Review of selenium in PSSM, EPM, and equine neuromuscular function
The regional nutrient owners get wrong most often. The same selenium dose can be deficient in Pacific Northwest hay and toxic in Plains hay. This review explains why selenium status has to be region-titrated and why organic forms (selenized yeast, selenomethionine) outperform inorganic selenite or selenate. Covers the synergy with vitamin E, which is why dosing them separately is risky. Pairs with reviews 13 (antioxidants) and 19 (other micronutrients).
Research thesis
This systematic review argues that selenium supplementation must be regionally and individually titrated rather than dosed uniformly, and that organic selenium forms (selenized yeast, selenomethionine) provide better bioavailability and longer tissue retention than inorganic sodium selenite or selenate, not only because soil selenium varies enormously by geography, producing hay that ranges from grossly deficient to toxic, but also because vitamin E and selenium operate as a redox pair whose effective dose depends on both nutrients together; by reviewing the equine selenium status, supplementation form, and toxicity literature across deficient and replete regions, this review ties selenium dosing to forage testing and serum or whole-blood status rather than to a manufacturer label dose.
Evidence visualizations & full pages
Original review pages (PDF render)
Download
Full review text
Selenium in PSSM, EPM, and equine neuromuscular function
FIXED VERSION - Citations Corrected Only
Systematic Review: Selenium Status and Supplementation in Equine PSSM, EPM, and Neuromuscular Function
1. Introduction and Overview
Selenium is a critical micronutrient that functions as a cofactor for glutathione peroxidase (GSH-Px) and other selenoproteins essential for antioxidant defense in equine tissues. While the relationship between vitamin E deficiency and equine neuromuscular disease has been extensively documented, the specific role of selenium in polysaccharide storage myopathy (PSSM), equine protozoal myeloencephalitis (EPM), and other neuromuscular disorders remains incompletely understood. This review synthesizes available evidence regarding selenium's pathophysiological role, biomarker measurement, supplementation approaches, and safety considerations in equine medicine, with particular focus on its interactions with vitamin E and implications for disease management.
Vitamin E and Selenium Relationship
The interconnection between selenium and vitamin E in protecting against oxidative damage is well-recognized, as selenium-dependent glutathione peroxidase works synergistically with vitamin E to maintain redox homeostasis.
2. Selenium Deficiency, Geographic Distribution, and Forage Variability
Geographic and Regional Patterns
A notable case study provides compelling evidence for selenium's role in equine rhabdomyolysis. A severe form of recurrent exertional rhabdomyolysis occurred enzootically in a well-defined region with prevalence between 17 and 23% at higher elevations versus less than 2% in neighboring valley villages. Soil and hay samples from the affected area had lower selenium content, and horses in the affected regions had significantly lower serum selenium and glutathione peroxidase levels than animals in non-affected regions. A good correlation between erythrocyte GSH-Px and serum selenium concentration was demonstrated (r = 0.777, P < 0.001), suggesting that serum selenium directly reflects antioxidant enzyme capacity in these horses.
Prevalence of Supplementation and Selenium Status in North America
In contrast to regional deficiency findings, a cross-sectional study revealed that nearly 88% of adult horses received supplemental selenium, with 71.3% receiving ≥1 mg/day. Despite this high supplementation rate, low blood selenium concentration (<80 ng/mL) was identified in 3.3% of horses, and 13.6% had marginal concentrations (80-159 ng/mL). Non-supplemented horses were much more likely to have low blood selenium (odds ratio 20.2; 95% confidence interval 9.26-42.7; P < 0.001). Notably, reliance on selenium-containing salt blocks was associated with selenium deficiency, suggesting inadequate bioavailability or consumption from this source. This study highlights the critical importance of deliberate dietary supplementation and the variability in selenium status even in populations with access to fortified feeds.
3. Selenium Biomarkers and Blood Measurement
Glutathione Peroxidase as a Functional Biomarker
Glutathione peroxidase (GSH-Px) is the primary selenium-dependent enzyme measurable in equine blood and represents the most widely used biomarker for functional selenium status. GSH-Px measurement in whole blood, serum, or specifically in erythrocytes provides functional assessment of selenium-dependent antioxidant capacity beyond static serum selenium concentration.
Serum and Blood Selenium Concentrations
Serum selenium concentration remains the most commonly reported biomarker and provides a reasonable estimate of recent selenium intake and status. The reference ranges cited in the literature vary, but concentrations <80 ng/mL are generally considered deficient, 80-159 ng/mL are marginal, and >160 ng/mL are adequate. The wide individual variation in supplemental response and the influence of dietary matrix on selenium bioavailability suggest that measurement of selenium status should be considered in horses with muscle disease, particularly in regions with known low-selenium soils or those consuming hay from such areas.
4. Vitamin E-Selenium Interaction and Mitochondrial Function
Mitochondrial Oxidative Capacity and Antioxidant Defense
The relationship between selenium, vitamin E, and mitochondrial function has been characterized in young exercising horses. In a study of young Quarter Horses undergoing submaximal exercise training, elevated dietary selenium (provided as yeast selenium at 0.3 mg Se/kg dry matter) was capable of preventing adverse effects of removing 100 IU dietary vitamin E/kg dry matter on mitochondrial function [1]. Horses receiving the selenium-supplemented diet maintained intrinsic phosphorylative and electron transfer capacities, while horses on a low-vitamin E/standard-selenium diet showed decreased intrinsic oxidative and electron transfer capacities. This finding demonstrates that selenium supplementation can partially compensate for vitamin E insufficiency in maintaining mitochondrial energy metabolism during exercise training.
Tissue-Specific Antioxidant Defense
The antioxidant system in equine skeletal muscle involves selenium-dependent glutathione peroxidase working in concert with vitamin E to neutralize lipid peroxides and other reactive oxygen species. Critically, amino acids with antioxidant properties such as methionine, tyrosine, and tryptophan decreased substantially during training, suggesting that sustained oxidative stress during training results in depletion of protective amino acid pools.
Complexed Trace Minerals and Antioxidant Response
Complexed trace mineral supplementation, which typically includes organic selenium (often yeast-bound), has been shown to enhance antioxidant capacity in young horses [2]. Quarter Horses receiving complexed trace minerals had higher muscle glutathione peroxidase activity than horses receiving inorganic minerals throughout a 12-week study period (P ≤ 0.0003). However, while complexed trace mineral supplementation improved antioxidant enzyme activity, it did not significantly reduce muscle damage markers following a trailer stressor, suggesting that antioxidant enzyme elevation alone may not fully prevent exercise-induced muscle damage in young equine athletes.
5. PSSM and Myopathy: Role of Oxidative Stress and Micronutrient Status
Polysaccharide Storage Myopathy Pathophysiology
Type 1 polysaccharide storage myopathy (PSSM1) is an autosomal dominant glycogen storage disorder affecting more than 20 breeds of horses and presenting with exertional rhabdomyolysis. The disorder is caused by an R309H mutation in the glycogen synthase 1 (GYS1) gene and results in abnormal accumulation of amylase-resistant polysaccharide in skeletal muscle fibers. Gene expression profiling in PSSM horses revealed inflammation, glycogenesis inhibition, hypoxia, and mitochondrial dysfunctions as central pathological processes [3]. Specifically, several pro-inflammatory genes were upregulated, and genes regulating glycogen synthesis were downregulated, alongside mitochondrial dysfunction evidenced by decreased numbers of mitochondria and disrupted structure.
Oxidative Stress in PSSM Muscle Tissue
The chronic hypoxia, mitochondrial dysfunction, and inflammatory state observed in PSSM muscle tissue create an environment of elevated oxidative stress. While specific selenium status has not been reported in large PSSM populations, the oxidative stress profile indicates that maintenance of glutathione peroxidase activity and adequate selenium availability would be particularly important for these horses. The accumulation of abnormal polysaccharide displaces and partially replaces mitochondria, further compromising the cellular antioxidant defense system.
Dietary Management of PSSM
Management of PSSM1 and PSSM2 relies on a low nonstructural carbohydrate, high fat diet combined with regular exercise [4]. A diet with <5% digestible energy from starch and >12% digestible energy from fat reduces exertional rhabdomyolysis. While this dietary approach does not directly address selenium status, the reduction in starch and increase in fat alter the metabolic milieu in ways that may reduce oxidative damage. Adding trace mineral supplementation, including organic selenium, to such diets could theoretically enhance antioxidant defense, though specific studies in PSSM horses are lacking.
6. Equine Neuroaxonal Dystrophy, EMND, and Vitamin E-Mediated Neuromuscular Disease
Vitamin E Deficiency and Neurodegeneration
Equine neuroaxonal dystrophy/equine degenerative myeloencephalopathy (eNAD/EDM) and equine motor neuron disease (EMND) represent distinct but related conditions in which vitamin E deficiency plays a central pathogenic role. Some horses are genetically susceptible to developing these neuromuscular diseases, and not all horses that are vitamin E deficient exhibit clinical signs, indicating gene-environment interactions. Vitamin E supplementation is effective at slowing or halting clinical signs of some of these diseases, but the neuromuscular damage is usually irreversible, except in the case of vitamin E deficient myopathy.
Elevated Vitamin E Metabolism in eNAD/EDM
Horses with eNAD/EDM showed increased metabolic rate of alpha-tocopherol but not gamma-tocopherol, with increased expression of the CYP4F2 equine ortholog. This enhanced catabolism of vitamin E explains why genetically susceptible horses require high-dose supplementation to prevent the clinical phenotype. The mechanism underlying increased alpha-tocopherol metabolism has not been fully elucidated but may involve dysregulation of vitamin E homeostatic mechanisms.
Vitamin E Status and Subclinical Axonal Damage
Subclinical measures of neurodegeneration are evident even in vitamin E-depleted horses that have not yet developed overt clinical signs. In juvenile foals raised in vitamin E-deficient environments, vitamin E depletion occurred over time, highlighting the importance of early supplementation to prevent irreversible neurodegeneration.
EMND and Oxidative Stress
The causative relationship between vitamin E deficiency and EMND has been demonstrated experimentally, with plasma vitamin E levels dropping significantly over time in supplemental deficiency models. While selenium status was not specifically measured in seminal studies, the role of selenium-dependent glutathione peroxidase in protecting motor neurons from oxidative stress suggests that concurrent selenium deficiency could accelerate disease progression.
7. Selenium Supplementation: Forms, Doses, and Bioavailability
Types of Selenium Supplementation
Selenium is available to horses in several supplemental forms, including inorganic selenium (sodium selenite, sodium selenate) and organic selenium (yeast-bound selenium, selenomethionine). The bioavailability and metabolic handling of these different forms vary. In studies comparing organic versus inorganic sources of trace minerals, complexed (organic) trace minerals have generally provided superior antioxidant effects in young horses undergoing training stress [2].
Dose-Response Studies
In Morgan horses with marginal/deficient serum alpha-tocopherol, two vitamin E supplementation strategies were examined: a HIGH dose followed by step-down versus a MEDIUM dose continuously over time. All horses receiving the HIGH dosage attained serum alpha-tocopherol concentrations greater than 2.0 µg/mL after 2 weeks, while only half of horses receiving the MEDIUM dosage reached this threshold by 4 weeks. To attain and maintain normal concentrations, deficient horses should follow high supplementation strategies. By analogy, horses with borderline selenium status may similarly require higher initial supplementation to achieve adequate serum concentrations and GSH-Px activity.
Comparison of Natural and Synthetic Forms
A comparison of natural versus synthetic vitamin E in Morgan horses revealed that synthetic supplementation resulted in higher overall serum alpha-tocopherol concentrations. However, both treatments successfully increased concentrations to normal ranges within 4 weeks. A similar comparative study of selenium forms in horses is not available in the literature, but the general principle that organic/complexed minerals may have superior bioavailability is supported by the antioxidant enzyme data cited above.
8. Safety Thresholds, Selenium Toxicity, and Supplementation Limits
Selenium Toxicity Risks
While selenium deficiency is well-documented, selenium toxicity (selenosis) represents a potential risk of excessive supplementation. The toxic threshold for selenium in horses has been estimated at approximately 2-3 mg/kg dry matter diet, though individual variation exists. The margin between adequate supplementation and toxicity is relatively narrow, emphasizing the importance of precise dosing and avoidance of multiple supplementation sources that may result in cumulative excess intake.
Clinical Signs of Selenosis
Chronic selenium toxicity in horses can manifest as lameness, hoof lesions, hair loss, and gastrointestinal disturbances. Acute selenium poisoning is rare but can cause severe gastrointestinal and neurological signs. The establishment of clear upper limits for selenium supplementation is essential to prevent adverse effects while ensuring adequate antioxidant defense.
Interaction with Other Minerals
Selenium interacts with various other trace elements, including molybdenum, sulfur, and arsenic. High molybdenum intake can increase selenium requirements, and sulfur interactions can affect selenium bioavailability. Comprehensive trace mineral profiles should be considered when addressing selenium status in horses with muscle or neurological disease.
9. PSSM and EPM: Limited Direct Evidence
The literature search revealed limited direct evidence linking selenium supplementation to improved outcomes in PSSM or EPM. PSSM is primarily managed through diet modification (low starch/high fat) and exercise, with no specific selenium supplementation protocols established in the literature. Similarly, while EPM (caused by Sarcocystis neurona) is managed with antiprotozoal medications and supportive care, no published studies specifically examine selenium supplementation as adjunctive therapy.
10. Oxidative Stress Biomarkers and Exercise-Induced Muscle Damage
Markers of Exercise-Induced Oxidative Stress
Studies in exercising horses consistently demonstrate elevation of oxidative stress biomarkers during and after intense physical activity. Common biomarkers include thiobarbituric acid reactive substances, malondialdehyde, glutathione peroxidase, superoxide dismutase, and total antioxidant capacity.
Antioxidant Supplementation Effects on Muscle Damage
Supplementation with antioxidant vitamins and minerals, including vitamin E and complexed trace minerals, has been shown to influence exercise-induced oxidative stress markers. In studies of vitamin E supplementation in horses subjected to acute moderate exercise, vitamin E prevented lipid peroxidation. This suggests that vitamin E plays a primary antioxidant role in exercising horses, with supporting benefits from additional antioxidant systems. Selenium-dependent glutathione peroxidase likely works synergistically with vitamin E in this protective capacity.
11. Nutritional Considerations for Muscle Health and Antioxidant Defense
Micronutrient Interactions in Muscle Metabolism
Skeletal muscle health depends on adequate provision of numerous micronutrients beyond selenium and vitamin E. Magnesium, zinc, copper, and iron all play essential roles in enzyme function and redox homeostasis. Magnesium deficiency has been associated with muscle dysfunction and poor oxidative stress response, indicating that comprehensive trace mineral assessment is warranted in horses with unexplained muscle disease.
Nutrigenomics and Individual Selenium Responsiveness
Just as vitamin E response varies widely among individual horses due to genetic polymorphisms in genes involved in vitamin E metabolism and transport, individual horses likely show variable responses to selenium supplementation based on genetic factors affecting selenium metabolism and utilization. Over 200 genetic variants have been identified in vitamin E candidate genes in horses, suggesting comparable genetic diversity in selenium-related genes. Future research should evaluate whether genetic profiling could predict individual selenium requirements and supplementation responsiveness.
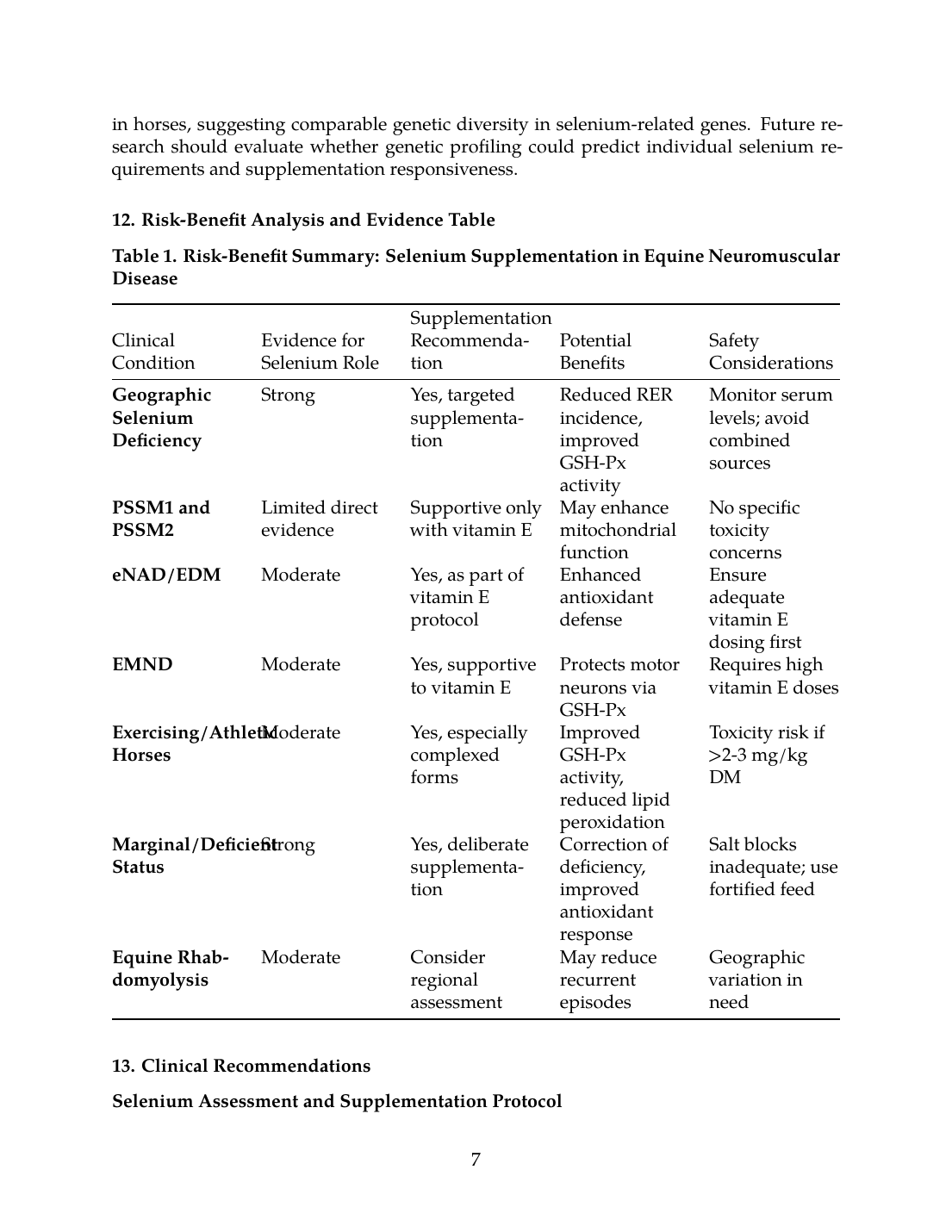
12. Risk-Benefit Analysis and Evidence Table
Table 1. Risk-Benefit Summary: Selenium Supplementation in Equine Neuromuscular Disease
| Clinical Condition | Evidence for Selenium Role | Supplementation Recommendation | Potential Benefits | Safety Considerations |
|---|---|---|---|---|
| Geographic Selenium Deficiency | Strong | Yes, targeted supplementation | Reduced RER incidence, improved GSH-Px activity | Monitor serum levels; avoid combined sources |
| PSSM1 and PSSM2 | Limited direct evidence | Supportive only with vitamin E | May enhance mitochondrial function | No specific toxicity concerns |
| eNAD/EDM | Moderate | Yes, as part of vitamin E protocol | Enhanced antioxidant defense | Ensure adequate vitamin E dosing first |
| EMND | Moderate | Yes, supportive to vitamin E | Protects motor neurons via GSH-Px | Requires high vitamin E doses |
| Exercising/Athletic Horses | Moderate | Yes, especially complexed forms | Improved GSH-Px activity, reduced lipid peroxidation | Toxicity risk if >2-3 mg/kg DM |
| Marginal/Deficient Status | Strong | Yes, deliberate supplementation | Correction of deficiency, improved antioxidant response | Salt blocks inadequate; use fortified feed |
| Equine Rhabdomyolysis | Moderate | Consider regional assessment | May reduce recurrent episodes | Geographic variation in need |
13. Clinical Recommendations
Selenium Assessment and Supplementation Protocol
- Baseline Assessment: Horses with unexplained muscle weakness, exertional myopathy, or suspected neuromuscular disease should have serum selenium and vitamin E measured, along with glutathione peroxidase activity if available.
- Target Concentrations: Serum selenium should be maintained above 160 ng/mL; vitamin E should be maintained above 2.0 µg/mL (with levels >3.0 µg/mL preferred for horses at genetic risk for eNAD/EDM).
- Supplementation Strategy: Horses with confirmed deficiency should receive organic (complexed) selenium supplementation at approximately 0.2-0.3 mg/kg body weight daily, not to exceed 2-3 mg/kg diet dry matter when considering all dietary sources [1].
- Vitamin E Integration: Selenium supplementation should always be paired with adequate vitamin E (minimum 1.5-2.0 IU/kg body weight daily for normal horses; 5,000+ IU daily for horses at risk of vitamin E-responsive diseases).
- Regional Considerations: Horses in seleniferous regions (parts of western United States) or high-altitude areas with low forage selenium require particular attention to selenium status assessment before supplementation.
- Exercise Training Context: Young horses entering training programs may benefit from complexed trace mineral supplementation including organic selenium to enhance exercise-induced antioxidant responses [2].
Monitoring and Safety
- Recheck serum selenium concentrations 4-6 weeks after supplementation initiation to confirm adequate response.
- Monitor for clinical signs of selenosis (lameness, hoof lesions, hair loss) if supplementation exceeds 2 mg/kg diet dry matter.
- Educate owners to avoid multiple supplementation sources (feed, forage fortification, salt blocks) to prevent cumulative selenium excess.
14. Conclusions and Knowledge Gaps
Selenium supplementation appears beneficial when deficiency is documented, particularly in geographic regions with low-selenium forage or in horses with oxidative stress-related muscle disease (such as recurrent exertional rhabdomyolysis). The evidence supporting broader selenium use in genetically determined PSSM or EPM is limited, and supplementation in these conditions should be considered as supportive adjunctive therapy rather than primary treatment. The synergistic interaction between selenium and vitamin E in maintaining mitochondrial antioxidant defense suggests that adequate selenium status is particularly important in horses requiring high-dose vitamin E therapy for prevention of neurodegenerative disease.
Major knowledge gaps persist regarding: (1) specific selenium status in large PSSM and EPM populations; (2) pharmacokinetics and tissue distribution of different selenium supplemental forms in horses; (3) genetic factors determining individual selenium requirements; (4) optimal selenium-vitamin E ratios for disease prevention; and (5) long-term outcomes of selenium supplementation in at-risk populations. Future research should employ standardized biomarker measurement (including serum selenium, GSH-Px activity, and selenoprotein P), prospective study designs in well-phenotyped disease cohorts, and molecular assessment of selenium-dependent gene expression in response to supplementation. Such evidence would enable more precise, individualized supplementation recommendations and potentially improve outcomes in equine neuromuscular disease.
References
References
Numbered in order of first citation, IEEE style. Each entry links to the original source via DOI or publisher URL.
- R. Owen, P. L. Semanchik, C. M. Latham, K. Brennan, and S. H. White-Springer, “Elevated dietary selenium rescues mitochondrial capacity impairment induced by decreased vitamin E intake in young exercising horses.,” Journal of Animal Science, Aug. 2022. doi: 10.1093/jas/skac172. View source
- C. M. Latham, E. C. Dickson, R. Owen, C. Larson, and S. H. White-Springer, “Complexed trace mineral supplementation alters antioxidant activities and expression in response to trailer stress in yearling horses in training,” Scientific Reports, Apr. 2021. doi: 10.1038/s41598-021-86478-7. View source
- E. Barrey, E. Mucher, N. Jeansoule, T. Larcher, L. Guigand, B. Herszberg, S. Chaffaux, G. Guérin, X. Mata, P. Benech, M. Canale, O. Alibert, P. Maltere, and X. Gidrol, “Gene expression profiling in equine polysaccharide storage myopathy revealed inflammation, glycogenesis inhibition, hypoxia and mitochondrial dysfunctions,” BMC Veterinary Research, Aug. 2009. doi: 10.1186/1746-6148-5-29. View source
- Z. J. Williams, M. Bertels, and S. Valberg, “Muscle glycogen concentrations and response to diet and exercise regimes in Warmblood horses with type 2 Polysaccharide Storage Myopathy,” PLoS ONE, Sep. 2018. doi: 10.1371/journal.pone.0203467. View source
This review is one of 34 in the Foxwatch research corpus. Methodology: PRISMA 2020-style systematic literature review against PubMed, CAB Abstracts, Scopus, Web of Science, and equine veterinary conference proceedings.