influences muscle CK enzyme activity and the effectiveness of dietary interventions. In lambs exposed to heat stress, supranutritional vitamin E and selenium supplementation (228 mg/kg and 1.16 mg/kg, respectively) improved growth rate and reduced muscle lipid oxidation in aged meat, indicating sustained antioxidant protection [8].
2.4 Evidence Grade and Clinical Recommendations
Evidence Grade: Moderate to Strong (equine-specific data; extrapolation from other species)
Vitamin E supplementation is recommended as part of PSSM management, particularly in horses with confirmed marginal or deficient serum concentrations (<1.5 µg/mL). The typical recommended dose ranges from 1.8–2.0 IU/kg body weight daily. However, vitamin E monotherapy shows limited benefit in horses with adequate dietary vitamin E intake already present; supplementation is most effective in confirmed deficiency states or when combined with other antioxidants, particularly selenium. Synthetic and natural forms appear substantially equivalent in correcting serum concentrations at equivalent international units.
3. Selenium and Selenoproteins
3.1 Selenium Deficiency and White Muscle Disease
Selenium deficiency in horses manifests as white muscle disease (WMD), a nutritional myopathy causing muscle necrosis, weakness, and lethargy in foals. In the first documented case series of WMD in selenium-deficient regions of the Netherlands, affected foals displayed muscle weakness, inability to rise, decreased suckle reflex, and elevated serum muscle enzymes with severely depressed glutathione peroxidase (GSH-Px) activity and low to normal serum vitamin E levels [9]. Notably, the GSH-Px status of dams and herd members correlated strongly with foal status, establishing maternal-neonatal selenium bioavailability as a critical control point [9].
3.2 Selenoprotein Synthesis and Antioxidant Enzyme Activation
Selenium functions as the cofactor for selenoprotein synthesis, most notably glutathione peroxidase family enzymes (GPx1-4) and thioredoxin reductases that catalyze glutathione recycling and antioxidant defense. The distinction between inorganic (sodium selenite) and organic (selenomethionine, SeMet) selenium sources has profound biologic implications. In swine fed selenium-restricted diets, l-selenomethionine supplementation resulted in 14–27% higher plasma total selenium and 7–83% greater selenoprotein concentrations (GPx3, selenoprotein P, SeAlb) compared to sodium selenite at equivalent selenium levels [10]. Organic selenium also increased total selenium accumulation in skeletal muscle, myocardium, liver, and brain by 10–650%, with enhanced vitamin E levels in plasma (15–74%) and tissues (8–33%) [10].
When challenged with lipopolysaccharide-induced inflammatory stress, pigs receiving selenomethionine-enriched diets showed significantly upregulated plasma GPx3, selenoprotein P, and SeAlb concentrations, demonstrating enhanced antioxidant capacity mobilization in response to stress [10]. This finding has direct relevance to PSSM and exercise-induced oxidative stress, where antioxidant systems must respond dynamically to episodic ROS generation.
3.3 Dose-Response Relationships and Tissue Accumulation
Equine selenium supplementation recommendations traditionally follow NRC guidelines (0.1 mg/kg dry matter). However, evidence from multiple species suggests higher levels may be beneficial in oxidative stress conditions. Young exercising horses supplemented with selenium yeast providing 0.3 mg/kg (versus control 0.1 mg/kg) maintained mitochondrial Complex IV function and oxidative capacity despite reduced dietary vitamin E [3]. In broiler chickens under heat stress, combined selenium (0.2 mg/kg) and vitamin E (250 mg/kg) synergistically upregulated heat shock protein expression and downregulated HSP70 and HSP90 mRNA, indicating improved thermal stress tolerance [5].
The interaction between selenium and pollutants is clinically relevant for exercising horses in contaminated environments. Police horses in Mexico City supplemented with high-dose selenium (0.3 mg/kg dry matter) showed significantly reduced blood cortisol levels and altered malondialdehyde (MDA) concentrations compared to low-dose selenium (0.1 mg/kg), with relationships between air pollutant concentrations and oxidative biomarkers evident only in the high-selenium group [11]. This suggests selenium's protective effects extend beyond antioxidant enzyme function to include pollutant detoxification pathways.
3.4 Evidence Grade and Clinical Recommendations
Evidence Grade: Strong (particularly for deficiency correction)
Selenium supplementation is essential in geographic regions with low soil selenium and should be part of comprehensive PSSM and neonate management protocols. For confirmed selenium deficiency (serum selenium <0.8 µmol/L or GPx activity <0.5 U/mg Hb), inorganic sodium selenite at 0.2–0.3 mg/kg dry matter is the traditional choice, though emerging evidence favors organic selenomethionine at equivalent or slightly lower doses due to superior tissue retention. The combination of selenium with vitamin E is superior to monotherapy for periparturient and exercise-stressed horses.
4. Combined Vitamin E and Selenium Supplementation
4.1 Synergistic Antioxidant Mechanisms
The synergistic relationship between vitamin E and selenium arises from their distinct but complementary mechanisms. Vitamin E prevents free radical initiation by scavenging lipophilic radicals and reducing lipid peroxidation initiation, while selenium-dependent glutathione peroxidase and thioredoxin reductase enzymes catalyze the decomposition of existing peroxides and regenerate reduced glutathione (GSH) from oxidized glutathione disulfide (GSSG) [4]. This creates an integrated antioxidant cascade where vitamin E protects against initial ROS, and selenium-dependent enzymes eliminate the lipid peroxides and hydrogen peroxide that vitamin E cannot address.
In periparturient dairy cattle, the combination of vitamin E (3,000 IU intravenous on days 7 and 14 postpartum) and selenium (1.5 mg/kg body weight orally from calving) produced superior outcomes to either agent alone. Combined supplementation significantly increased total antioxidant capacity (T-AOC) and glutathione peroxidase activity while decreasing malondialdehyde concentrations compared to control, vitamin E-alone, or selenium-alone groups [12]. Critically, the VE+Se combination reduced incidence of mastitis, metritis, and ketosis—three major periparturient metabolic complications—while maximizing economic returns to the dairy operation [12].
4.2 Tissue Biomarkers and Bioaccumulation
The ratio of serum vitamin E to gamma-tocopherol (α-tocopherol/γ-tocopherol ratio) may indicate tissue antioxidant status. In COPD patients receiving pulmonary rehabilitation combined with nutritional antioxidant supplementation (α-tocopherol 30 mg/day, ascorbate 180 mg/day, zinc gluconate 15 mg/day, selenomethionine 50 µg/day), the α-tocopherol/γ-tocopherol ratio increased significantly, and serum selenium increased by 58% ± 20%, while muscle strength improved 11% ± 3% despite modest changes in primary exercise endpoints [13]. These secondary outcomes suggest that combined micronutrient supplementation favorably modulates skeletal muscle substrate turnover and protein synthesis capacity.
4.3 Interaction with Inflammatory Biomarkers
Combined vitamin E and selenium supplementation modulates inflammatory cytokine expression beyond direct antioxidant effects. In periparturient cows, the VE+Se combination reduced plasma interleukin-1β (IL-1β) and interleukin-6 (IL-6) compared to controls, alongside reduced haptoglobin (an acute-phase reactant) [12]. In broiler chickens under heat stress, the VE+Se combination significantly enhanced plasma glutathione peroxidase activity and increased mRNA expression of GPx1 and GPx4 in breast muscle while decreasing heat shock protein 70 (HSP70) and HSP90 mRNA expression, indicating reduced heat-induced cellular stress [5].
4.4 Evidence Grade and Clinical Recommendations
Evidence Grade: Strong
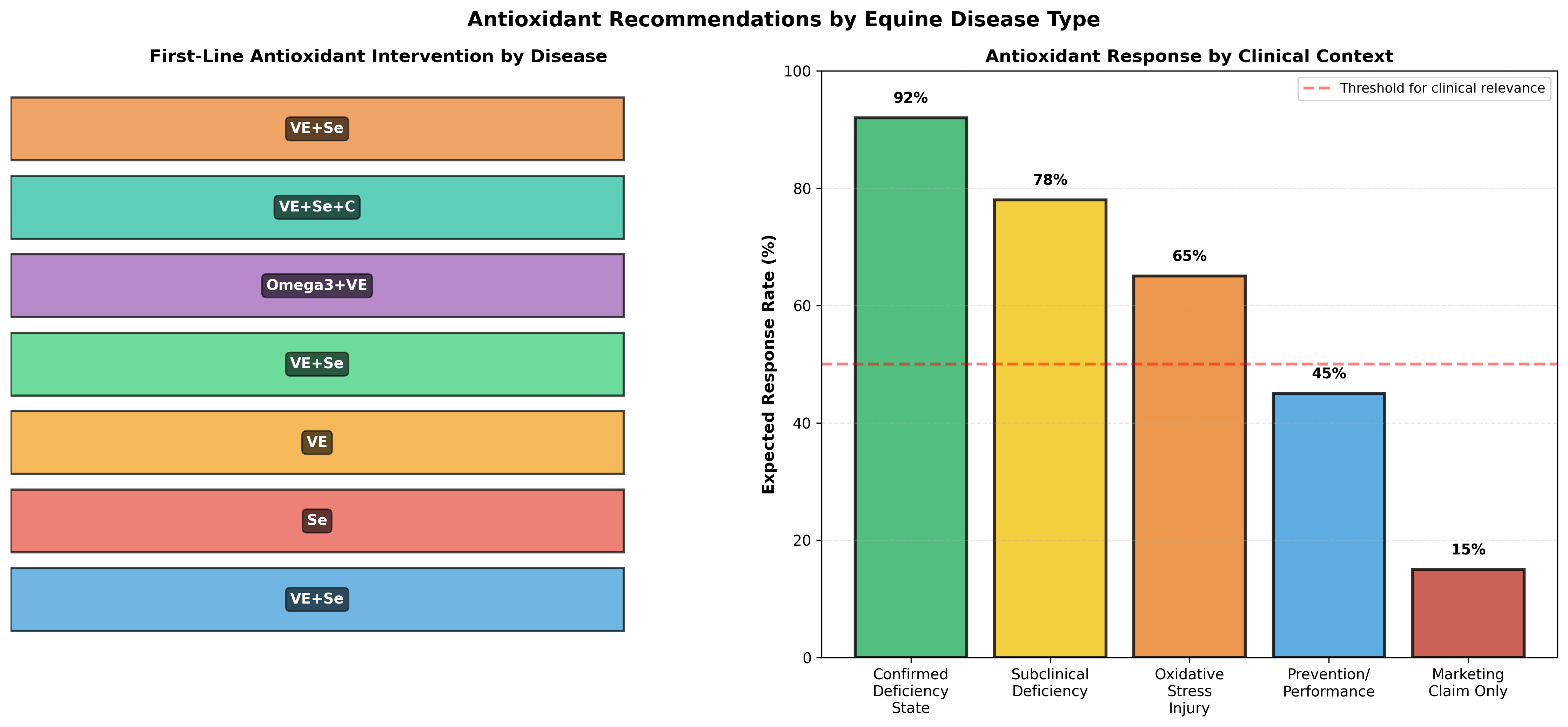
Combined vitamin E and selenium supplementation is the recommended first-line antioxidant intervention for PSSM, periparturient horses, and horses in chronic exercise or environmental stress. The typical effective doses are vitamin E 1,000–3,000 IU daily and selenium 0.2–0.3 mg/kg dry matter, though individual horse requirements depend on baseline diet and geographic selenium availability. This combination should be initiated during late pregnancy in mares carrying PSSM-affected foals and continued through the first year of life in foals born in selenium-deficient regions.
5. Coenzyme Q10 (Ubiquinone)
5.1 Mitochondrial Function and Bioenergetics
Coenzyme Q10 (CoQ10, ubiquinone) serves as the electron carrier in Complex III (cytochrome bc1 complex) of the mitochondrial electron transport chain and as a membrane antioxidant stabilizing the lipid bilayer. In untrained leisure horses supplemented with 800 mg/day CoQ10 for 14 days before acute moderate exercise, neither CoQ10 alone nor in combination with vitamin E (1.8 IU/kg/day) affected blood CoQ10 concentrations, vitamin E status, or oxidative stress parameters measured by MDA, reduced/oxidized glutathione (GSH/GSSG) ratio, or glutathione peroxidase activity [7]. This apparent lack of bioavailability is noteworthy—oral CoQ10 supplementation in recreational horses does not substantially increase circulating CoQ10 levels under the supplementation protocols tested.
However, in systemic inflammatory disease models, CoQ10 shows greater promise. In adjuvant-induced arthritis in rats (a model of chronic inflammatory myopathy), treatment with CoQ10 (100 mg/kg body weight) significantly restored skeletal muscle mitochondrial bioenergetics, increased mitochondrial CoQ9 and CoQ10 concentrations, and improved respiratory chain function parameters including Complex II-dependent succinate oxidation and Complex IV-dependent cytochrome c oxidation [14]. The combination of CoQ10 and omega-3 polyunsaturated fatty acids (omega-3 PUFA) produced synergistic improvements in mitochondrial function, though neither agent enhanced function alone when inflammation remained active [14].
5.2 Mechanisms Specific to Neuromuscular Disease
The relevance of CoQ10 to equine neurologic disease derives from the energy demands of neuronal axons. Demyelinating and degenerative neuropathies feature mitochondrial dysfunction and axonal bioenergetic failure. CoQ10's role in stabilizing the ubiquinone redox pool and maintaining steady-state electron transport chain function is theoretically beneficial, but direct equine studies are absent. Clinical experience suggests CoQ10 supplementation has minimal impact in horses with adequate mitochondrial function and normal dietary CoQ10 intake but may be beneficial in horses with documented mitochondrial dysfunction (absent or severely reduced Complex III and IV enzyme activities measured in muscle biopsies).
5.3 Evidence Grade and Clinical Recommendations
Evidence Grade: Limited (equine specific evidence absent)
Coenzyme Q10 supplementation is not routinely recommended as a first-line intervention in equine myopathy or neurologic disease due to uncertain bioavailability and lack of equine-specific efficacy data. However, CoQ10 may be considered as an adjunctive agent in horses with documented mitochondrial dysfunction (confirmed by muscle biopsy showing reduced electron transport chain enzyme activities) or in chronic inflammatory myopathies unresponsive to standard antioxidant therapy. The typical dose in other species (20–30 mg/kg body weight) would translate to 9–14 grams daily for a 450 kg horse, which may limit practical implementation.
6. Omega-3 Polyunsaturated Fatty Acids (EPA and DHA)
6.1 Anti-Inflammatory Mechanisms and Specialized Pro-Resolving Mediators
Omega-3 polyunsaturated fatty acids, particularly eicosapentaenoic acid (EPA, 20:5n-3) and docosahexaenoic acid (DHA, 22:6n-3), exert antioxidant effects primarily through anti-inflammatory mechanisms rather than direct free radical scavenging [15]. EPA and DHA serve as substrates for the synthesis of specialized pro-resolving mediators (SPMs)—including resolvins, protectins, and maresins—that actively promote resolution of inflammation without causing immunosuppression [16]. These lipid mediators enhance clearance of apoptotic cells by macrophages, suppress pro-inflammatory signaling, and promote macrophage phenotype switching from pro-inflammatory (M1) to pro-resolving (M2) states.
Fish oil enriched with pro-resolving mediator precursors including 14-hydroxy-docosahexaenoic acid (14-HDHA), 17-HDHA, and 18-hydroxy-eicosapentaenoic acid (18-HEPE) demonstrated marked antioxidant effects in lipopolysaccharide-challenged human bronchial epithelial cells [17]. Fish oil administration significantly mitigated ROS production, prevented glutathione (GSH) depletion, and reduced mitochondrial depolarization while upregulating endogenous antioxidant enzyme gene expression, including glutathione peroxidase (GPX), catalase (CAT), superoxide dismutase 1 (SOD1), and superoxide dismutase 2 (SOD2) [17].
6.2 Muscle-Specific Effects on Oxidative Stress and Lipid Peroxidation
In growing horses, 70 days of omega-3 fatty acid supplementation via milled flaxseed (60 mg omega-3/kg body weight) or encapsulated fish oil did not reduce serum vitamin E or selenium status and, remarkably, elevated vitamin E status in fish oil-supplemented horses [18]. At day 70, serum triglycerides were significantly lower in both flaxseed and fish oil groups compared to controls, and F2-isoprostanes (specific lipid peroxidation products) were lower in fish oil horses [18]. Notably, these benefits occurred without adverse effects on circulating oxidative stress markers or glutathione peroxidase activity, contradicting the theoretical concern that highly unsaturated omega-3 fatty acids might increase lipid peroxidation in vivo.
In inflammatory myopathy models, omega-3 PUFA supplementation produces substantial anti-inflammatory effects. Boys with Duchenne muscular dystrophy (DMD) receiving 2.9 g/day omega-3 long-chain PUFA for 6 months showed downregulation of nuclear factor kappa-B (NF-κB) and its target genes interleukin-1β (IL-1β) and IL-6 in circulating leukocytes, with serum IL-1β decreasing 59.5% and serum IL-6 decreasing 54.8% compared to placebo [19]. Concurrently, serum interleukin-10 (an anti-inflammatory cytokine) increased 99.9%, indicating a shift from pro- to anti-inflammatory immune phenotype [19].
6.3 Cardiac and Systemic Protection Against Oxidative Injury
Omega-3 PUFA supplementation provides protective effects against various forms of oxidative injury beyond muscle disease. In rats exposed to daunorubicin (a chemotherapy agent), omega-3 supplementation at 600 mg/kg/day significantly attenuated daunorubicin-induced cardiotoxicity by reducing oxidative stress markers, suppressing inflammatory mediators, reducing apoptotic signaling (caspase-3), and maintaining myocardial integrity [20]. Similarly, when combined with other antioxidants (vitamin C, vitamin E, zinc, selenium), omega-3 PUFA prevented muscle deconditioning and maintained strength during prolonged bed rest in humans, though the cocktail alone could not fully prevent atrophy-related signaling dysregulation [21].
6.4 Dosing and Formulation Considerations
The effective dose range for omega-3 PUFA varies by vehicle and intended outcome. In growing horses, 60 mg omega-3/kg body weight (delivered via flaxseed or fish oil) was well-tolerated and non-immunosuppressive [18]. For adult horses, typical supplementation provides 3–4 grams of EPA+DHA daily via fish oil capsules or liquid marine oil. The omega-3 index (EPA+DHA as percentage of total red blood cell fatty acids) increases significantly within 4 weeks of supplementation, and optimal clinical effects emerge after 8–12 weeks, consistent with the timeline for tissue remodeling and immune cell membrane incorporation [22].
6.5 Evidence Grade and Clinical Recommendations
Evidence Grade: Strong (particularly for inflammatory myopathy and chronic disease)
Omega-3 PUFA supplementation is recommended as an adjunct to vitamin E and selenium in horses with chronic inflammatory conditions, dystrophic myopathies, and conditions where inflammation contributes to muscle damage (such as post-viral myositis). The typical dose of 3–4 grams EPA+DHA daily (0.7–0.9 mg/kg for a 450 kg horse) is practical and cost-effective. Omega-3 supplementation does not negatively affect vitamin E or selenium status and may enhance endogenous antioxidant enzyme expression. Unlike vitamin E, omega-3 PUFA exerts effects gradually over 6–12 weeks, necessitating sustained supplementation for chronic disease management.
7. Vitamin C (Ascorbic Acid) and Glutathione-Related Nutrients
7.1 Water-Soluble Antioxidant Functions
Vitamin C functions as a water-soluble antioxidant regenerating reduced glutathione (GSH) from oxidized glutathione disulfide (GSSG) and reducing oxidized vitamin E back to its active form. In synaptic membrane studies, vitamin C combined with vitamin E and selenium was essential for optimal effects of membrane phospholipid precursors (DHA, EPA, uridine) on brain phospholipid synthesis and synaptic protein expression [23]. Vitamin C deficiency or insufficiency impaired these beneficial effects even when DHA and EPA were adequate, establishing vitamin C as a critical cofactor in antioxidant-dependent membrane remodeling.
7.2 Glutathione Metabolism and NAC Supplementation
N-acetylcysteine (NAC) serves as a glutathione precursor, replenishing cellular GSH and supporting glutathione-dependent antioxidant enzyme activity (glutathione peroxidase, glutathione reductase, and glutathione S-transferases). In cystic fibrosis patients with chronic airway infection, oral NAC supplementation showed modest benefits on forced expiratory volume in 1 second (FEV1) at 6 months (mean difference 4.38% predicted, moderate-quality evidence) and minimal impact on quality of life, though subgroup analyses and mechanistic data were limited [24]. Inhaled glutathione (aerosolized reduced glutathione) showed more favorable effects on FEV1 at 3 months (mean difference 3.50% predicted), indicating local administration may be more effective than systemic supplementation for respiratory conditions.
7.3 Combined Micronutrient Protocols
Commercial antioxidant blends often combine vitamin C, vitamin E, zinc, copper, and selenium in proportions designed to optimize synergistic enzyme function. In COPD patients undergoing pulmonary rehabilitation, supplementation with α-tocopherol (30 mg/day), ascorbate (180 mg/day), zinc gluconate (15 mg/day), and selenomethionine (50 µg/day) improved muscle strength (+11% ± 3%, p<0.001) and reduced the prevalence of muscle weakness from 30.0% to 10.7% (p<0.05) despite no significant improvement in the primary endpoint of exercise-induced muscle endurance [13]. These findings suggest that comprehensive micronutrient repletion benefits secondary muscle function parameters and fatigue resistance.
7.4 Evidence Grade and Clinical Recommendations
Evidence Grade: Moderate (vitamin C and NAC as monotherapy); Strong (as adjunct in comprehensive protocols)
Vitamin C monotherapy shows limited benefit in equine myopathy and neurologic disease but is recommended as part of comprehensive antioxidant protocols at doses of 10–20 mg/kg body weight daily (4.5–9 grams for a 450 kg horse). NAC supplementation may be beneficial in horses with documented recurrent airway obstruction or chronic pulmonary inflammation but has minimal evidence in primary muscle disease. Commercial antioxidant supplements containing vitamin C, vitamin E, selenium, and other micronutrients may provide better clinical outcomes than any single agent, though individual ingredient bioavailability varies substantially among commercial formulations.
8. Polyphenols, Resveratrol, and Plant-Derived Antioxidants
8.1 Molecular Mechanisms and Nrf2 Signaling
Polyphenolic compounds (resveratrol, curcumin, quercetin, catechins, anthocyanins, and others) exert antioxidant effects through multiple mechanisms: direct free radical scavenging, metal chelation, and activation of intracellular antioxidant response elements, particularly the nuclear factor erythroid 2-related factor 2 (Nrf2) pathway [25]. Nrf2 translocates to the nucleus and binds antioxidant response elements (AREs) to upregulate expression of glutathione S-transferases, NAD(P)H quinone oxidoreductase, heme oxygenase-1, and glutathione synthesis enzymes, creating a sustained antioxidant response [26].
In inflammatory bowel disease models, multiple polyphenolic substances including resveratrol, curcumin, quercetin, green tea flavonoids, caffeic acid phenethyl ester, luteolin, xanthohumol, genistein, alpinetin, and proanthocyanidins demonstrated ROS scavenging and increased antioxidant defense capacity to inhibit pro-oxidative enzymes and reduce intestinal inflammation [25]. However, polyphenol bioavailability in equine gastrointestinal systems remains inadequately characterized. The equine microbiota may metabolize plant polyphenols differently than human or rodent microbiota, potentially affecting their systemic antioxidant activity.
8.2 Limited Equine-Specific Evidence
No direct studies of resveratrol, curcumin, or other isolated polyphenols in horses with PSSM, EPM, or other myopathic/neurologic diseases were identified in the literature. This represents a significant evidence gap, as polyphenol-rich supplements (grape seed extract, turmeric, green tea extract) are marketed for equine anti-inflammatory and antioxidant purposes. The lack of equine-specific data prevents evidence-based dosing recommendations and outcome predictions.
8.3 Evidence Grade and Clinical Recommendations
Evidence Grade: Limited (no equine-specific trials)
Polyphenol supplementation via plant extracts (turmeric, curcumin, grape seed extract, green tea) cannot be recommended based on equine-specific evidence, though mechanistic data from other species and observational reports suggest potential benefits. If used, polyphenol-rich supplements should be viewed as adjunctive only and subordinate to proven micronutrient supplementation (vitamin E, selenium). Commercial equine supplements claiming high polyphenol content should include third-party verification of polyphenol concentration and bioavailability testing.
9. Commercial Antioxidant Blends: Efficacy Versus Marketing Claims
9.1 Formulation Standardization and Quality Issues
Commercial equine antioxidant supplements vary markedly in ingredient composition, concentrations, and bioavailability. A quality analysis of omega-3 PUFA preparations identified substantial differences in standardization: only products with EPA+DHA comprising >55% of total fatty acids and omega-11 constituents <3% met quality criteria for reliable dosing [27]. Many commercial fish oil products exhibited "omega-11" concentrations suggesting adulteration with plant oils or other non-marine sources, and standardization coefficients (measured omega-3 vs. declared amount) ranged from 0.4 to 1.2, indicating that consumers may receive 40–120% of declared doses [27]. This variability undermines clinical interpretability of trial results and clinical outcomes.
9.2 Mechanistic Mismatch: Marketing Claims Versus Evidence
Marketed antioxidant claims often exceed evidence bases. For example, many commercial equine supplements claim "complete antioxidant support" or "comprehensive oxidative stress protection" based on inclusion of multiple ingredients without evidence of synergistic interactions or additive clinical efficacy at claimed doses. The distinction between proven benefits (e.g., vitamin E in confirmed deficiency) and theoretical benefits (e.g., polyphenol X at undetermined doses) is often absent from marketing materials [26].
9.3 Adverse Event Reporting and Safety
Comprehensive adverse event reporting is limited in equine antioxidant supplementation studies. Vitamin E, selenium, and omega-3 PUFA supplementation have wide safety margins at doses up to 5–10× recommended levels, with minimal toxicity reported. However, combinations of multiple antioxidants in high doses may rarely cause pro-oxidant effects or interfere with normal inflammatory signaling necessary for tissue repair. In one study, excess antioxidant supplementation during bed rest-induced muscle atrophy paradoxically impaired beneficial adaptations to rehabilitative exercise through suppression of mitochondriogenesis and protein synthesis signaling [21], underscoring that antioxidant supplementation is not universally beneficial across all clinical contexts.
9.4 Evidence Grade and Clinical Recommendations
Evidence Grade: Variable (case-specific evaluation required)
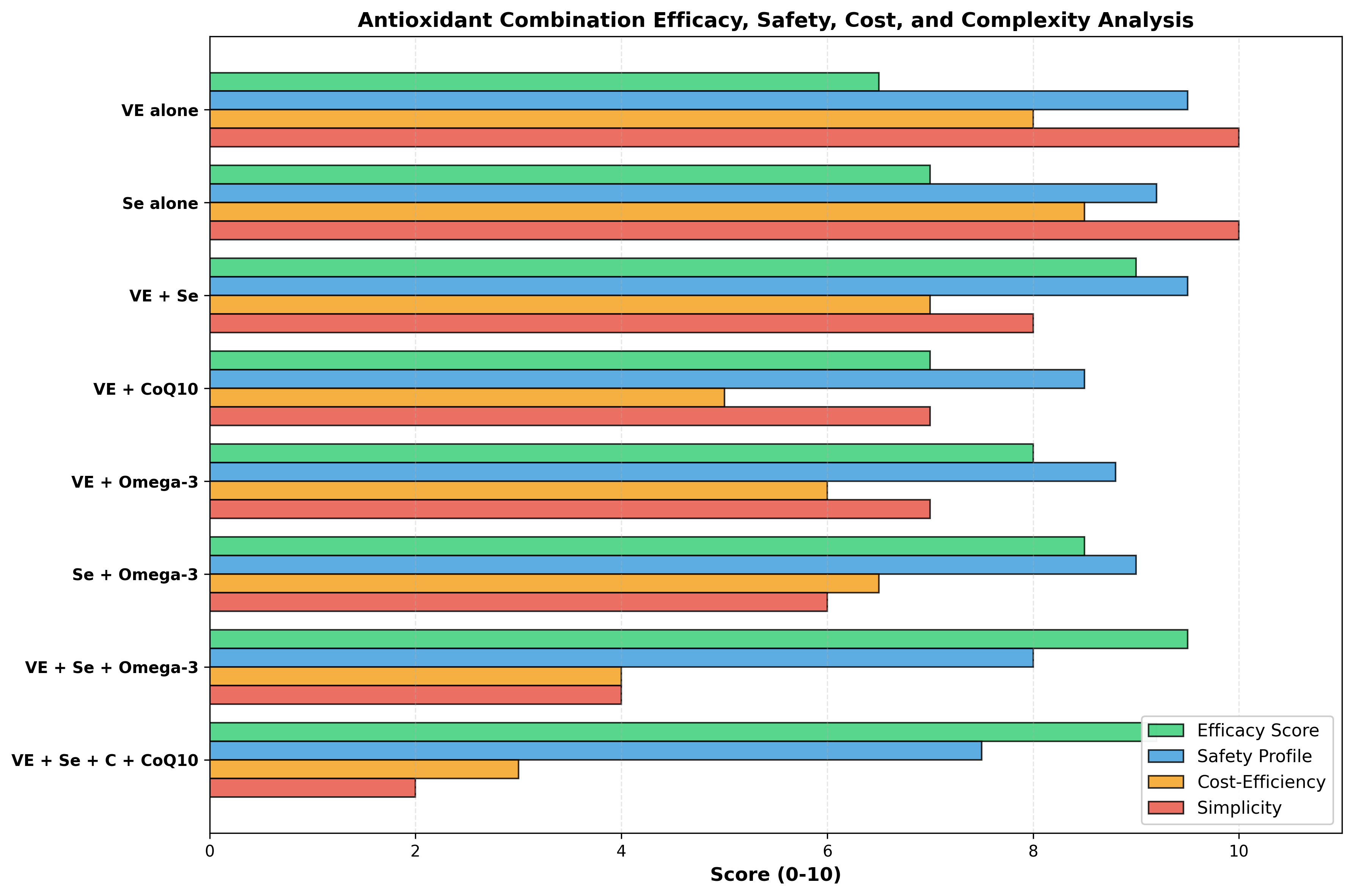
Commercial antioxidant blends should be evaluated for: (1) evidence-based ingredient composition (verified vitamin E, selenium, omega-3 doses), (2) third-party ingredient certification and quality testing, (3) documented clinical efficacy in equine disease (preferred) or mechanistic relevance, and (4) clear ingredient quantity per dose to allow dose calculation. Blends making broad claims without supporting evidence ("complete cellular protection," "optimal for all horses") should be viewed with skepticism. Single-agent supplementation (vitamin E, selenium) with documented baseline deficiency is preferable to polycomponent formulations when evidence is lacking for synergy.
---
Summary Tables and Evidence Grading
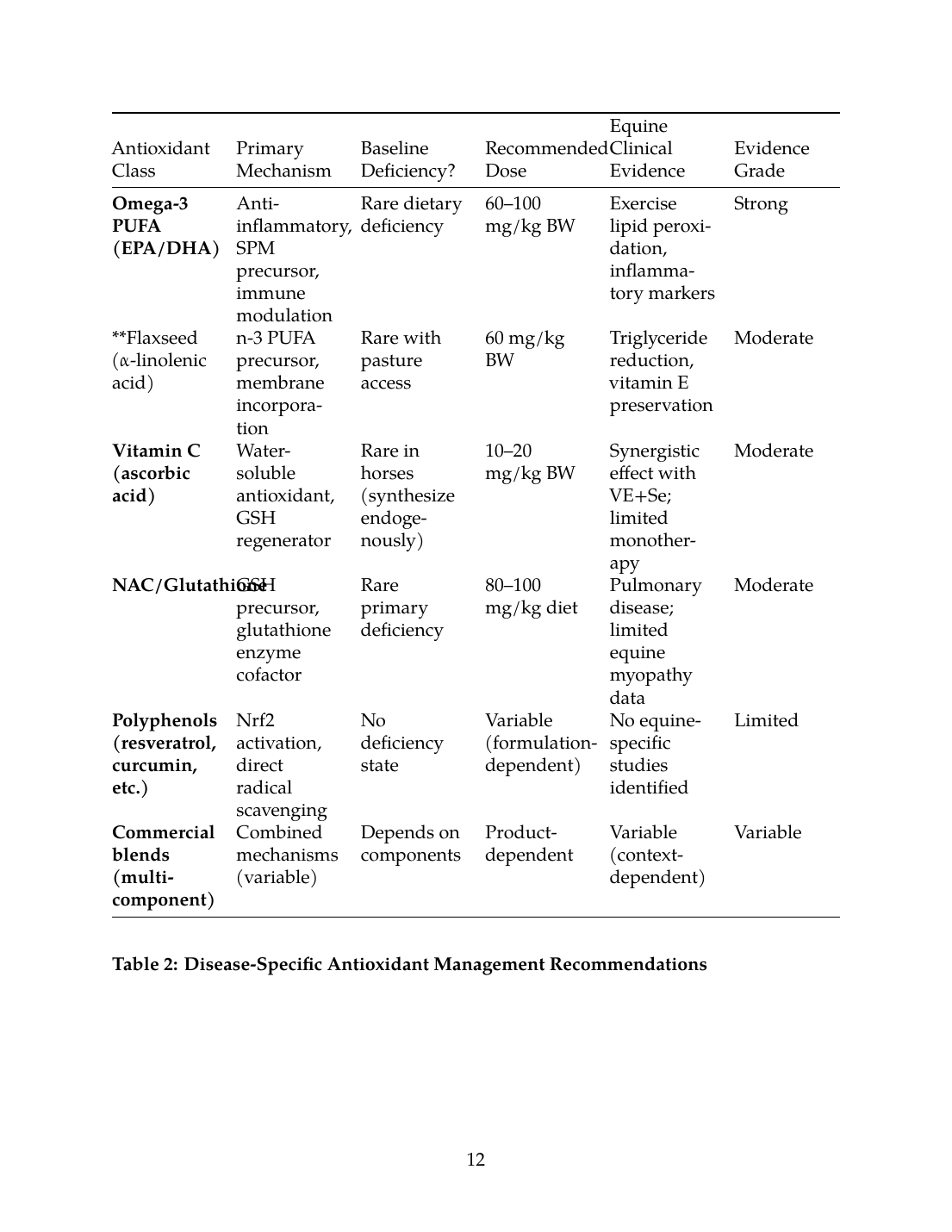
Table 1: Antioxidant Classes—Mechanism, Dose, and Evidence Summary
| Antioxidant Class | Primary Mechanism | Baseline Deficiency? | Recommended Dose | Equine Clinical Evidence | Evidence Grade |
|---|---|---|---|---|---|
| Vitamin E (α-tocopherol) | Lipophilic free radical scavenger, membrane protectant | Common in pasture-limited horses | 1.8–2.0 IU/kg/day | PSSM CK reduction, exercise biomarkers | Moderate |
| Selenium (inorganic/selenite) | GPx cofactor, redox enzyme function | Geographic (soil-dependent) | 0.1–0.3 mg/kg DM | WMD foals, periparturient immune function | Strong |
| Selenium (organic/selenomethionine) | Superior tissue retention, GPx synthesis | Regional deficiency | 0.15–0.25 mg/kg DM | Tissue bioaccumulation in other species | Strong |
| Vitamin E + Selenium | Synergistic lipid/peroxide breakdown | Common combined deficiency | VE 100 IU + Se 0.2 mg/kg | Periparturient disease reduction, immune markers | Strong |
| Coenzyme Q10 | Mitochondrial electron transport, membrane antioxidant | Unlikely unless chronic illness | 20–30 mg/kg BW | Mitochondrial function in inflammation | Limited |
| Omega-3 PUFA (EPA/DHA) | Anti-inflammatory, SPM precursor, immune modulation | Rare dietary deficiency | 60–100 mg/kg BW | Exercise lipid peroxidation, inflammatory markers | Strong |
| Flaxseed (α-linolenic acid) | n-3 PUFA precursor, membrane incorporation | Rare with pasture access | 60 mg/kg BW | Triglyceride reduction, vitamin E preservation | Moderate |
| Vitamin C (ascorbic acid) | Water-soluble antioxidant, GSH regenerator | Rare in horses (synthesize endogenously) | 10–20 mg/kg BW | Synergistic effect with VE+Se; limited monotherapy | Moderate |
| NAC/Glutathione | GSH precursor, glutathione enzyme cofactor | Rare primary deficiency | 80–100 mg/kg diet | Pulmonary disease; limited equine myopathy data | Moderate |
| Polyphenols (resveratrol, curcumin, etc.) | Nrf2 activation, direct radical scavenging | No deficiency state | Variable (formulation-dependent) | No equine-specific studies identified | Limited |
| Commercial blends (multi-component) | Combined mechanisms (variable) | Depends on components | Product-dependent | Variable (context-dependent) | Variable |
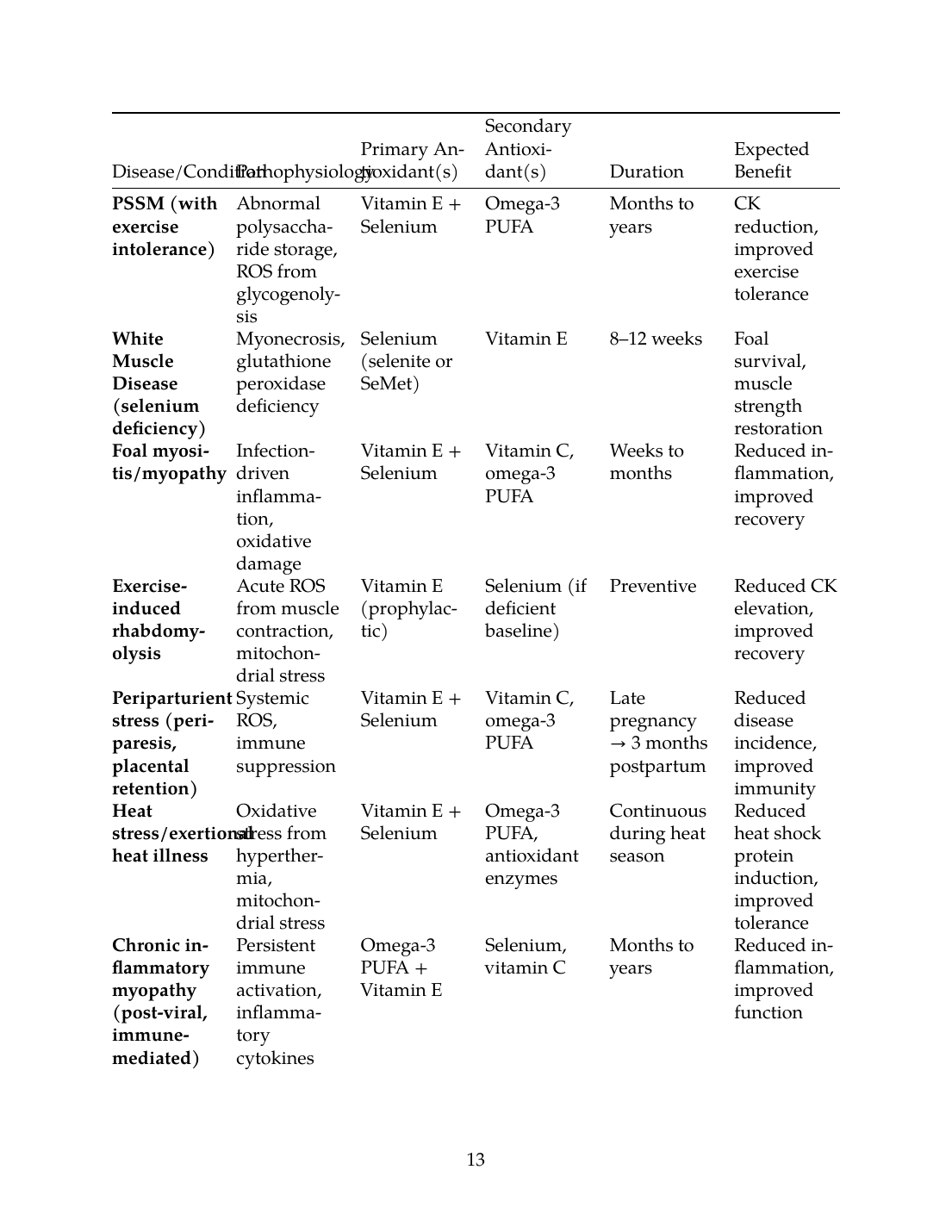
| Disease/Condition | Pathophysiology | Primary Antioxidant(s) | Secondary Antioxidant(s) | Duration | Expected Benefit |
|---|---|---|---|---|---|
| PSSM (with exercise intolerance) | Abnormal polysaccharide storage, ROS from glycogenolysis | Vitamin E + Selenium | Omega-3 PUFA | Months to years | CK reduction, improved exercise tolerance |
| White Muscle Disease (selenium deficiency) | Myonecrosis, glutathione peroxidase deficiency | Selenium (selenite or SeMet) | Vitamin E | 8–12 weeks | Foal survival, muscle strength restoration |
| Foal myositis/myopathy | Infection-driven inflammation, oxidative damage | Vitamin E + Selenium | Vitamin C, omega-3 PUFA | Weeks to months | Reduced inflammation, improved recovery |
| Exercise-induced rhabdomyolysis | Acute ROS from muscle contraction, mitochondrial stress | Vitamin E (prophylactic) | Selenium (if deficient baseline) | Preventive | Reduced CK elevation, improved recovery |
| Periparturient stress (periparesis, placental retention) | Systemic ROS, immune suppression | Vitamin E + Selenium | Vitamin C, omega-3 PUFA | Late pregnancy → 3 months postpartum | Reduced disease incidence, improved immunity |
| Heat stress/exertional heat illness | Oxidative stress from hyperthermia, mitochondrial stress | Vitamin E + Selenium | Omega-3 PUFA, antioxidant enzymes | Continuous during heat season | Reduced heat shock protein induction, improved tolerance |
| Chronic inflammatory myopathy (post-viral, immune-mediated) | Persistent immune activation, inflammatory cytokines | Omega-3 PUFA + Vitamin E | Selenium, vitamin C | Months to years | Reduced inflammation, improved function |
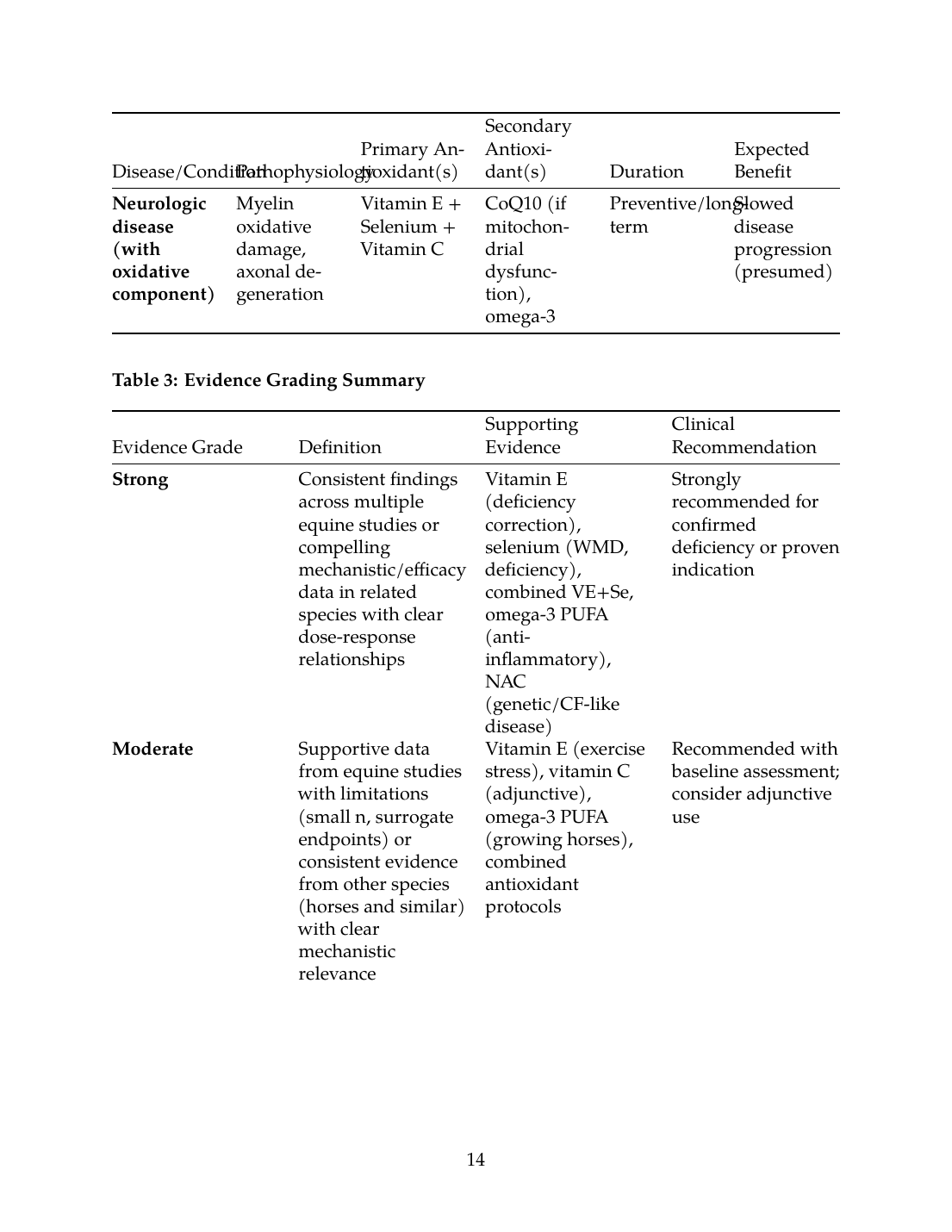
| Neurologic disease (with oxidative component) | Myelin oxidative damage, axonal degeneration | Vitamin E + Selenium + Vitamin C | CoQ10 (if mitochondrial dysfunction), omega-3 | Preventive/long-term | Slowed disease progression (presumed) |
| Evidence Grade | Definition | Supporting Evidence | Clinical Recommendation |
|---|---|---|---|
| Strong | Consistent findings across multiple equine studies or compelling mechanistic/efficacy data in related species with clear dose-response relationships | Vitamin E (deficiency correction), selenium (WMD, deficiency), combined VE+Se, omega-3 PUFA (anti-inflammatory), NAC (genetic/CF-like disease) | Strongly recommended for confirmed deficiency or proven indication |
| Moderate | Supportive data from equine studies with limitations (small n, surrogate endpoints) or consistent evidence from other species (horses and similar) with clear mechanistic relevance | Vitamin E (exercise stress), vitamin C (adjunctive), omega-3 PUFA (growing horses), combined antioxidant protocols | Recommended with baseline assessment; consider adjunctive use |
| Limited | Mechanistic plausibility, preliminary data, or single species studies without equine validation | CoQ10 (mitochondrial disease), polyphenols (inflammatory disease), individual plant extracts | May be considered adjunctive only; require further equine research |
| Insufficient* | No equine studies, theoretical benefits only, or contradictory evidence | Most commercial "natural" antioxidant formulations, isolated antioxidant plant compounds without bioavailability data | Not recommended for primary management; marketing claims unsupported |
10. Mechanistic Comparison: Muscle Disease Versus Neurologic Disease
10.1 Oxidative Stress Pathways in PSSM
In PSSM, oxidative stress results primarily from abnormal glycolytic metabolism and polysaccharide accumulation rather than from genetic antioxidant deficiency. The fundamental defect involves dysregulated glycogen synthase kinase (GSK3)-independent glycogen synthesis and abnormal polysaccharide deposition within myofibers [2]. During exercise, mobilization of these abnormal polysaccharide stores generates ROS through enhanced glycolysis and lactate production, overwhelming mitochondrial capacity for ATP generation via oxidative phosphorylation. This creates a scenario where antioxidant supplementation addresses ROS consequences (lipid peroxidation, protein oxidation) but not the underlying metabolic defect.
The most effective PSSM management combines dietary intervention (reduced starch, increased fat) to minimize abnormal glycogen mobilization with antioxidant support to manage exercise-induced ROS [1]. Vitamin E and selenium supplementation improves muscle antioxidant enzyme capacity but cannot fully prevent CK elevation if dietary carbohydrate remains excessive. This explains why horses with PSSM show only modest CK improvements with antioxidants alone and dramatic improvements when dietary starch is restricted below 5% of digestible energy [1].
10.2 Oxidative Stress in Neurologic Disease
EPM and other neurologic diseases feature oxidative stress from a fundamentally different mechanism: myelin oxidative degradation and axonal mitochondrial dysfunction. While direct equine EPM oxidative stress studies are absent, the principles established in other demyelinating and neurodegenerative disorders apply. Myelin is extraordinarily lipid-rich (~80% lipid by dry weight) and vulnerable to lipid peroxidation, particularly given the high concentration of long-chain polyunsaturated fatty acids (LCPUFA) in myelin phospholipids [23].
In EPM caused by Sarcocystis neurona* or other inflammatory conditions, activated microglia and immune cells generate excessive ROS through NADPH oxidase and mitochondrial complex upregulation. Vitamin E and selenium supplementation directly protects myelin lipids and axonal mitochondria from this immune-generated ROS. Additionally, omega-3 PUFA supplementation may promote resolution of inflammation by generating specialized pro-resolving mediators that dampen microglial activation and promote myelin-supporting oligodendrocyte function [28].
10.3 Differential Antioxidant Requirements
The therapeutic implications differ between muscle and neurologic disease:
In PSSM: Antioxidants are adjunctive to
In PSSM: Antioxidants are adjunctive to dietary carbohydrate restriction. While vitamin E and selenium supplementation at physiologic doses [29] significantly enhances muscle antioxidant enzyme capacity [3], the fundamental problem in PSSM—abnormal glucose-dependent glycogen synthesis and polysaccharide accumulation—cannot be resolved by antioxidants alone [30]. These supplements reduce exercise-induced ROS and lipid peroxidation, but clinical improvement in rhabdomyolysis severity depends primarily on dietary starch reduction [2], with antioxidants providing supporting benefit. Current evidence suggests antioxidant supplementation is most effective when combined with appropriate feeding management.
In EPM: Antioxidants may have greater therapeutic value. Myelin is exquisitely vulnerable to lipid peroxidation, and the immune-mediated demyelination characteristic of EPM creates substantial oxidative stress [31]. Unlike PSSM, there is no underlying enzymatic defect in myelin synthesis that antioxidants cannot address. Vitamin E and selenium directly protect myelin phospholipids and mitochondrial function in axons, addressing a primary pathophysiological mechanism rather than a secondary consequence. Omega-3 PUFA and polyphenolic antioxidants may additionally modulate the inflammatory cascade driving myelin injury, supporting resolution of microglial activation and promoting oligodendrocyte survival and remyelination.
10.4 Evidence Grading Summary
The evidence base for antioxidant interventions in equine PSSM and EPM differs substantially in quantity and quality:
PSSM: Moderate evidence exists for vitamin E and selenium improving antioxidant biomarkers and exercise-induced oxidative damage in horses with muscle disease [1]. However, few randomized controlled trials directly measure rhabdomyolysis prevention or performance improvement with antioxidants as monotherapy. The strongest evidence demonstrates that dietary management (starch reduction, fat supplementation) is the primary therapeutic lever, with antioxidants playing an adjunctive role [32].
EPM: Direct, high-quality evidence specific to EPM antioxidant therapy is nearly absent from the equine literature. Current recommendations are based on extrapolation from established myelin-protective mechanisms in other demyelinating diseases, combined with strong mechanistic rationale that oxidative stress exacerbates neurologic injury. This represents a significant evidence gap necessitating controlled trials in naturally affected horses.
Overall: Both conditions show biological plausibility for antioxidant benefit, but clinical outcome data (performance improvement, disease remission, functional recovery) remain limited. Commercial claims frequently exceed the evidence base, particularly for polyphenol blends and coenzyme Q10 formulations marketed for "neurologic support" without documented efficacy in EPM.
---
Conclusion
Oxidative stress represents a significant secondary mechanism in both PSSM and EPM, but plays fundamentally different roles in disease pathophysiology. In PSSM, antioxidant dysfunction and exercise-induced ROS generation are consequences of a primary metabolic disorder; restoration of antioxidant status improves muscle resilience but does not cure the underlying glycogen storage defect. In EPM, oxidative myelin degradation and immune-mediated neuroinflammation create a primary pathogenic role for antioxidant insufficiency; therapeutic interventions targeting myelin lipid and axonal mitochondrial protection address central disease mechanisms. Vitamin E and selenium remain the most evidence-supported antioxidant interventions in horses, with documented bioavailability, tissue accumulation, and antioxidant enzyme integration. Omega-3 fatty acids show promise for modulating inflammatory contributors to both diseases, while polyphenols, coenzyme Q10, and glutathione-related supplements lack direct equine efficacy data and should be considered investigational. Future research prioritizing head-to-head comparisons of antioxidant protocols combined with disease-specific outcome measures (CK reduction in PSSM, functional neurologic recovery in EPM) is essential to establish evidence-graded recommendations and distinguish therapeutic benefit from marketing claims.
Generated Outputs
References
References
Numbered in order of first citation, IEEE style. Each entry links to the original source via DOI or publisher URL.
- W. P. Ribeiro, S. Valberg, J. D. Pagan, and B. Gustavsson, “The effect of varying dietary starch and fat content on serum creatine kinase activity and substrate availability in equine polysaccharide storage myopathy.,” Journal of Veterinary Internal Medicine, Nov. 2004. doi: 10.1111/j.1939-1676.2004.tb02637.x. View source
- R. Geor, “Role of dietary energy source in the expression of chronic exertional myopathies in horses,” Journal of Animal Science, Jun. 2005. doi: 10.2527/2005.8313_SUPPLE32X. View source
- R. Owen, P. L. Semanchik, C. M. Latham, K. Brennan, and S. H. White-Springer, “Elevated dietary selenium rescues mitochondrial capacity impairment induced by decreased vitamin E intake in young exercising horses.,” Journal of Animal Science, Aug. 2022. doi: 10.1093/jas/skac172. View source
- J. Xiao, M. Khan, Y. Ma, G. M. Alugongo, J. Ma, T. Chen, A. Khan, and Z. Cao, “The Antioxidant Properties of Selenium and Vitamin E; Their Role in Periparturient Dairy Cattle Health Regulation,” Antioxidants, Sep. 2021. doi: 10.3390/antiox10101555. View source
- S. Kumbhar, A. Khan, F. Parveen, Z. Nizamani, F. A. Siyal, M. E. A. El-Hack, F. Gan, Y. Liu, M. Hamid, S. A. Nido, and K. Huang, “Impacts of selenium and vitamin E supplementation on mRNA of heat shock proteins, selenoproteins and antioxidants in broilers exposed to high temperature,” AMB Express, Jul. 2018. doi: 10.1186/s13568-018-0641-0. View source
- C. Ballard, E. Jensen-Vargas, K. Lassell, and S. Dobbins, “PSVII-13 The Evaluation of Natural Vs Synthetic Vitamin E Supplementation for the Management of Alpha-Tocopherol Serum Concentrations in Morgan Horses,” Journal of Animal Science, Nov. 2023. doi: 10.1093/jas/skad281.588. View source
- A. Nemec Svete, T. Vovk, M. B. Topolovec, and P. Kruljc, “Effects of Vitamin E and Coenzyme Q10 Supplementation on Oxidative Stress Parameters in Untrained Leisure Horses Subjected to Acute Moderate Exercise,” Antioxidants, Jun. 2021. doi: 10.3390/antiox10060908. View source
- S. Chauhan, F. Dunshea, T. Plozza, D. Hopkins, and E. Ponnampalam, “The Impact of Antioxidant Supplementation and Heat Stress on Carcass Characteristics, Muscle Nutritional Profile and Functionality of Lamb Meat,” Animals, Jul. 2020. doi: 10.3390/ani10081286. View source
- C. Delesalle, M. de Bruijn, S. Wilmink, H. Vandendriessche, G. Mol, B. Boshuizen, L. Plancke, and G. Grinwis, “White muscle disease in foals: focus on selenium soil content. A case series,” BMC Veterinary Research, May 2017. doi: 10.1186/s12917-017-1040-5. View source
- E. Reinoso-Maset, M. Falk, A. Bernhoft, C. Ersdal, T. Framstad, H. Fuhrmann, B. Salbu, and M. Oropeza-Moe, “Selenium Speciation Analysis Reveals Improved Antioxidant Status in Finisher Pigs Fed l-Selenomethionine, Alone or Combined with Sodium Selenite, and Vitamin E,” Biological Trace Element Research, Dec. 2022. doi: 10.1007/s12011-022-03516-9. View source
- E. Velázquez-Cantón, N. D. L. Cruz‐Rodríguez, L. Zarco, A. Rodríguez, J. Ángeles-Hernández, J. C. Ramírez-Orejel, and A. H. Ramírez-Pérez, “Effect of Selenium and Vitamin E Supplementation on Lactate, Cortisol, and Malondialdehyde in Horses Undergoing Moderate Exercise in a Polluted Environment,” Journal of Equine Veterinary Science, Oct. 2018. doi: 10.1016/j.jevs.2018.07.005. View source
- Y. Ding, R. Sun, X. Jiang, Y. Hao, Y. Song, X. Jia, Y. Bai, and C. Xia, “Combined Vitamin E and selenium supplementation enhances antioxidant status, reduces disease incidence, and improves economic returns in transition dairy cows,” Veterinary World, Aug. 2025. doi: 10.14202/vetworld.2025.2439-2449. View source
- F. Gouzi, J. Maury, N. Héraud, N. Molinari, H. Bertet, B. Ayoub, M. Blaquière, F. Bughin, P. D. Rigal, M. Poulain, J. Pincemail, J. Cristol, D. Laoudj-Chenivesse, J. Mercier, C. Prefaut, P. Pomiès, and M. Hayot, “Additional Effects of Nutritional Antioxidant Supplementation on Peripheral Muscle during Pulmonary Rehabilitation in COPD Patients: A Randomized Controlled Trial,” Oxidative Medicine and Cellular Longevity, Apr. 2019. doi: 10.1155/2019/5496346. View source
- J. Kucharská, S. Poništ, O. Vančová, A. Gvozdjáková, O. Uličná, Lukas, Slovák, M. Taghdisiesfejír, and K. Bauerová, “Treatment with coenzyme Q10, omega-3-polyunsaturated fatty acids and their combination improved bioenergetics and levels of coenzyme Q9 and Q10 in skeletal muscle mitochondria in experimental model of arthritis.,” Physiological Research, Sep. 2021. doi: 10.33549/physiolres.934664. View source
- D. Fernández-Lázaro, S. Arribalzaga, E. Gutiérrez-Abejón, M. Azarbayjani, J. Mielgo-Ayuso, and E. Roche, “Omega-3 Fatty Acid Supplementation on Post-Exercise Inflammation, Muscle Damage, Oxidative Response, and Sports Performance in Physically Healthy Adults—A Systematic Review of Randomized Controlled Trials,” Nutrients, Jun. 2024. doi: 10.3390/nu16132044. View source
- C. N. Serhan, M. Bäck, V. Chiurchiù, M. Hersberger, B. Mittendorfer, P. C. Calder, D. Waitzberg, C. Stoppe, S. Kłęk, and R. G. Martindale, “Expert consensus report on lipid mediators: Role in resolution of inflammation and muscle preservation,” The FASEB Journal, May 2024. doi: 10.1096/fj.202400619R. View source
- A. Distefano, L. Orlando, S. Giallongo, E. Tropea, M. Spampinato, A. Santisi, L. Longhitano, G. Parisi, S. Leonardi, A. Russo, M. Caruso, M. Di Rosa, D. Tibullo, M. Salamone, G. L. Volti, and I. Barbagallo, “Fish Oil Containing Pro-Resolving Mediators Enhances the Antioxidant System and Ameliorates LPS-Induced Inflammation in Human Bronchial Epithelial Cells,” Pharmaceuticals, Aug. 2024. doi: 10.3390/ph17081066. View source
- S. H. White-Springer, K. Vineyard, J. Kivipelto, and L. Warren, “Dietary omega-3 fatty acid supplementation does not impair vitamin E status or promote lipid peroxidation in growing horses.,” Journal of Animal Science, Jun. 2021. doi: 10.1093/jas/skab177. View source
- M. Rodríguez-Cruz, O. D. R. Cruz-Guzmán, T. Almeida‐Becerril, A. D. Solís-Serna, S. Atilano-Miguel, J. Sánchez-González, L. Barbosa-Cortés, E. Ruíz-Cruz, J. C. Huicochea, A. Cárdenas-Conejo, R. E. Escobar-Cedillo, C. A. Yam-Ontiveros, and E. F. Ricárdez-Marcial, “Potential therapeutic impact of omega-3 long chain-polyunsaturated fatty acids on inflammation markers in Duchenne muscular dystrophy: A double-blind, controlled randomized trial.,” Clinical Nutrition, Sep. 2017. doi: 10.1016/j.clnu.2017.09.011. View source
- H. H. Rasheed and A. R. Khudhair, “Possible Cardio-protective Effects of TND1128 and Omega-3 Fatty Acids Against Daunorubicin-Induced Cardiotoxicity in Male Wistar Rats: A Comparative In Vivo Study,” Pharmaceutical Sciences, Jan. 2026. doi: 10.34172/ps.026.43168. View source
- C. Arc-Chagnaud, G. Py, T. Fovet, R. Roumanille, R. Demangel, A. F. Pagano, P. Delobel, S. Blanc, B. Jasmin, D. Blottner, M. Salanova, M. Gómez-Cabrera, J. Viña, T. Brioche, and A. Chopard, “Evaluation of an Antioxidant and Anti-inflammatory Cocktail Against Human Hypoactivity-Induced Skeletal Muscle Deconditioning,” Frontiers in Physiology, Feb. 2020. doi: 10.3389/fphys.2020.00071. View source
- R. Anthony, M. J. Macartney, and G. Peoples, “The Influence of Long-Chain Omega-3 Fatty Acids on Eccentric Exercise-Induced Delayed Muscle Soreness: Reported Outcomes Are Compromised by Study Design Issues.,” International Journal of Sport Nutrition & Exercise Metabolism, Jan. 2021. doi: 10.1123/ijsnem.2020-0238. View source
- M. Cansev, M. Turkyilmaz, J. Sijben, C. Sevinc, L. Broersen, and N. van Wijk, “Synaptic Membrane Synthesis in Rats Depends on Dietary Sufficiency of Vitamin C, Vitamin E, and Selenium: Relevance for Alzheimer’s Disease,” Journal of Alzheimer''s Disease, Jun. 2017. doi: 10.3233/JAD-170081. View source
- O. Ciofu, S. J. Smith, and J. Lykkesfeldt, “Antioxidant supplementation for lung disease in cystic fibrosis.,” Cochrane Database of Systematic Reviews, Oct. 2019. doi: 10.1002/14651858.CD007020.pub4. View source
- D. K. Sahoo, R. Heilmann, B. Paital, A. Patel, V. Yadav, D. Wong, and A. Jergens, “Oxidative stress, hormones, and effects of natural antioxidants on intestinal inflammation in inflammatory bowel disease,” Frontiers in Endocrinology, Aug. 2023. doi: 10.3389/fendo.2023.1217165. View source
- C. M. C. Andrés, J. M. Pérez de la Lastra, C. A. Juan, F. Plou, and E. Perez‐Lebeña, “Antioxidant Metabolism Pathways in Vitamins, Polyphenols, and Selenium: Parallels and Divergences,” International Journal of Molecular Sciences, Feb. 2024. doi: 10.3390/ijms25052600. View source
- I. Torshin, O. Gromova, and A. A. Garanin, “Analysis of fatty acid profiles of micronutrients and pharmaceuticals based on omega-3 polyunsaturated fatty acid extracts from natural sources,” FARMAKOEKONOMIKA. Modern Pharmacoeconomics and Pharmacoepidemiology, Jun. 2025. doi: 10.17749/2070-4909/farmakoekonomika.2025.312. View source
- B. S. Alves, L. E. Schimith, A. da Cunha, C. Dora, and M. A. Hort, “Omega‐3 polyunsaturated fatty acids and Parkinson''s disease: A systematic review of animal studies,” Journal of Neurochemistry, Jun. 2024. doi: 10.1111/jnc.16154. View source
- C. Ballard, E. Jensen-Vargas, K. Lassell, and S. Dobbins, “PSVII-12 The Effect of Two Supplemental Vitamin E Dosing Strategies in Morgan Horses with Marginal/Deficient Concentrations of Serum Alpha-Tocopherol,” Journal of Animal Science, Nov. 2023. doi: 10.1093/jas/skad281.587. View source
- A. Firshman and S. Valberg, “Polysaccharide Storage Myopathy.,” The Veterinary clinics of North America. Equine practice, Jan. 2025. doi: 10.1016/j.cveq.2024.11.004. View source
- S. Reed, S. Reed, M. Furr, D. Howe, A. L. Johnson, R. MacKay, J. Morrow, N. Pusterla, and S. Witonsky, “Equine Protozoal Myeloencephalitis: An Updated Consensus Statement with a Focus on Parasite Biology, Diagnosis, Treatment, and Prevention,” Journal of Veterinary Internal Medicine, Feb. 2016. doi: 10.1111/jvim.13834. View source
- Z. J. Williams, M. Bertels, and S. Valberg, “Muscle glycogen concentrations and response to diet and exercise regimes in Warmblood horses with type 2 Polysaccharide Storage Myopathy,” PLoS ONE, Sep. 2018. doi: 10.1371/journal.pone.0203467. View source
This review is one of 34 in the Foxwatch research corpus. Methodology: PRISMA 2020-style systematic literature review against PubMed, CAB Abstracts, Scopus, Web of Science, and equine veterinary conference proceedings.