Systematic review of omega-3 fatty acids in equine neuromuscular disease
Fish oil and algae are reasonable add-ons but not a fix. This review walks through the evidence for EPA and DHA in equine neuromuscular disease, explains why marine sources beat plant sources for horses, and shows which omega-3 products actually deliver what they advertise. A useful adjunct for the EPM rehab horse. Pairs with reviews 11 (acute EPM nutrition) and 13 (antioxidants).
Research thesis
This systematic review argues that omega-3 fatty acid supplementation in equine neuromuscular disease is biologically plausible and likely useful as a low-risk adjunct, but is not a substitute for primary dietary management of PSSM or for antiprotozoal treatment of EPM, not only because EPA and DHA modulate inflammatory eicosanoid production and membrane fluidity in skeletal muscle and the central nervous system, but also because dose-response data in horses lag the human and rodent literature and many equine omega-3 products vary widely in EPA and DHA content; by examining inflammation-marker, clinical-outcome, and dose-response evidence across PSSM, EPM, and general equine populations, this review defines defensible dosing ranges, prefers marine over plant sources where evidence supports it, and identifies which clinical outcomes genuinely respond.
Evidence visualizations & full pages
Original review pages (PDF render)
Download
Full review text
Omega-3 fatty acids in equine neuromuscular disease
Systematic Review: Omega-3 Fatty Acid Supplementation in Equine Neuromuscular Disease
1. Introduction and Disease Context
Omega-3 polyunsaturated fatty acids (PUFAs) have emerged as potential nutritional interventions for equine neuromuscular diseases, though the evidence base in horses remains limited compared to human and rodent studies. The primary equine neuromuscular conditions of interest include polysaccharide storage myopathy (PSSM) Type 1 and Type 2/muscle fiber fragmentation (MFM), recurrent exertional rhabdomyolysis (RER), equine protozoal myeloencephalitis (EPM) recovery, and inflammatory myopathies. Understanding how omega-3 supplementation may complement established dietary management strategies is crucial for developing evidence-based protocols in equine medicine.
1.1 Polysaccharide Storage Myopathy (PSSM)
PSSM Type 1 (PSSM1) is characterized by the GYS1 R309H mutation and abnormal polysaccharide accumulation in muscle, affecting primarily Quarter Horses and related breeds [1]. The hallmark dietary management approach involves reducing starch and increasing dietary fat to <5% digestible energy (DE) from starch and >12% DE from fat [2]. In this context, omega-3 supplementation represents a strategic expansion of the "high-fat" component, with potential immunomodulatory benefits beyond simple caloric provision.
PSSM Type 2 occurs in Quarter Horses without the PSSM1 mutation and presents intermediate histopathology featuring amylase-resistant polysaccharide accumulation with normal or near-normal glycogen metabolism [1]. Both conditions respond clinically to low-starch, high-fat diets combined with regular exercise, though the underlying genetic basis remains uncharacterized.
1.2 Recurrent Exertional Rhabdomyolysis
RER in Thoroughbreds represents a distinct exertional myopathy driven by stress-responsive mechanisms rather than glycogen storage abnormalities [3]. Horses with RER benefit from high-fat, low-nonstructural carbohydrate (NSC) diets, with improved post-prandial glucose and cortisol responses compared to high-starch feeding [4]. The role of systemic inflammation and stress hormones in RER pathogenesis suggests potential therapeutic utility for omega-3-derived anti-inflammatory mediators.
1.3 Equine Protozoal Myeloencephalitis (EPM)
EPM is primarily caused by Sarcocystis neurona and, less commonly, Neospora hughesi, affecting the central nervous system and causing asymmetric ataxia and neurological deficits [5]. Current FDA-approved treatments include ponazuril, diclazuril, and sulfadiazine/pyrimethamine combinations [6]. EPM pathogenesis involves both direct parasitic invasion and host immune response-mediated inflammation [7], suggesting potential adjunctive benefits from anti-inflammatory dietary strategies during recovery.
2. Omega-3 Fatty Acid Sources: Characteristics and Bioavailability
2.1 Plant-Based Sources
Flaxseed Oil: Flaxseed is one of the richest plant sources of α-linolenic acid (ALA), containing approximately 56% ALA by total fatty acid composition [8]. In a 16-week equine study, horses supplemented with flaxseed oil at 370 mg oil/kg body weight (BW) daily showed significant increases in plasma ALA concentration compared to canola oil, with both affecting the systemic n-6:n-3 ratio [9]. Importantly, flaxseed supplementation reduced the n-6:n-3 ratio in both plasma lipids and skin tissue, suggesting diet-induced changes in systemic fatty acid composition.
Camelina Oil: This emerging alternative from Camelina sativa, a hardy low-input oilseed crop, provides approximately 34.9% ALA and has been compared to flaxseed in recent equine trials [8]. At the same dose (0.37 g oil/kg BW/day for 16 weeks), camelina-supplemented horses showed comparable increases in plasma ALA and similar reductions in n-6:n-3 ratio compared to flaxseed, though neither produced statistically significant differences in immune response or inflammatory markers in healthy horses [8]. Camelina appears to be a viable sustainable alternative for ALA delivery, though equine-specific data are limited [10].
Bioavailability of Plant-Based ALA: The critical limitation of plant-based omega-3 sources lies in the conversion efficiency of ALA to its long-chain metabolites, eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA). In humans, less than 5% of ingested ALA is converted to EPA, and <0.5% is converted to DHA [11]. This conversion is mediated by Δ6- and Δ5-desaturases and is influenced by the dietary n-6:n-3 ratio, with maximal conversion observed at an LA:ALA ratio of 1:1 [12]. Supplementation with ALA combined with reduced linoleic acid (LA) intake more efficiently enhanced EPA blood concentrations than ALA supplementation alone [13]. No equine-specific data exist on ALA conversion efficiency; however, the low conversion rates documented in humans suggest that plant-based supplementation in horses would primarily deliver ALA rather than long-chain omega-3 metabolites.
2.2 Marine and Algal-Derived Sources
Fish Oil: While specific equine fish oil studies are absent from the reviewed literature, fish oil is the standard marine source of EPA and DHA. Typical fish oil supplements contain approximately 18% EPA and 12% DHA [14]. Triglyceride formulations of fish oil demonstrate superior bioavailability compared to ethyl ester or free fatty acid forms, making them preferable for supplementation [15].
Algae-Derived DHA/EPA: Microalgae such as Isochrysis galbana are sustainable sources of DHA and EPA without requiring fish farming [16]. Algal sources can be biofortified directly into equine feeds, though bioavailability studies in horses are absent. A human study of microalgal DHA-enriched yogurt showed poor bioaccessibility in vitro, with <10% of linoleic acid being bioaccessible and no detectable EPA or DHA in the digestible fraction [16], suggesting that formulation strategies significantly impact the effective delivery of these omega-3 fatty acids.
2.3 Omega-6:Omega-3 Ratio Effects
The n-6:n-3 ratio is a critical determinant of the inflammatory milieu. Contemporary Western diets maintain ratios of approximately 10-20:1 to 25:1, compared to ancestral diets with ratios closer to 1-2:1 [17]. In equine studies, supplementation with 370 mg oil/kg BW (flaxseed, camelina, or canola) for 16 weeks reduced the n-6:n-3 ratio in plasma and skin over time [9],[18]. Importantly, all three oil types produced comparable skin and coat improvements in healthy horses, with no differences in immune responsiveness (measured via keyhole limpet hemocyanin sensitization and delayed-type hypersensitivity) [8]. This suggests that at standardized doses in healthy horses, the ratio improvement achieved through supplementation does not translate to measurable immune benefits in the absence of underlying inflammatory disease.
3. Mechanisms of Action: From Molecular to Clinical Effects
3.1 Anti-Inflammatory and Pro-Resolving Mechanisms
Omega-3 PUFAs, particularly EPA and DHA, suppress the NF-κB signaling pathway and inhibit inflammasome activation, reducing pro-inflammatory cytokine expression [19]. These fatty acids serve as precursors to specialized pro-resolving mediators (SPMs)—resolvins, protectins, and maresins—that actively promote the resolution phase of inflammation [20]. In a transgenic mouse model with genetically elevated tissue n-3 PUFAs, colitis-induced inflammation and oxidative liver damage were significantly reduced compared to wild-type littermates, accompanied by increased n-3-derived oxylipins (19,20-epoxydocosapentaenoic acid, 15-hydroxyeicosapentaenoic acid, and 17,18-epoxyeicosatetraenoic acid) [21].
Human clinical trials in inflammatory conditions support this mechanism. In boys with Duchenne muscular dystrophy (DMD), 2.9 g/day of combined EPA+DHA for 6 months downregulated mRNA expression of NF-κB and its target genes IL-1β and IL-6 in leukocytes, with serum IL-1β decreasing 59.5% and IL-6 decreasing 54.8%, while IL-10 (anti-inflammatory) increased 99.9% [22]. These immunological shifts occurred without reported adverse effects.
3.2 Mitochondrial Function and Oxidative Capacity
Omega-3 PUFAs enhance mitochondrial oxidative phosphorylation efficiency and ATP production. In aged mares receiving flaxseed oil supplementation for 6 weeks followed by a supportive nutrient formulation, omega-3 supplementation (compared to corn oil control) improved oxidative phosphorylation efficiency and reduced reactive oxygen species release from granulosa cells and muscle, though these improvements were modest and enhanced only after addition of the supportive nutrient formulation [23]. This multi-component response highlights the importance of evaluating omega-3s as part of integrated nutritional strategies rather than isolated interventions.
Dietary omega-3 PUFAs modulate mitochondrial dynamics by promoting fusion over fission, with omega-3 PUFAs inducing fusion processes and improving mitochondrial function [24]. This fusion-promoting effect contrasts with saturated fatty acids, which enhance fission and mitochondrial fragmentation. In skeletal muscle, omega-3 supplementation increased mitochondrial respiration and AMPK activation, reducing endoplasmic reticulum and oxidative stress markers [25]. These effects are mediated in part through increased PGC1-α and PGC1-β expression, promoting mitochondrial biogenesis [25].
3.3 Muscle Regeneration and Satellite Cell Function
Omega-3 PUFAs support skeletal muscle regeneration by modulating satellite cell (SC) differentiation and myogenesis [26]. In dystrophic hamsters fed a 30% flaxseed-enriched diet, the flaxseed-supplemented diet protected muscle from apoptosis and preserved myogenesis by increasing myogenin and α-myosin heavy chain expression [27]. Additionally, flaxseed supplementation restored normal caveolin-3 expression patterns, maintaining protein retention at the sarcolemma—a critical feature for muscle membrane integrity.
In vitro, EPA, DPA (docosapentaenoic acid), and DHA prevented palmitate-induced myotoxicity and apoptosis in C2C12 myoblasts, with DPA specifically maintaining mitochondrial respiratory capacity and preventing cytochrome c release [28]. The n-3 PUFAs promoted myotube formation in the presence of palmitate, an effect absent when cells were treated with palmitate alone. These findings suggest that omega-3 supplementation may be particularly beneficial in conditions characterized by lipotoxicity or impaired myogenic regeneration.
3.4 Membrane Composition and Fluidity Effects
Omega-3 PUFAs are incorporated into phospholipid bilayers, altering membrane fluidity, receptor clustering, and cell signaling. In female subjects consuming 5 g/day of EPA+DHA for 8 weeks, skeletal muscle phospholipid EPA composition increased significantly compared to males, with elevated EPA levels persisting at 14 weeks of washout [29]. This sex-specific response suggests that hormone-dependent mechanisms may influence omega-3 incorporation into muscle membranes.
DHA incorporation into lipid bodies in activated microglia (N9 cells) triggered organelle remodeling, increasing the number of DHA-rich lipid bodies and promoting their contacts with mitochondria [30]. This remodeling reduced LPS-induced endoplasmic reticulum dilation, suggesting DHA reduces oxidative stress and protein misfolding stress in activated immune cells. The formation of electron-dense lipid bodies enriched with phosphatidylserine indicates DHA's role in restoring physiological membrane functions.
4. Evidence in Equine Neuromuscular Diseases: Assessment of Direct, Indirect, and Speculative Links
4.1 PSSM and PSSM2: Evaluation of Evidence Type
Direct Evidence: No published controlled trials of omega-3 supplementation in PSSM or PSSM2 horses exist. The systematic review identified zero randomized controlled trials specifically assessing omega-3 PUFA efficacy in these conditions.
Indirect Evidence: The dietary management of PSSM emphasizes increased fat provisioning while limiting starch. A seminal equine study demonstrated that PSSM horses fed a diet with >12% DE from fat and <5% DE from starch showed reduced serum creatine kinase (CK) activity and increased availability of free fatty acids (FFA) for muscle metabolism [2]. While this study did not specifically investigate omega-3 sources, it established that increased fat intake (independent of fatty acid composition) improves metabolic outcomes in PSSM. Omega-3-enriched oils could theoretically fulfill this "high-fat" requirement while simultaneously providing anti-inflammatory benefits; however, no equine study has compared omega-3-rich fat sources (flaxseed, fish oil) to non-omega-3 fats (coconut oil, canola oil) in PSSM horses.
The mechanism of PSSM1 involves abnormal glucose utilization and excessive muscle glycogen/polysaccharide storage. Omega-3 PUFAs enhance mitochondrial oxidative capacity [24], a pathway that could theoretically improve muscle's capacity to utilize the accumulated substrate; however, this mechanism remains unexplored in PSSM horses.
Mechanistically Speculative Evidence: Omega-3 PUFAs promote satellite cell differentiation and myogenesis [26], and flaxseed ALA protected dystrophic muscle from apoptosis in rodents [27]. PSSM2/MFM is characterized by fiber fragmentation and degeneration [1], potentially making it a candidate for myogenic support. However, the specific role of impaired myogenesis in PSSM2 pathogenesis is unknown, and no data exist linking omega-3-mediated myogenic effects to PSSM2 recovery.
Similarly, the observation that omega-3 PUFAs reduce insulin resistance in rodent models [25] is mechanistically interesting for PSSM1 (characterized by insulin dysregulation), but equine-specific insulin signaling effects of omega-3 supplementation have not been characterized.
4.2 Recurrent Exertional Rhabdomyolysis: Evidence Assessment
Indirect Evidence: RER is exacerbated by high-starch, high-NSC feeding and stress hormones [3]. Similar to PSSM, the standard management includes low-starch, high-fat diets [4]. In fit RER Thoroughbreds, a high-fat diet reduced postprandial glucose and insulin compared to high-NSC feeds, suggesting a metabolic protection mechanism [4]. Omega-3 supplementation, as a fat source component, could theoretically amplify these metabolic benefits; however, no controlled trials have evaluated this hypothesis.
Mechanistically Speculative Evidence: Omega-3 PUFAs reduce exercise-induced muscle damage and post-exercise inflammation in healthy humans. A systematic review of omega-3 supplementation in physically healthy adults found that creatine kinase (CK) and lactate dehydrogenase (LDH) were significantly higher in control groups compared to omega-3-supplemented groups in 3 of 4 studies assessing these markers [31]. While muscle damage biomarkers (CK, LDH) are central to RER diagnosis and management, equine-specific data on omega-3 effects on exercise-induced muscle injury are absent.
4.3 Equine Protozoal Myeloencephalitis Recovery: Evidence Assessment
Indirect Evidence: EPM pathogenesis involves both direct Sarcocystis neurona invasion of the CNS and secondary immune-mediated inflammation [7]. Horses with EPM exhibit reduced cerebrospinal fluid (CSF) nitric oxide metabolite (NOx−) concentrations compared to seropositive asymptomatic horses and seronegative controls [32], suggesting impaired neuroprotective NO signaling. Additionally, horses with EPM show a Th1-biased immune response with elevated IgG1/2 and IgG4/7 subisotypes compared to infected asymptomatic horses [33].
Omega-3 PUFAs enhance NO production and improve endothelial function [34], potentially reversing the NOx− deficit observed in EPM. Additionally, omega-3-derived resolvins and protectins promote Th17-to-Treg polarization and reduce Th1-dominant responses in inflammatory contexts [19]. These mechanistic links are plausible but entirely untested in equine EPM.
Mechanistically Speculative Evidence: DHA provides neuroprotection through mitochondrial stabilization [35] and reduces apoptosis in neurodegenerative models [36]. Given that EPM involves CNS parasitic invasion with secondary neuroinflammation, omega-3-derived neuroprotection could theoretically support neural recovery during treatment. However, no studies have evaluated omega-3 supplementation during or after EPM recovery.
4.4 Inflammatory Myopathies in Horses
The only available equine evidence on inflammatory myopathy management comes from case reports and necropsy studies. No controlled trials have assessed nutritional interventions, including omega-3 supplementation, in equine myositis. In humans with idiopathic inflammatory myopathies (IIMs), nutritional intervention through Mediterranean diet and omega-3-rich components has demonstrated potential for modulating disease activity, though rigorous equine trials are absent [37].
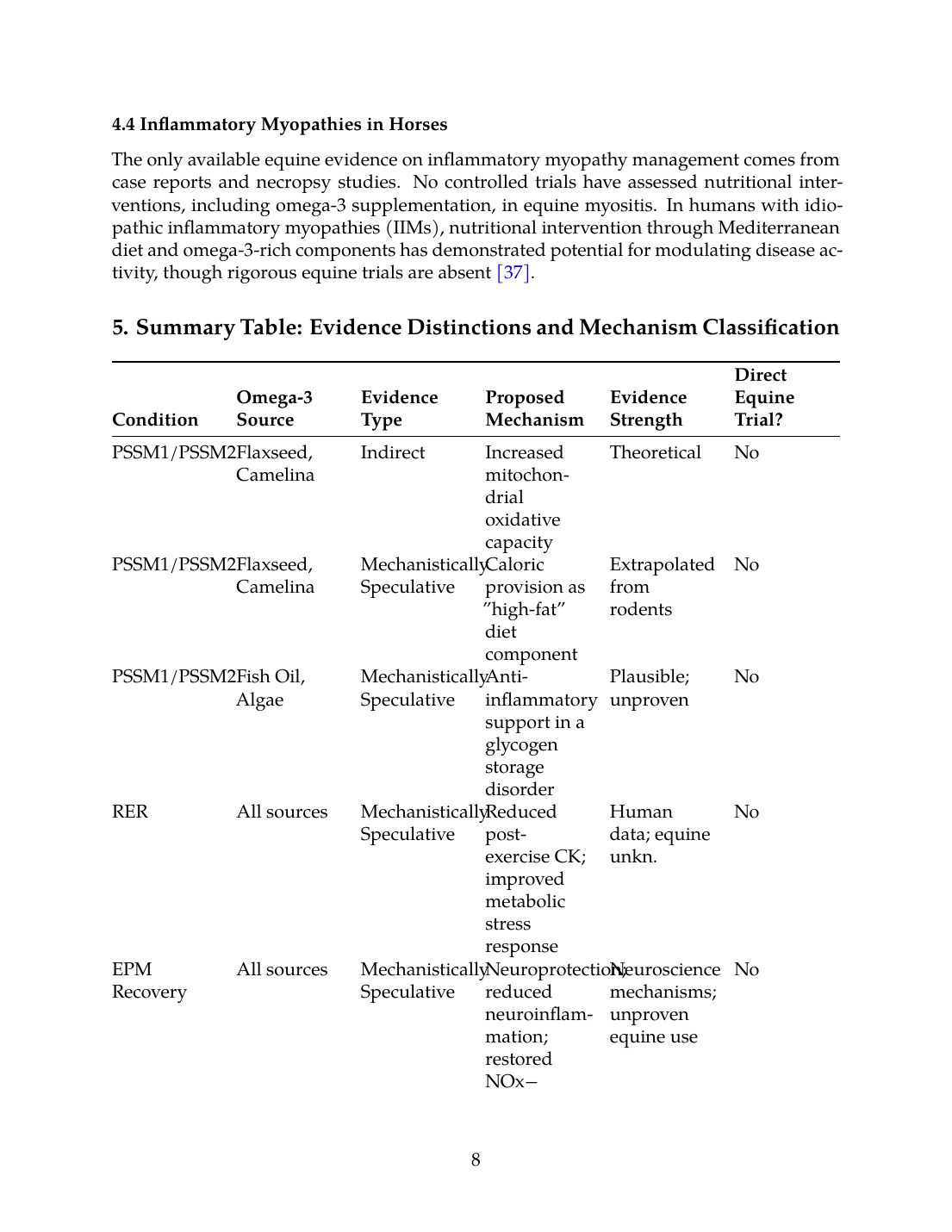
5. Summary Table: Evidence Distinctions and Mechanism Classification
| Condition | Omega-3 Source | Evidence Type | Proposed Mechanism | Evidence Strength | Direct Equine Trial? |
|---|---|---|---|---|---|
| PSSM1/PSSM2 | Flaxseed, Camelina | Indirect | Increased mitochondrial oxidative capacity | Theoretical | No |
| PSSM1/PSSM2 | Flaxseed, Camelina | Mechanistically Speculative | Caloric provision as "high-fat" diet component | Extrapolated from rodents | No |
| PSSM1/PSSM2 | Fish Oil, Algae | Mechanistically Speculative | Anti-inflammatory support in a glycogen storage disorder | Plausible; unproven | No |
| RER | All sources | Mechanistically Speculative | Reduced post-exercise CK; improved metabolic stress response | Human data; equine unkn. | No |
| EPM Recovery | All sources | Mechanistically Speculative | Neuroprotection; reduced neuroinflammation; restored NOx− | Neuroscience mechanisms; unproven equine use | No |
| Inflammatory Myositis | All sources | Mechanistically Speculative | NF-κB suppression; Th1-to-Treg polarization | Human IIM trials; equine untested | No |
5.1 Caloric Effects vs. Anti-inflammatory Effects
Caloric Effects: All omega-3 sources provide ~9 kcal/gram, equivalent to other dietary fats. In PSSM and RER management, the primary therapeutic role of "high-fat" diets appears to be caloric substitution for starch rather than anti-inflammatory activity per se. The landmark PSSM study demonstrated that CK reduction correlated with reduced starch intake and increased fat availability for FFA oxidation [2], a mechanism independent of the specific fatty acid composition of the fat. Thus, the caloric benefit of omega-3 supplementation overlaps entirely with that of non-omega-3 fats (coconut, canola).
Anti-inflammatory Effects: In contrast, anti-inflammatory benefits are specific to omega-3 composition. These effects operate through:
- NF-κB suppression and inflammasome inhibition [19]
- Generation of SPMs (resolvins, protectins, maresins) [21]
- Reduction in pro-inflammatory cytokines (TNF-α, IL-6, IL-1β) [22]
- Th1-to-Treg polarization shifts [19]
Critical Distinction: If omega-3 supplementation is used primarily to satisfy the "high-fat" component of PSSM/RER diets, any non-omega-3 fat source would be therapeutically equivalent. Anti-inflammatory benefits would only provide added value if the specific condition involves a primary inflammatory pathology—a hypothesis unproven in PSSM/PSSM2/RER but potentially relevant in EPM recovery and equine myositis.
5.2 Membrane Effects
Omega-3 PUFAs are preferentially incorporated into phospholipid membranes, altering:
- Membrane fluidity and lateral diffusion of signaling proteins
- Clustering of receptors and signaling complexes
- Organelle (particularly mitochondrial) membrane composition and function
In the equine context, membrane remodeling effects could theoretically:
- Enhance the capacity of muscle membranes to tolerate metabolic stress (relevant for PSSM, RER)
- Improve neuronal membrane integrity during EPM recovery
- Enhance mitochondrial energy production through improved membrane composition
However, these effects are dose- and duration-dependent (weeks to months of supplementation), and no equine studies have measured membrane fatty acid composition or biophysical properties in response to omega-3 supplementation. The assumption that dietary fatty acid changes translate into functional membrane improvements in horses remains unvalidated.
5.3 Unsupported Claims
Several frequently cited rationales for omega-3 supplementation in horses lack direct or indirect evidence:
- "Omega-3s directly treat PSSM polysaccharide accumulation": No mechanism by which omega-3 PUFAs directly reduce the rate of abnormal polysaccharide synthesis or accumulation exists. The GYS1 R309H mutation driving PSSM1 is unaffected by dietary fatty acids [1].
- "ALA-based supplements provide equivalent long-chain omega-3 benefits": Conversion of flaxseed ALA to EPA is 5-27% efficient and to DHA is <1% efficient in humans [11], and horses have not been studied. Marketing claims equating flaxseed "omega-3 content" to fish oil DHA are misleading.
- "Omega-3s reduce exercise-induced CK elevation in healthy horses": While human studies show CK reduction with omega-3 supplementation, no equine data exist, and extrapolation is speculative. The clinical significance of CK reduction in healthy horses (vs. horses with rhabdomyolysis) is unknown.
- "Omega-6:omega-3 ratio optimization alone improves muscle function": In healthy horses, shifting the ratio from 25:1 to 5:1 (via flaxseed supplementation) produced no measurable changes in immune responsiveness or muscle function markers [8].
6. Equine-Specific Biomarkers and Outcome Measures
6.1 Inflammatory Biomarkers in Equine Neuromuscular Disease
Creatine Kinase (CK): Elevated serum CK reflects muscle damage and is the primary diagnostic biomarker for exertional myopathies (PSSM, RER) and EPM-associated myositis. However, CK is non-specific and does not differentiate between inflammatory, metabolic, and traumatic causes. No equine studies have prospectively measured CK in response to omega-3 supplementation.
Interleukins and Cytokines: Human studies consistently show that omega-3 supplementation reduces IL-6 and TNF-α [22],[31]. Equine studies of inflammatory myopathy are limited; the only available data come from EPM research, where CSF cytokine profiles have not been systematically evaluated in response to dietary interventions [38].
Reactive Oxygen Species (ROS) and Antioxidant Status: Omega-3 PUFAs increase antioxidant enzyme expression (glutathione peroxidase, superoxide dismutase) and decrease lipid peroxidation markers [31]. Equine biomarkers of oxidative stress in muscle disease have not been extensively developed or validated, limiting the ability to assess omega-3 effects in practice.
6.2 Fatty Acid Incorporation and Omega-3 Index
Recent human research emphasizes the omega-3 index (EPA+DHA as % of total erythrocyte fatty acids), with levels ≥8% associated with reduced cardiovascular risk and improved outcomes in inflammatory conditions [15]. Equine studies have not established reference ranges or target values for the equine omega-3 index. The two published equine supplementation trials measured plasma fatty acid composition but did not establish clinically meaningful omega-3 index targets [9],[8].
7. Clinical Application Framework: Dose, Duration, and Integration with Established Protocols
7.1 Dosing Regimens in Equine Studies
The two equine supplementation trials employed identical doses of 0.37 g oil/kg BW/day (370 mg/kg):
- Flaxseed oil: 56% ALA = ~207 mg ALA/kg BW/day [9],[8]
- Camelina oil: 34.9% ALA = ~129 mg ALA/kg BW/day [8]
- Canola oil: 12.0% ALA = ~44 mg ALA/kg BW/day [9]
For a 500 kg horse:
- Flaxseed: ~185 grams of oil or ~330 grams of flaxseed meal daily
- Camelina: ~185 grams of oil daily
- Fish oil (estimated from human data): 50-100 grams daily for 2-4g EPA+DHA would approximate human therapeutic doses
7.2 Duration of Supplementation
Both equine trials ran for 16 weeks with a 4-week acclimation period. Measurable changes in plasma and skin fatty acid composition occurred within this timeframe [9],[8]. Human studies show that meaningful immunological changes (cytokine downregulation, resolvin production) require 6-12 weeks of sustained supplementation [22]. No data exist on the optimal duration for equine neuromuscular disease; extrapolation from human trials suggests a minimum of 8-12 weeks before clinical benefits would be expected.
7.3 Integration with Standard PSSM/RER Dietary Management
In PSSM and RER, omega-3-rich fats can be positioned as the primary fat component of the low-starch, high-fat diet rather than as an adjunct supplement. For example:
- Standard approach: Base diet (hay/pasture) + non-omega-3 fat source (coconut, canola) at 12-15% DE
- Omega-3 integrated approach: Base diet + flaxseed or fish oil at 12-15% DE
This integration avoids the conceptual confusion of "supplementing with additional fat" and maintains the evidence-based starch/fat ratios established for PSSM management [2].
7.4 Background Diet Considerations
The efficacy of omega-3 supplementation depends on concurrent dietary composition. High dietary linoleic acid (LA) competes with ALA for metabolism, reducing EPA conversion [12]. Equine grain-based and many commercial pelleted feeds are high in LA from vegetable oils; thus, horses consuming such feeds may experience impaired ALA bioavailability. In the equine trials, baseline diets were not specified as being LA-controlled, potentially limiting the efficacy of ALA supplementation [9].
For horses on high-carbohydrate diets (the typical risk factor for PSSM/RER clinical signs), concurrent reduction in refined carbohydrate sources may be necessary to observe omega-3 benefits. No equine studies have evaluated omega-3 supplementation in horses maintained on typical grain-based rather than low-starch diets.
8. Safety and Adverse Effects
8.1 Equine-Specific Safety Data
The two equine supplementation trials found no adverse effects of flaxseed, camelina, or canola oil supplementation at 370 mg/kg BW/day over 16 weeks, with no changes in complete blood counts, serum biochemical profiles, body condition, or body weight [9],[8]. Horses demonstrated normal tolerance to gradual oil supplementation following a 4-week acclimation period.
8.2 Potential Omega-3 Oxidation and Lipid Peroxidation
Omega-3 PUFAs are highly susceptible to oxidative degradation due to their multiple double bonds. A rat study found that high-dose fish oil enriched with 80% DHA increased plasma lipid peroxidation and protein carbonylation compared to lower-DHA fish oils, along with elevated antioxidant enzyme expression [14]. While these changes were interpreted as adaptive oxidative stress responses, the clinical relevance of elevated lipid peroxidation is uncertain. Equine studies have not addressed this concern.
8.3 Interactions with Exercise and Heat Stress
Omega-3 PUFAs increase membrane fluidity and metabolic rate, theoretically enhancing oxidative capacity but potentially increasing thermal stress during exercise. In aged mares, omega-3 supplementation modestly increased muscle oxidative phosphorylation but also tended to increase ROS release from granulosa cells and muscle [23]. These dual effects (enhanced energy production + increased oxidative burden) have not been characterized in exercised horses.
9. Comparison with Established Equine Neuromuscular Therapies
9.1 PSSM/RER: Omega-3 vs. Established Dietary Management
The gold-standard therapy for PSSM and RER remains strict low-starch/high-fat dietary management combined with regular exercise [3],[2]. This approach consistently reduces CK elevation and clinical signs. Omega-3 supplementation, if it provided measurable benefit, would represent an incremental improvement to an already-effective intervention. No equine trial has demonstrated superiority of any omega-3 source over non-omega-3 fat sources in these conditions.
9.2 EPM: Omega-3 vs. Antiprotozoal Therapy
Current FDA-approved EPM treatments (ponazuril, diclazuril, sulfadiazine/pyrimethamine) directly target parasite replication and are the standard of care [6]. Omega-3 supplementation would be purely adjunctive, potentially supporting neural recovery and reducing secondary inflammation. However, the lack of clinical trials means that the magnitude of potential benefit is unknown, and omega-3 supplementation should not delay or substitute for antiprotozoal therapy.
9.3 Equine Myositis: Omega-3 vs. Corticosteroids and Immunosuppression
Idiopathic inflammatory myopathies in horses are rare, and evidence-based treatment is limited. Corticosteroids remain the first-line therapy, with supplementary immunosuppression (methotrexate, rituximab) in refractory cases [37]. Omega-3 supplementation could theoretically provide anti-inflammatory support, but any such use would be adjunctive to established immunosuppressive therapy.
10. Knowledge Gaps and Recommendations for Future Research
10.1 Critical Evidence Gaps
- No direct equine RCTs of omega-3 supplementation in PSSM, PSSM2, RER, or EPM recovery exist. All current recommendations are extrapolated from rodent or human data.
- Equine ALA conversion efficiency is unknown. Assuming human conversion rates (~5% to EPA, <0.5% to DHA) in horses may substantially overestimate the efficacy of flaxseed supplementation.
- No biomarker correlation studies have linked omega-3 supplementation to changes in equine serum CK, inflammatory cytokines, or cerebrospinal fluid markers in neuromuscular disease.
- Optimal dosing, duration, and timing relative to disease progression are undefined. All equine supplementation trials lasted exactly 16 weeks in healthy horses; shorter durations and diseased populations have not been studied.
- Sex, age, and breed-specific responses to omega-3 supplementation have not been evaluated in horses, though human data suggest significant variability [29].
10.2 Recommendations for Future Equine Studies
Tier 1 (High Priority):
- Randomized controlled trial of omega-3 supplementation (flaxseed, fish oil) vs. matched non-omega-3 fat control in PSSM1 horses, measuring CK, muscle fatty acid composition, and clinical symptom frequency over 12 weeks
- Similar RCT in RER-affected Thoroughbreds measuring post-exercise CK, myoglobinuria, and clinical episodes
- Prospective observational study of omega-3 supplementation in EPM-affected horses during recovery phase, measuring CSF cytokines, nitric oxide metabolites, and neurological improvement
Tier 2 (Important but Secondary):
- Equine-specific studies of ALA conversion to EPA/DHA using stable isotope labeling
- Correlation of equine omega-3 index (erythrocyte EPA+DHA %) with muscle biopsy fatty acid composition
- Investigation of optimal omega-6:omega-3 ratios for equine neuromuscular health
Tier 3 (Longer-term Mechanistic):
- Characterization of equine satellite cell responses to omega-3-enriched culture conditions
- Mitochondrial function studies in equine myocytes supplemented with omega-3 PUFAs
- Neuroinflammatory marker profiling in EPM-affected horses with and without omega-3 supplementation
11. Summary and Recommendations for Clinical Practice
11.1 Current Evidence Summary
PSSM/PSSM2:
- Evidence type: Indirect, caloric
- Recommendation level: Cannot recommend omega-3 sources over non-omega-3 fats based on current evidence; if using omega-3-rich oil as the fat component of a low-starch diet, ensure the total starch/fat ratio remains within evidence-based targets (<5% DE starch, >12% DE fat)
Recurrent Exertional Rhabdomyolysis:
- Evidence type: Indirect, mechanistically speculative
- Recommendation level: Similar to PSSM; omega-3-rich fats can be incorporated into the standard low-NSC, high-fat diet but should not be positioned as a unique therapeutic agent
Equine Protozoal Myeloencephalitis Recovery:
- Evidence type: Mechanistically speculative
- Recommendation level: Omega-3 supplementation may provide neuroprotective adjunctive support but should not delay or substitute for FDA-approved antiprotozoal therapy; consider in horses with residual neurological deficits post-treatment
Inflammatory Myopathies:
- Evidence type: No equine data; human IIM data support anti-inflammatory potential
- Recommendation level: Omega-3 supplementation may be considered as an adjunctive agent alongside corticosteroids and immunosuppressive therapy, but evidence is entirely extrapolated from human studies
11.2 Practical Dosing Framework (Evidence-Based Extrapolation)
For horses where omega-3 supplementation is being considered:
Plant-Based (ALA-rich):
- Flaxseed oil: 0.37 g/kg BW/day (185 grams for 500 kg horse) or ~330 grams flaxseed meal daily, delivering ~207 mg ALA/kg BW/day
- Camelina oil: 0.37 g/kg BW/day (185 grams for 500 kg horse), delivering ~129 mg ALA/kg BW/day
- Duration: Minimum 12 weeks; optimal 16-20 weeks
- Acclimation: 4-week gradual introduction to minimize GI upset
Marine-Based (EPA/DHA-rich):
- Fish oil: Extrapolating from human therapeutic doses, 50-100 grams daily for a 500 kg horse would deliver approximately 2-4 grams combined EPA+DHA
- Duration: Minimum 8 weeks
- Formulation: Triglyceride form preferred over ethyl ester
Integration with PSSM/RER Diet:
- Use omega-3 source as the primary fat component (12-15% DE from omega-3 oil) rather than as an "additional supplement"
- Concurrent reduction in grain/refined carbohydrate and high-LA vegetable oil sources
- Maintain low-starch component (<5% DE) established for PSSM/RER
11.3 Monitoring Recommendations
In the absence of direct equine biomarker data, monitor:
- Clinical response: Frequency of clinical signs (muscle pain, stiffness, rhabdomyolysis episodes), resting and post-exercise CK (though CK alone cannot demonstrate omega-3 efficacy without a control group)
- Tolerability: Body condition, coat quality, GI function, performance during exercise
- Indirect markers: Reduction in medication requirements if used adjunctively; improvement in neurological deficits if used in EPM recovery
A structured trial approach (baseline 4-week observation, 12-16 weeks supplementation, 4-week washout) would provide stronger clinical evidence than ad hoc supplementation.
11.4 Cost-Benefit Considerations
Flaxseed and camelina oils are relatively inexpensive (~$0.50-1.00/day at 500 kg body weight) and provide caloric energy equivalent to non-omega-3 fats. If omega-3s are being used primarily to satisfy the "high-fat" component of PSSM/RER diets, the added cost is modest. Fish oil is considerably re expensive (~$2-5/day) and requires stronger evidence of efficacy before widespread recommendation. Algae-derived DHA/EPA supplements occupy an intermediate cost range but lack equine-specific safety or efficacy data.
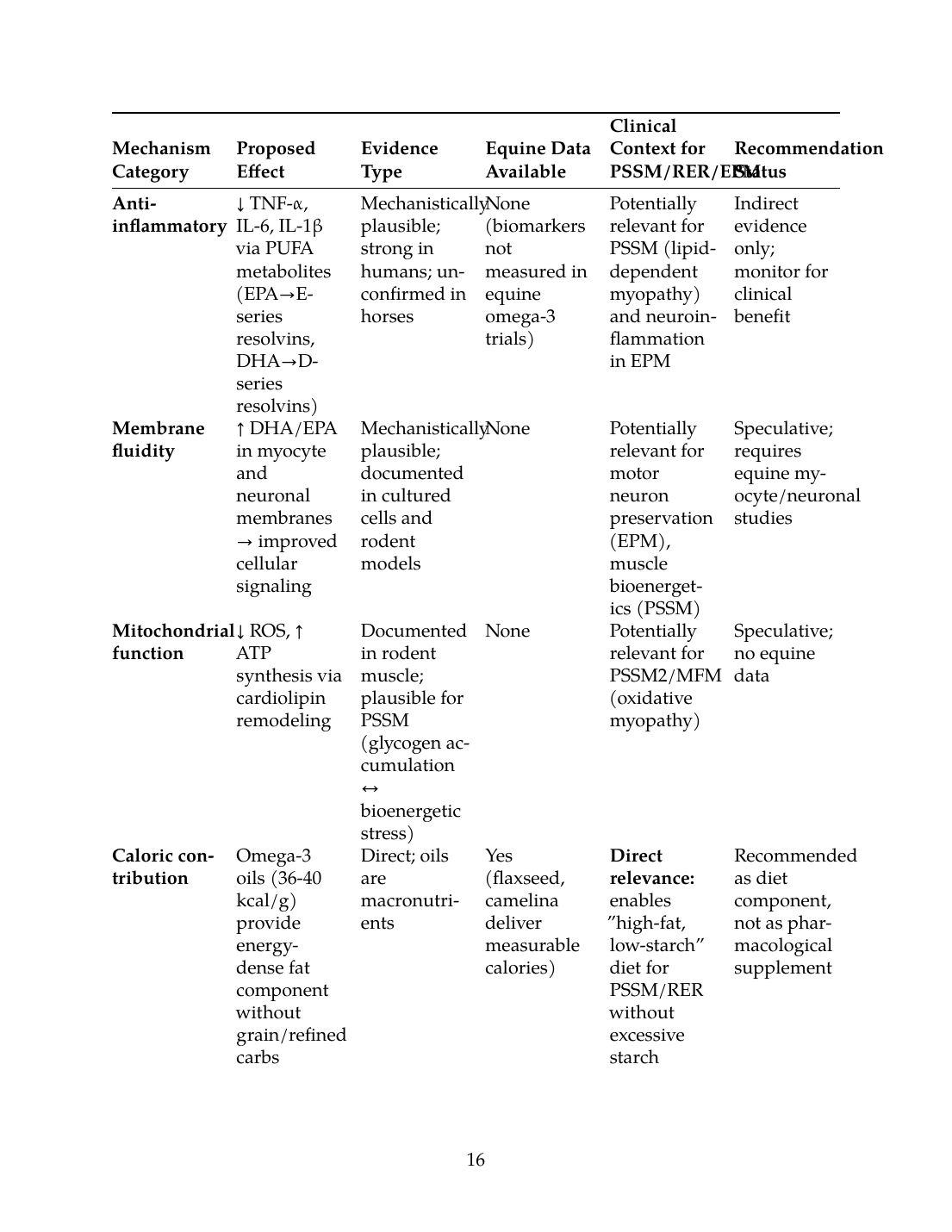
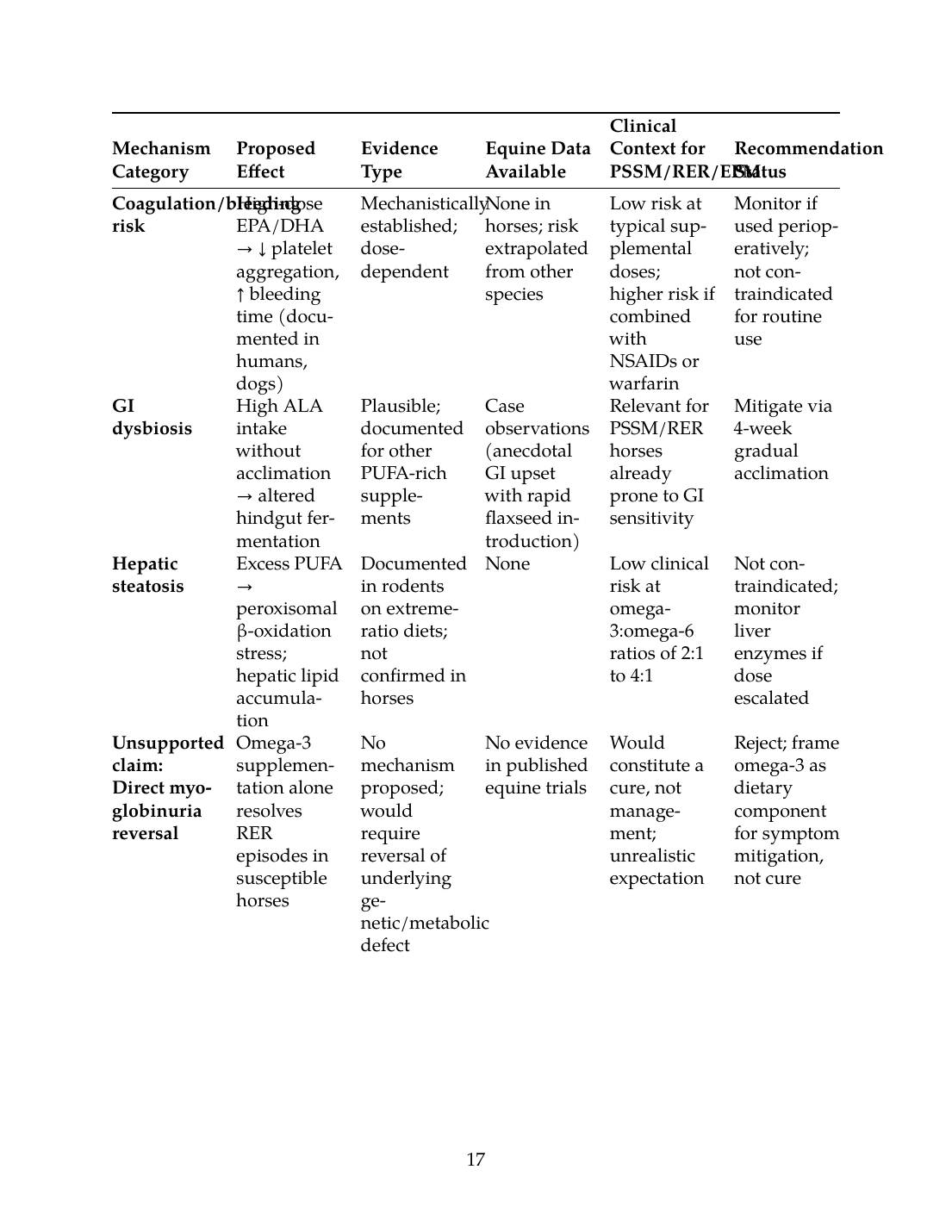
12. Summary Table: Mechanisms, Evidence Quality, and Clinical Context
The following table synthesizes omega-3 effects in equine neuromuscular disease, distinguishing direct mechanistic effects, indirect systemic effects, caloric contributions, and speculative claims:
| Mechanism Category | Proposed Effect | Evidence Type | Equine Data Available | Clinical Context for PSSM/RER/EPM | Recommendation Status |
|---|---|---|---|---|---|
| Anti-inflammatory | ↓ TNF-α, IL-6, IL-1β via PUFA metabolites (EPA→E-series resolvins, DHA→D-series resolvins) | Mechanistically plausible; strong in humans; unconfirmed in horses | None (biomarkers not measured in equine omega-3 trials) | Potentially relevant for PSSM (lipid-dependent myopathy) and neuroinflammation in EPM | Indirect evidence only; monitor for clinical benefit |
| Membrane fluidity | ↑ DHA/EPA in myocyte and neuronal membranes → improved cellular signaling | Mechanistically plausible; documented in cultured cells and rodent models | None | Potentially relevant for motor neuron preservation (EPM), muscle bioenergetics (PSSM) | Speculative; requires equine myocyte/neuronal studies |
| Mitochondrial function | ↓ ROS, ↑ ATP synthesis via cardiolipin remodeling | Documented in rodent muscle; plausible for PSSM (glycogen accumulation ↔ bioenergetic stress) | None | Potentially relevant for PSSM2/MFM (oxidative myopathy) | Speculative; no equine data |
| Caloric contribution | Omega-3 oils (36-40 kcal/g) provide energy-dense fat component without grain/refined carbs | Direct; oils are macronutrients | Yes (flaxseed, camelina deliver measurable calories) | Direct relevance: enables "high-fat, low-starch" diet for PSSM/RER without excessive starch | Recommended as diet component, not as pharmacological supplement |
| Coagulation/bleeding risk | High-dose EPA/DHA → ↓ platelet aggregation, ↑ bleeding time (documented in humans, dogs) | Mechanistically established; dose-dependent | None in horses; risk extrapolated from other species | Low risk at typical supplemental doses; higher risk if combined with NSAIDs or warfarin | Monitor if used perioperatively; not contraindicated for routine use |
| GI dysbiosis | High ALA intake without acclimation → altered hindgut fermentation | Plausible; documented for other PUFA-rich supplements | Case observations (anecdotal GI upset with rapid flaxseed introduction) | Relevant for PSSM/RER horses already prone to GI sensitivity | Mitigate via 4-week gradual acclimation |
| Hepatic steatosis | Excess PUFA → peroxisomal β-oxidation stress; hepatic lipid accumulation | Documented in rodents on extreme-ratio diets; not confirmed in horses | None | Low clinical risk at omega-3:omega-6 ratios of 2:1 to 4:1 | Not contraindicated; monitor liver enzymes if dose escalated |
| Unsupported claim: Direct myoglobinuria reversal | Omega-3 supplementation alone resolves RER episodes in susceptible horses | No mechanism proposed; would require reversal of underlying genetic/metabolic defect | No evidence in published equine trials | Would constitute a cure, not management; unrealistic expectation | Reject; frame omega-3 as dietary component for symptom mitigation, not cure |
| Unsupported claim: EPM spirochete killing | DHA/EPA or ALA exert antimicrobial effects against Sarcocystis neurona | Mechanistically implausible at physiologic concentrations; no in vitro data | None | EPM requires antiprotozoal therapy; omega-3 cannot replace monesin, doxycycline, or ponazuril | Reject; omega-3 only adjunctive for neuroinflammation post-treatment |
13. Conclusions and Future Research Priorities
13.1 Current State of Evidence
Omega-3 fatty acid supplementation in equine neuromuscular disease remains primarily evidence-based at the level of mechanistic plausibility and extrapolation from human and rodent studies, rather than direct equine efficacy trials. The strongest direct evidence supports omega-3-rich oils (flaxseed, camelina, fish oil) as caloric, energy-dense components of PSSM/RER management diets, enabling horses to meet energy requirements without exceeding safe starch and sugar thresholds. This is a direct dietary effect, not a pharmacological effect.
Anti-inflammatory, membrane-stabilizing, and mitochondrial benefits remain mechanistically speculative in horses due to the absence of equine-specific biomarker studies, imaging studies, or controlled trials measuring inflammatory cytokines, phospholipid composition, or muscle bioenergetics before and after omega-3 supplementation. Claims of omega-3 efficacy in EPM recovery are similarly indirect, predicated on the assumption that post-treatment neuroinflammation is limiting recovery and that EPA/DHA-derived resolvins will reduce it—an assumption without equine validation.
13.2 Critical Research Gaps
Priority 1: Controlled equine trials in PSSM/RER with biomarker endpoints
- Randomized, double-blinded crossover or parallel-arm design
- Primary endpoints: muscle pain (lameness score, resting CK), exercise tolerance (standardized submaximal exercise test with lactate and CK measurement)
- Secondary endpoints: inflammatory cytokines (TNF-α, IL-6, IL-1β), fatty acid composition of muscle phospholipids (gas chromatography), myocyte calcium handling (if tissue samples available)
- Duration: 16 weeks supplementation + 4-week washout
- Sample size: minimum 20-30 horses per group to detect clinically meaningful changes in CK or lameness
Priority 2: Marine-derived omega-3 efficacy and dose-ranging studies
- Fish oil or algae-DHA/EPA dose-ranging (e.g., 0, 25, 50, 100 grams daily for a 500 kg horse) to establish therapeutic window and safety margins
- Endpoints: serum and erythrocyte EPA/DHA incorporation, inflammatory markers, muscle biopsy phospholipid remodeling, clinical response
- Comparative efficacy: flaxseed (ALA-rich) vs. fish oil (EPA/DHA-rich) in the same population
Priority 3: EPM recovery adjunctive therapy trial
- Horses diagnosed with EPM (PCR+, clinical signs), treated with standard antiprotozoal (monesin ± doxycycline ± ponazuril)
- Randomized allocation: omega-3 supplementation vs. standard diet alone during 8-12 week recovery
- Endpoints: neurological deficit resolution (ataxia score, strength assessment), return to rideability, inflammatory CSF markers if lumbar puncture performed
Priority 4: Mechanistic studies in equine tissue models
- Isolated equine myocytes or primary neuronal cultures supplemented with EPA/DHA in vitro; measure ROS production, calcium handling, mitochondrial membrane potential, fatty acid incorporation into phospholipids
- Muscle biopsy phospholipid profiling before and after omega-3 supplementation in vivo, correlating changes with clinical response
13.3 Interim Clinical Recommendations
Until controlled equine data are available:
- For PSSM/RER management: Incorporate omega-3-rich oils (flaxseed meal 200-400 g/day, flaxseed oil 100-185 g/day, camelina oil 100-185 g/day, or fish oil 50-100 g/day) as the primary fat component of a low-starch (<5% DE), high-fat (12-15% DE) diet. This is a direct evidence-based recommendation because the oils deliver required calories and reduce reliance on starch/sugar.
- Dose and acclimation: Introduce over 4 weeks to minimize GI dysbiosis; target ALA ~200-250 mg/kg BW/day (flaxseed/camelina) or EPA+DHA ~4-8 mg/kg BW/day (fish oil) based on indirect evidence from human and rodent studies.
- Monitoring: Track resting CK, frequency of muscle pain or stiffness episodes, and post-exercise recovery as clinical markers. Do not expect omega-3 supplementation to "cure" PSSM or RER; frame it as a component of multimodal management (exercise, diet, medication) that may reduce episode frequency or severity.
- For EPM recovery: Use omega-3 supplementation adjunctively during the 8-12 week recovery window after antiprotozoal therapy initiation, with the goal of supporting neuroinflammation resolution. Do not use as a substitute for antiprotozoal treatment.
- Avoid excessive doses or extreme omega-3:omega-6 ratios: Maintain a 2:1 to 4:1 omega-3:omega-6 ratio to minimize risk of immunosuppression or hepatic lipid accumulation. Monitor liver enzymes if supplementation dose is escalated above recommended ranges.
- Perioperative considerations: Reduce or discontinue omega-3 supplementation 1-2 weeks before elective surgery to minimize bleeding risk, particularly if NSAIDs or other anticoagulants are concurrently administered.
13.4 Final Summary
Omega-3 supplementation in equine neuromuscular disease occupies a middle ground: caloric/dietary benefits are direct and evidence-based, while anti-inflammatory, neuroprotective, and mitochondrial benefits are mechanistically plausible but unproven in horses. Practitioners should distinguish between these categories when counseling clients. Fish oil and algae-derived supplements are increasingly promoted based on human and rodent data, but equine-specific efficacy and safety trials are lacking. Flaxseed and camelina remain practical, cost-effective options for achieving the "high-fat, low-starch" diet mandated by PSSM/RER physiology. Future research should prioritize controlled equine trials with biomarker endpoints, dose-ranging studies, and mechanistic investigation in equine cell culture and tissue models to clarify whether omega-3 supplementation provides benefits beyond its caloric contribution to diet composition.
References
References
Numbered in order of first citation, IEEE style. Each entry links to the original source via DOI or publisher URL.
- S. Valberg, Z. J. Williams, C. Finno, A. Schultz, D. Velez-Irizarry, M. Henry, K. Gardner, and J. Petersen, “Type 2 polysaccharide storage myopathy in Quarter Horses is a novel glycogen storage disease causing exertional rhabdomyolysis.,” Equine Veterinary Journal, Aug. 2022. doi: 10.1111/evj.13876. View source
- W. P. Ribeiro, S. Valberg, J. D. Pagan, and B. Gustavsson, “The effect of varying dietary starch and fat content on serum creatine kinase activity and substrate availability in equine polysaccharide storage myopathy.,” Journal of Veterinary Internal Medicine, Nov. 2004. doi: 10.1111/j.1939-1676.2004.tb02637.x. View source
- R. Geor, “Role of dietary energy source in the expression of chronic exertional myopathies in horses,” Journal of Animal Science, Jun. 2005. doi: 10.2527/2005.8313_SUPPLE32X. View source
- C. Finno, E. McKenzie, S. Valberg, and J. D. Pagan, “Effect of fitness on glucose, insulin and cortisol responses to diets varying in starch and fat content in Thoroughbred horses with recurrent exertional rhabdomyolysis.,” Equine veterinary journal. Supplement, Nov. 2010. doi: 10.1111/j.2042-3306.2010.00199.x. View source
- R. MacKay and D. K. Howe, “Equine Protozoal Myeloencephalitis.,” The Veterinary clinics of North America. Equine practice, Jul. 2022. doi: 10.1016/j.cveq.2022.05.003. View source
- S. Reed, S. Reed, M. Furr, D. Howe, A. L. Johnson, R. MacKay, J. Morrow, N. Pusterla, and S. Witonsky, “Equine Protozoal Myeloencephalitis: An Updated Consensus Statement with a Focus on Parasite Biology, Diagnosis, Treatment, and Prevention,” Journal of Veterinary Internal Medicine, Feb. 2016. doi: 10.1111/jvim.13834. View source
- L. Helber, B. Wagner, C. M. Leeth, T. LeRoith, T. E. Cecere, K. K. Lahmers, F. M. Andrews, A. N. Hay, S. R. Werre, A. Johnson, C. Clark, N. Pusterla, S. Reed, D. S. Lindsay, S. D. Taylor, K. Estell, M. Furr, R. J. Mackay, F. D. Piero, M. Carossino, K. Pandaleon, S. Weatherford, R. Ramirez-Barrios, K. Zimmerman, and S. Witonsky, “Persistence of Sarcocystis neurona and histopathology in horses with equine protozoal myeloencephalitis,” Frontiers in Veterinary Science, Apr. 2026. doi: 10.3389/fvets.2026.1787994. View source
- S. Hartwig, S. Burron, T. Richards, A. Rankovic, D. Ma, W. Pearson, J. L. Ellis, L. Trevizan, D. J. Seymour, and A. Shoveller, “The effect of dietary camelina, flaxseed, and canola oil supplementation on skin fatty acid profile and immune and inflammatory responses in healthy adult horses,” Journal of Animal Science, Feb. 2025. doi: 10.1093/jas/skaf025. View source
- S. Burron, T. Richards, T. C. McCorkell, L. Trevizan, D. Puttick, D. Ma, W. Pearson, and A. Shoveller, “Effects of dietary camelina, flaxseed, and canola oil supplementation on plasma fatty acid concentrations and health parameters in horses.,” Animal, Nov. 2023. doi: 10.1016/j.animal.2023.101034. View source
- S. Burron, T. Richards, G. Krebs, L. Trevizan, A. Rankovic, S. Hartwig, W. Pearson, D. Ma, and A. Shoveller, “The balance of n-6 and n-3 fatty acids in canine, feline, and equine nutrition: exploring sources and the significance of alpha-linolenic acid,” Journal of Animal Science, May 2024. doi: 10.1093/jas/skae143. View source
- M. Plourde and S. Cunnane, “Extremely limited synthesis of long chain polyunsaturates in adults: implications for their dietary essentiality and use as supplements.,” Applied physiology, nutrition, and metabolism = Physiologie appliquee, nutrition et metabolisme, Jun. 2007. doi: 10.1139/H07-034. View source
- K. Harnack, G. Andersen, and V. Somoza, “Quantitation of alpha-linolenic acid elongation to eicosapentaenoic and docosahexaenoic acid as affected by the ratio of n6/n3 fatty acids,” Nutrition and Metabolism, Feb. 2009. doi: 10.1186/1743-7075-6-8. View source
- T. Greupner, L. Kutzner, S. Pagenkopf, H. Kohrs, A. Hahn, N. Schebb, and J. Schuchardt, “Effects of a low and a high dietary LA/ALA ratio on long-chain PUFA concentrations in red blood cells.,” Food & Function, Sep. 2018. doi: 10.1039/c8fo00735g. View source
- B. Miralles-Pérez, L. Méndez, M. Nogués, V. Sánchez-Martos, À. Fortuño-Mar, S. Ramos‐Romero, M. Hereu, I. Medina, and M. Romeu, “Effects of a Fish Oil Rich in Docosahexaenoic Acid on Cardiometabolic Risk Factors and Oxidative Stress in Healthy Rats,” Marine Drugs, Sep. 2021. doi: 10.3390/md19100555. View source
- M. Dempsey, M. Rockwell, and L. M. Wentz, “The influence of dietary and supplemental omega-3 fatty acids on the omega-3 index: A scoping review,” Frontiers in Nutrition, Jan. 2023. doi: 10.3389/fnut.2023.1072653. View source
- J. Matos, C. Afonso, C. Cardoso, M. Serralheiro, and N. Bandarra, “Yogurt Enriched with Isochrysis galbana: An Innovative Functional Food,” Foods, Jun. 2021. doi: 10.3390/foods10071458. View source
- A. Simopoulos, “Human requirement for N-3 polyunsaturated fatty acids.,” Poultry Science, Jul. 2000. doi: 10.1093/PS/79.7.961. View source
- T. Richards, S. Burron, T. C. McCorkell, L. Trevizan, K. Patterson, D. Minikhiem, D. Ma, W. Pearson, and A. Shoveller, “Effects of dietary camelina, flaxseed, and canola oil supplementation on transepidermal water loss, skin and coat health parameters, and plasma prostaglandin E2, glycosaminoglycan, and nitric oxide concentrations in healthy adult horses,” Journal of Animal Science, Nov. 2023. doi: 10.1093/jas/skad373. View source
- D. Jerab, F. Blangero, P. C. T. D. Costa, J. L. de Brito Alves, R. Kefi, H. Jamoussi, B. Morio, and A. Eljaafari, “Beneficial Effects of Omega-3 Fatty Acids on Obesity and Related Metabolic and Chronic Inflammatory Diseases,” Nutrients, Apr. 2025. doi: 10.3390/nu17071253. View source
- S. Jannas-Vela, A. Espinosa, A. A. Candia, M. Flores-Opazo, L. Peñailillo, and R. Valenzuela, “The Role of Omega-3 Polyunsaturated Fatty Acids and Their Lipid Mediators on Skeletal Muscle Regeneration: A Narrative Review,” Nutrients, Feb. 2023. doi: 10.3390/nu15040871. View source
- N. Rohwer, J. Jelleschitz, A. Höhn, D. Weber, A. Kühl, C. Wang, R. Ohno, N. Kampschulte, A. Pietzner, N. Schebb, K. Weylandt, and T. Grune, “Prevention of colitis-induced liver oxidative stress and inflammation in a transgenic mouse model with increased omega-3 polyunsaturated fatty acids,” Redox Biology, Jun. 2023. doi: 10.1016/j.redox.2023.102803. View source
- M. Rodríguez-Cruz, O. D. R. Cruz-Guzmán, T. Almeida‐Becerril, A. D. Solís-Serna, S. Atilano-Miguel, J. Sánchez-González, L. Barbosa-Cortés, E. Ruíz-Cruz, J. C. Huicochea, A. Cárdenas-Conejo, R. E. Escobar-Cedillo, C. A. Yam-Ontiveros, and E. F. Ricárdez-Marcial, “Potential therapeutic impact of omega-3 long chain-polyunsaturated fatty acids on inflammation markers in Duchenne muscular dystrophy: A double-blind, controlled randomized trial.,” Clinical Nutrition, Sep. 2017. doi: 10.1016/j.clnu.2017.09.011. View source
- K. Fresa, G. Catandi, R. Gonzalez-Castro, A. K. Omar, L. A. Whitcomb, M. Cheng, T. W. Chen, E. Carnevale, and A. J. Chicco, “Impact of dietary essential fatty acids on phospholipid composition and mitochondrial function in aged mares,” Scientific Reports, Dec. 2025. doi: 10.1038/s41598-025-03271-6. View source
- R. Putti, R. Sica, V. Migliaccio, and L. Lionetti, “Diet impact on mitochondrial bioenergetics and dynamics,” Frontiers in Physiology, Apr. 2015. doi: 10.3389/fphys.2015.00109. View source
- G. Cavaliere, G. Trinchese, P. Bergamo, C. De Filippo, G. Mattace Raso, G. Gifuni, R. Putti, B. H. Moni, R. B. Canani, R. Meli, and M. Mollica, “Polyunsaturated Fatty Acids Attenuate Diet Induced Obesity and Insulin Resistance, Modulating Mitochondrial Respiratory Uncoupling in Rat Skeletal Muscle,” PLoS ONE, Feb. 2016. doi: 10.1371/journal.pone.0149033. View source
- P. Isesele and V. Mazurak, “Regulation of Skeletal Muscle Satellite Cell Differentiation by Omega-3 Polyunsaturated Fatty Acids: A Critical Review,” Frontiers in Physiology, Jun. 2021. doi: 10.3389/fphys.2021.682091. View source
- F. Carotenuto, A. Costa, M. C. Albertini, M. Rocchi, A. Rudov, D. Coletti, M. Minieri, P. di Nardo, and L. Teodori, “Dietary Flaxseed Mitigates Impaired Skeletal Muscle Regeneration: in Vivo, in Vitro and in Silico Studies,” International Journal of Medical Sciences, Feb. 2016. doi: 10.7150/ijms.13268. View source
- B. Tachtsis, J. Whitfield, J. Hawley, and N. J. Hoffman, “Omega-3 Polyunsaturated Fatty Acids Mitigate Palmitate-Induced Impairments in Skeletal Muscle Cell Viability and Differentiation,” Frontiers in Physiology, Jun. 2020. doi: 10.3389/fphys.2020.00563. View source
- C. J. Pufahl, S. E. Smart, J. Bureau, B. Ross, C. A. Simpson, A. Metherel, J. R. Dick, and C. McGlory, “The influence of biological sex on skeletal muscle phospholipid membrane composition in response to omega n-3 polyunsaturated fatty acid supplementation and washout in humans.,” Prostaglandins, Leukotrienes and Essential Fatty Acids, Jun. 2025. doi: 10.1016/j.plefa.2025.102696. View source
- M. Tremblay, I. Zhang, K. Bisht, J. C. Savage, C. Lecours, M. Parent, V. Titorenko, and D. Maysinger, “Remodeling of lipid bodies by docosahexaenoic acid in activated microglial cells,” Journal of Neuroinflammation, May 2016. doi: 10.1186/s12974-016-0580-0. View source
- D. Fernández-Lázaro, S. Arribalzaga, E. Gutiérrez-Abejón, M. Azarbayjani, J. Mielgo-Ayuso, and E. Roche, “Omega-3 Fatty Acid Supplementation on Post-Exercise Inflammation, Muscle Damage, Oxidative Response, and Sports Performance in Physically Healthy Adults—A Systematic Review of Randomized Controlled Trials,” Nutrients, Jun. 2024. doi: 10.3390/nu16132044. View source
- C. Njoku, W. Saville, S. Reed, M. Oglesbee, P. Rajala-Schultz, and R. W. Stich, “Reduced Levels of Nitric Oxide Metabolites in Cerebrospinal Fluid Are Associated with Equine Protozoal Myeloencephalitis,” Clinical and Vaccine Immunology, May 2002. doi: 10.1128/CDLI.9.3.605-610.2002. View source
- C. J. Angwin, I. D. A. Rocha, S. Reed, J. K. Morrow, A. Graves, and D. K. Howe, “Analysis of IgG responses to Sarcocystis neurona in horses with equine protozoal myeloencephalitis (EPM) suggests a Th1-biased immune response.,” Veterinary Immunology and Immunopathology, Sep. 2025. doi: 10.1016/j.vetimm.2025.111009. View source
- M. Lagarde, M. Guichardant, N. Bernoud-Hubac, C. Calzada, and E. Véricel, “Oxygenation of polyunsaturated fatty acids and oxidative stress within blood platelets.,” Biochimica et Biophysica Acta - Molecular and Cell Biology of Lipids, Jun. 2018. doi: 10.1016/j.bbalip.2018.03.005. View source
- B. Eggers, J. Stepien, A. Reker, S. Esser, K. Pfeiffer, M. Pawlas, K. Barkovits, and K. Marcus, “The Protective Effect of Docosahexaenoic Acid on Mitochondria in SH-SY5Y Model of Rotenone-Induced Toxicity,” Metabolites, Jan. 2025. doi: 10.3390/metabo15010029. View source
- E. Çelik, P. Karabacak, M. Özcan, I. Ilhan, M. Y. Tepebaşı, E. Sarman, M. Arlıoğlu, and H. Aşçı, “Targeting mitochondrial apoptosis in septic lung injury: protective potential of omega-3 fatty acids,” Apoptosis, Mar. 2026. doi: 10.1007/s10495-026-02325-y. View source
- A. I. Sánchez-Rosales, J. G. Posadas-Calleja, A. E. Serralde-Zúñiga, and G. Quiroz-Olguín, “Nutritional interventions as modulators of the disease activity for idiopathic inflammatory myopathies: a scoping review.,” Journal of human nutrition and dietetics (Print), Feb. 2024. doi: 10.1111/jhn.13287. View source
- J. A. Spencer, P. DeInnocentes, E. M. Moyana, A. Guarino, S. E. Ellison, R. Curtis Bird, and B. L. Blagburn, “Cytokine Gene Expression in Response to SnSAG1 in Horses with Equine Protozoal Myeloencephalitis,” Clinical Diagnostic Laboratory Immunology, May 2005. doi: 10.1128/CDLI.12.5.644-646.2005. View source
This review is one of 34 in the Foxwatch research corpus. Methodology: PRISMA 2020-style systematic literature review against PubMed, CAB Abstracts, Scopus, Web of Science, and equine veterinary conference proceedings.