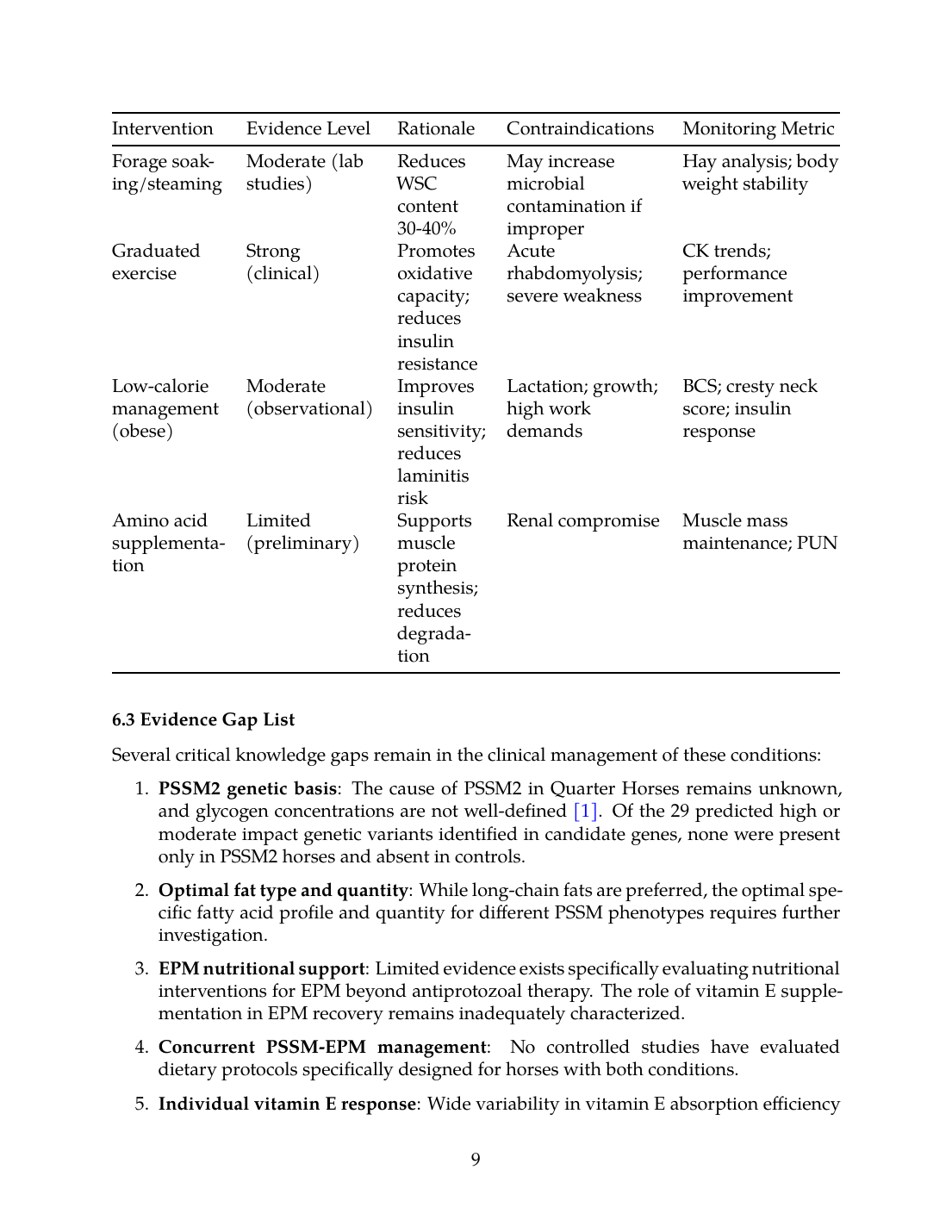
Clinical decision framework for feeding PSSM, EPM, or both
Pulls all 34 reviews into one decision tree. Confirmed PSSM Type 1, suspected PSSM2, confirmed EPM, suspected EPM, overlapping signs, uncertain diagnosis: each branch has its own diagnostic, dietary, and monitoring path. Veterinarians and barn managers building a feeding plan against a real diagnosis use this review as the master flow. Cross-references every other review on the site.
Research thesis
This evidence-based literature review argues that feeding decisions for horses with PSSM, EPM, or both should follow a single integrated decision framework, not two parallel disease-specific protocols, not only because diagnostic uncertainty (confirmed PSSM1, suspected PSSM2 or MFM, confirmed EPM, suspected EPM, overlapping signs) is the dominant clinical reality, but also because forage testing, NSC control, calorie strategy, fat use, protein and amino acid adequacy, vitamin E and selenium status, trace mineral balancing, body-condition management, and rehabilitation coordination interact with each other in ways single-disease guidance does not capture; by integrating the systematic-review evidence base across diagnostic, dietary, and monitoring domains and by treating the obese easy keeper, the underweight hard keeper, the senior horse, the performance horse, and the neurologic horse as separate cases, this review builds a step-by-step clinical decision tree with stated evidence levels, contraindications, and monitoring metrics.
Evidence visualizations & full pages
Evidence figures (2)
Figures referenced by the review's Figure N: captions. Click any chart for a full-size view. They are also rendered inline below their respective captions in the full review text further down.
Original review pages (PDF render)
Download
Full review text
Clinical decision framework for feeding PSSM, EPM, or both
Clinical Decision Framework for Feeding Horses with PSSM, EPM, or Both: An Evidence-Based Literature Review
1. Introduction and Diagnostic Clarification
1.1 Overview of Diagnostic Categories
The clinical management of horses presenting with muscle and/or neurologic signs requires precise diagnostic clarification before implementing dietary interventions. Polysaccharide storage myopathy (PSSM) encompasses two distinct conditions: Type 1 PSSM (PSSM1), a glycogen storage disorder caused by a dominant gain-of-function mutation (R309H) in the glycogen synthase 1 gene (GYS1), and Type 2 PSSM (PSSM2), characterized by histopathologic abnormalities without the GYS1 mutation [1]. Equine protozoal myeloencephalitis (EPM) remains one of the most frequently diagnosed neurological disorders of horses in North America, caused primarily by Sarcocystis neurona and less commonly Neospora hughesi [2]. The clinical presentation of EPM is highly variable due to multifocal involvement of both grey and white matter, with asymmetric ataxia and weakness being most common.
The genetic basis of PSSM1 is now well-established, with the GYS1 mutation resulting in increased insulin sensitivity, excessive muscle glycogen storage, and accumulation of amylase-resistant polysaccharide in muscle [3]. Testing has revealed significant breed variation in mutation prevalence, with Percheron horses showing up to 90% carrier rates, Russian heavy draft horses at 49.9%, and Soviet heavy draft horses at 30% [4]. A third of horses presenting with muscle disease at neuromuscular laboratories have alleles that explain their condition, constituting 45% of horses with confirmed histological myopathic processes [5]. Importantly, diagnostic confirmation of EPM remains challenging; paired serum and cerebrospinal fluid analysis is most reliable, while PCR lacks sensitivity [2].
1.2 Distinguishing PSSM1, PSSM2/MFM, and EPM
PSSM2 in Quarter Horses represents a novel glycogen storage disorder characterized by exertional rhabdomyolysis occurring predominantly in barrel racing and working cow/roping performance types [1]. Unlike PSSM1, PSSM2 shows intermediate histopathological scores including amounts of amylase-resistant polysaccharide and glycogen concentrations that differ significantly among diagnostic groups. In Warmblood horses with PSSM2, abnormal exercise responses reported by owners began at approximately 6 years of age and included a decline in performance, reluctance to collect, and reluctance to go forward in over 50% of horses [6]. The same diet and exercise regime prescribed for PSSM1 is recommended for PSSM2; however, the benefit of these recommendations for PSSM2 remains incompletely documented.
EPM can be caused by either of two related protozoan parasites, with S. neurona being the most common etiologic pathogen [7]. Horses are commonly infected, but clinical disease occurs only infrequently; the factors influencing disease occurrence are not well understood. Epidemiological studies have identified risk factors for EPM development, including the presence of opossums and prior stressful health-related events. Molecular detection using real-time PCR on cerebrospinal fluid from horses with suspected neurological disease has shown that 25 horses tested positive for S. neurona by PCR only, while 30 horses had intrathecally-derived antibodies only, and 13 horses tested positive by both methods [8].
2. Core Dietary Interventions
2.1 Forage Testing and NSC Control
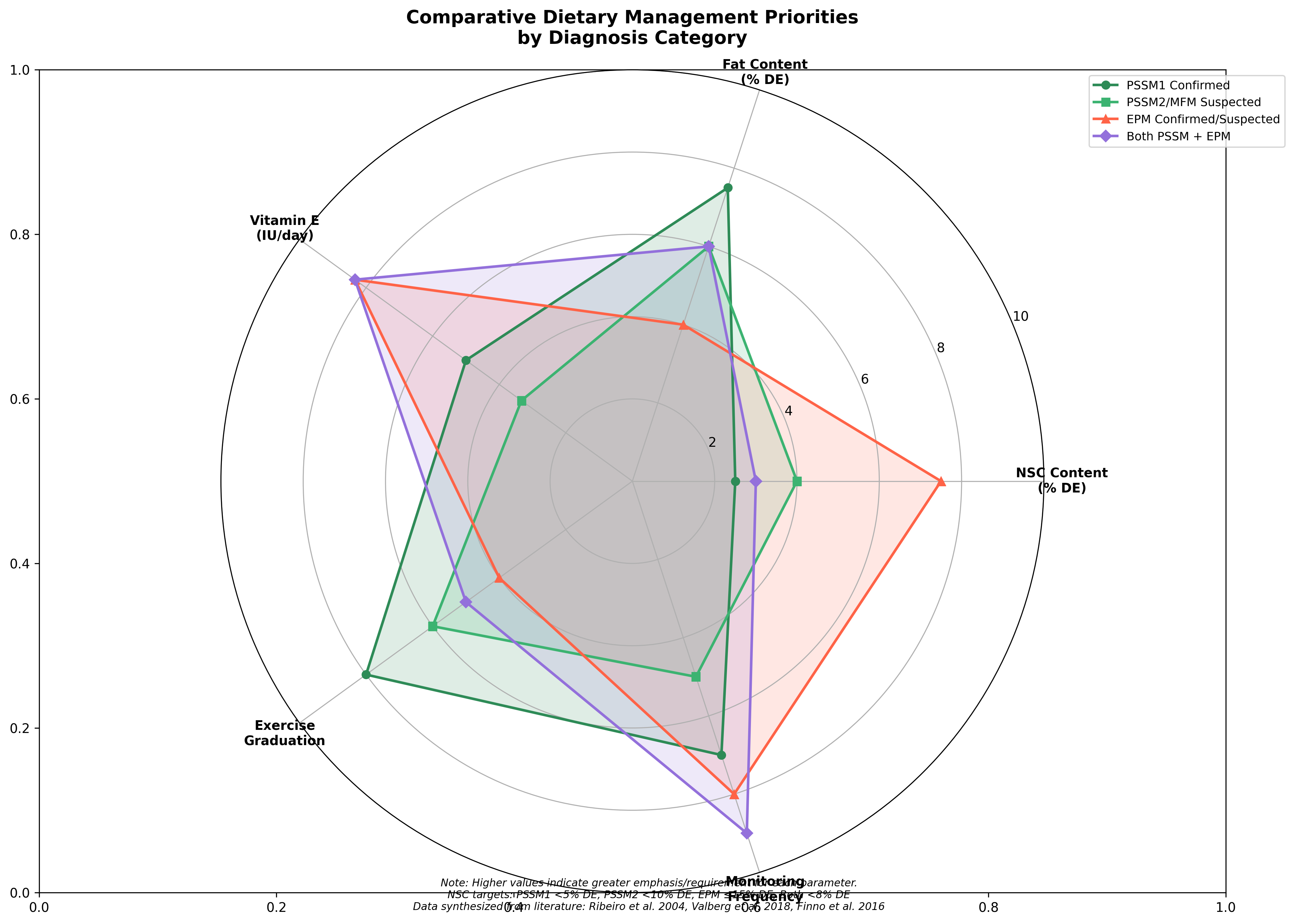
The foundation of dietary management for PSSM begins with controlling non-structural carbohydrate (NSC) intake. For horses with clinical manifestations of PSSM, a diet with <5% digestible energy (DE) from starch and >12% DE from fat can reduce exertional rhabdomyolysis, potentially by increasing availability of free fatty acids for muscle metabolism [9]. Postprandial insulin and glucose responses are significantly higher with high-starch diets compared to low-starch alternatives. The feeding of concentrates rich in hydrolyzable carbohydrates may enhance PSSM disease expression by increasing the quantity of glucose available for muscle glycogen synthesis [3].
Forage processing methods significantly impact NSC content. Soaking hay at 16°C for 9 hours and steaming effectively reduce water-soluble carbohydrates, with mean content dropping from 126 g/kg DM in dry hay to 79-83 g/kg DM after treatment [10]. The most effective method for reducing carbohydrates while maintaining hygienic quality involves steaming followed by soaking. For horses requiring strict NSC control, forage analysis should be performed to ensure NSC content remains below 10-12% on a dry matter basis. The fecal microbiota of grazing horses shows distinct shifts in response to different forages, with shifts most influenced by forage non-structural carbohydrates and crude protein rather than fiber content [11].
2.2 Fat Supplementation Strategy
Fat supplementation represents a cornerstone of PSSM dietary management. Four isocaloric diets ranging from 21.2% to 3.9% DE from starch demonstrated that log CK activity was significantly higher with diets containing >5% starch compared to the lowest-starch option [9]. Muscle oxidative capacity and lipid stores were found to be low in PSSM horses, while muscle glycogen and abnormal polysaccharide content remained high regardless of dietary starch level. A decrease in the frequency and severity of exertional rhabdomyolysis has been observed when horses with PSSM are fed diets with reduced DE from hydrolyzable carbohydrates (<10 to 15% of total diet) and increased DE from fat (15 to 20%) and other energy sources such as beet pulp and soybean hulls [3].
The type of fat matters clinically. Triheptanoin, an odd-chain fatty acid triglyceride, demonstrated detrimental effects in PSSM horses, reflecting decreased availability of non-esterified fatty acids, increased insulin stimulation of glycogen synthesis, and potential inhibition of lipid oxidation [12]. Long-chain fats remain the best dietetic choice for PSSM management. Rice bran oil and flaxseed oil supplementation in training horses has shown potential for reducing training-related increases in interleukin-1β and exercise-induced creatine kinase, with omega-3 fatty acid inclusion demonstrating anti-inflammatory benefits [13]. In endurance horses, mitochondrial β-oxidation increases approximately eight-fold during long-distance exercise, highlighting the importance of adequate fat adaptation for sustained performance [14].
2.3 Protein and Amino Acid Requirements
Protein and amino acid adequacy is essential for maintaining muscle mass and supporting recovery, particularly in horses with myopathies. Studies examining markers of whole-body and muscle protein metabolism in aged horses found that amino acid availability was not limiting protein synthesis in sedentary aged horses fed typical North American diets [15]. However, atrogin-1 abundance tended to be higher in the post-absorptive state for control treatments, indicating that amino acid supplementation resulted in less muscle protein degradation. For horses with pituitary pars intermedia dysfunction (PPID), appropriate nutritional management highlights the importance of providing adequate amounts and forms of energy, carbohydrate, and protein, with further practical considerations regarding feeding animals that are often aged and may have particular clinical sequelae [16].
The challenge in establishing protein requirements for senior horses relates to age-related loss of lean body mass, or sarcopenia. Lean body mass serves as a protein reservoir and provides amino acids used continuously for endogenous protein synthesis, while dietary protein maintains or restores this reservoir [17]. Loss of lean body mass is associated with greater morbidity and mortality, making preservation of lean body mass an important goal. For horses with myopathies, quality protein sources providing adequate lysine and threonine become particularly important for muscle maintenance and repair.
3. Micronutrient Management
3.1 Vitamin E Testing and Supplementation
Vitamin E status is critically important in both PSSM and EPM management, as well as in preventing other neuromuscular conditions. A vitamin E-deficient myopathy characterized histologically by a moth-eaten appearance in the mitochondria and anguloid myofiber atrophy has been found in horses with clinical signs of equine motor neuron disease (EMND) that were highly responsive to vitamin E treatment [18]. In horses, examples of nutrigenomics applications include PSSM1, where low starch and high fat diets are recommended to prevent episodes of rhabdomyolysis, and conditions where personalized nutrition tailors nutrition advice based on genetic makeup [19].
There is a wide range of individual response to vitamin E supplementation in horses. Some horses obtain very high serum vitamin E concentrations with minimal intake, whereas others require high doses of supplementation to remain in the normal range. Serum vitamin E concentrations show large fluctuations even in horses with deficient levels (<1.5 μg/mL), with the coefficient of variation being significantly larger in deficient horses (mean CV: 41%) compared to normal horses (14%) [20]. A single measurement does not always provide reliable information about the true vitamin E status; analyzing multiple samples allows for more accurate estimation. For horses affected by equine neuroaxonal dystrophy/degenerative myeloencephalopathy, increased α-tocopherol metabolism provides novel insight into alterations in vitamin E processing, highlighting the need for high-dose supplementation to prevent the clinical phenotype in genetically susceptible horses [21].
3.2 Selenium and Trace Mineral Considerations
Selenium deficiency has been implicated in various equine myopathies and plays a critical role in antioxidant defense. A study of recurrent exertional rhabdomyolysis in Transylvania found that horses in affected regions had significantly lower glutathione peroxidase levels than animals living in non-affected regions, with a strong correlation between erythrocyte selenium concentration and GSH-Px activity (r = 0.777, P < 0.001) [22]. Serum vitamin E concentrations were low independently of the origin of the horse, suggesting that both selenium and vitamin E deficiencies may contribute to the development of this enzootic syndrome.
Selenium is an essential trace element important for many physiological processes, especially the functions of immune and reproductive systems, metabolism of thyroid hormones, and antioxidant defense [23]. In foals, selenium deficiency manifests as white muscle disease (WMD), while selenium toxicity must also be carefully avoided given the narrow margin of safety. Certain regions have been identified as sufficiently selenium-deficient to predispose newborn foals to WMD, especially when fed diets consisting mainly of locally harvested roughage [24]. Results of studies evaluating risk factors for equine motor neuron disease suggest that lack of access to pasture, dietary deficiency of vitamin E, or excessive copper are likely risk factors [25].
Microelement deficiencies remain a current problem in horse breeding, causing various health complications. Studies of broodmares in southeastern Poland found zinc concentrations below reference ranges across all studied regions, with additional selenium and copper deficiencies identified in specific areas [26]. Despite identified deficiencies, mares gave birth on time with registered newborn foals, suggesting subclinical deficiency states may be more common than recognized. Trace element status and metabolic milieu are sometimes overlooked in common veterinary clinical practice, yet evaluation of requirements may be useful to prevent tissue-specific impairment [27].
4. Body Condition and Exercise Management
4.1 Body Condition Score Targets and Calorie Strategy
Equine metabolic syndrome represents a health problem affecting many horses considered "easy keepers," consisting of large regional fat deposits, obesity, insulin resistance or high fasting insulin concentrations, and if not controlled, recurrent episodes of laminitis [28]. Obese horses have become more the rule rather than the exception, and researchers have uncovered yet another reason to keep mature horses slim and conditioned. The prevalence of overconditioned and obese horses has been found to be higher than reported in previous studies, indicating that obesity might be an emerging problem [29].
Regular exercise is critical for metabolic health. The risk of insulin dysregulation was higher in horses with no exercise compared to horses with exercise (OR 7.6, 95% CI 1.2-49.3, P = 0.03) [30]. Neither oral sugar test results nor adiponectin varies with season; however, there are a substantial number of horses with variable insulin dysregulation status throughout the year, in which repeated testing may be beneficial. Lack of exercise was identified as a significant risk factor for insulin dysregulation. Ponies are especially known as "easy keepers" and are often affected by obesity and its related metabolic disorders, with the richness of the fecal microbiota decreasing in ponies during body weight gain [31].
Equine metabolic syndrome-affected horses benefit from interventions that improve insulin sensitivity and reduce systemic inflammation. Supplementation with bioactive compounds has shown potential for reducing baseline insulin and glucose levels, contributing to decreased adipose tissue inflammation, along with decreases in body weight and improvements in overall body condition scores and cresty neck scores [32]. A single bout of exercise, as well as chronic exercise conditioning, increases insulin sensitivity and glucose disposal via both contraction- and insulin-mediated uptake pathways [33].
4.2 Exercise Protocol Integration
For PSSM horses, exercise management is as important as dietary modification. With the recommended diet and exercise regime, 80% of PSSM2 Warmblood owners reported an overall improvement with significant decreases in the proportion of horses showing a decline in performance and rhabdomyolysis [6]. However, 53% of PSSM2 horses were still not advancing as expected, with reluctance to go forward and collect persisting in approximately one third of horses. Diet and exercise recommendations ideal for PSSM1 improve but do not eliminate the decline in performance and reluctance to go forward under saddle characteristic of PSSM2.
Rations high in grain and formulated to exceed daily energy requirements may increase episodes of rhabdomyolysis in Thoroughbred horses susceptible to recurrent exertional rhabdomyolysis [34]. Feeding low-carbohydrate, high-fat diets has been demonstrated to decrease the severity of exercise-induced muscle injury in horses with exertional rhabdomyolysis, with adaptation requiring time and serum CK activities being significantly lower than initial values after dietary change [35]. A high-energy, forage-only diet alters the metabolic response to exercise and, with the exception of lowered glycogen stores, appears to have positive rather than negative effects on performance traits [36].
5. Special Case Considerations
5.1 Obese Easy Keeper and Underweight Hard Keeper
The obese easy keeper with PSSM presents unique management challenges requiring careful caloric restriction while maintaining adequate nutrition. Horses should be fed low NSC feeds/feedstuffs as well as fresh or preserved forage with NSC content <10-12% on a dry matter basis [16]. For these horses, hay soaking or steaming becomes essential to reduce NSC content while still providing adequate forage volume. Exercise becomes particularly important for weight loss while maintaining muscle function.
The underweight hard keeper requires increased caloric density while maintaining low NSC content. Fat supplementation becomes the primary strategy for increasing calories without increasing glycemic load. For horses in negative energy balance, maintaining a positive anabolic hormonal balance while providing sufficient time to restore glycogen stores is important [37]. When horses consume a normal basal diet, neither extra carbohydrate nor fat will enhance glycogen stores, and may, especially in unadapted horses, produce undesirable effects.
5.2 Senior Horse and Performance Horse Considerations
Senior horses require special attention to protein quality and quantity, vitamin E status, and potential concurrent conditions such as PPID. Nutritional management for PPID proposes a stepwise approach to building an individual feeding plan, starting with considerations relating to current and desired body condition and muscle mass [16]. The next important step is to determine the degree of insulin dysregulation, as this is a major factor for increased risk of laminitis. Animals with insulin dysregulation should be fed low NSC feeds.
The energy requirements in dogs decrease with age, such that senior dogs need about 25% fewer calories on average than young adult dogs, and similar age-related changes likely occur in horses [17]. One challenge in establishing protein requirements for senior pets is age-related loss of lean body mass. While increasing protein intake can slow this loss, increasing protein intake alone cannot prevent loss of lean body mass.
Performance horses with PSSM require careful management of caloric intake relative to work demands while maintaining low NSC feeding principles. The single greatest nutrient need above maintenance is energy, and it appears that sufficient starch must be included in the diet to maintain normal muscle glycogen levels [38]. Above-normal levels of muscle glycogen created through feeding of high-starch diets do not appear to be beneficial to performance. The addition of fat to the diet can have a protective effect on muscle glycogen and blood glucose, but the effects on performance remain variable.
5.3 Neurologic Horse at Fall Risk
Horses with neurologic involvement from EPM or concurrent conditions require environmental modifications alongside nutritional management. Affected horses can have focal or multifocal central nervous system disease [39]. EPM can be difficult to diagnose antemortem, and treatment with approved antiprotozoal agents frequently leads to improvement, but complete resolution is uncommon and relapse may occur. Prognosis is guarded, particularly in cases with cranial nerve involvement, where outcomes are less favorable than for spinal cord deficits [2].
For horses with both muscle and neurologic signs, the diagnostic workup must address both systems. If concurrent PSSM and EPM are suspected, both histological and genetic analyses are essential in the investigation of muscle disease, since 10% of horses with muscle disease and normal histology had a muscle disease-causing genetic variant, and 63% of horses with histologically confirmed muscle alterations had no known genetic variants [5]. Management priorities should address the most acute condition first while implementing long-term dietary strategies for both conditions.
6. Monitoring, Reassessment, and Evidence Gaps
6.1 Monitoring Metrics and Reassessment Schedule
Serum creatine kinase (CK) activity serves as the primary monitoring metric for muscle damage assessment. Blood biochemical parameters provide baseline data for horses undergoing physical training, with standard reference ranges established for aspartate aminotransferase (AST: 306.27±84.66 U/L) and creatine kinase (CK: 181.06±80.38 U/L) in training sport horses [40]. Monitoring of creatine kinase and lactate dehydrogenase can reveal the state of muscle and its biochemical adaptation to physical load [41].
The classic triad of rhabdomyolysis symptoms includes muscle pain, weakness, and myoglobinuria; however, this triad is found in less than 10% of patients with diagnosed rhabdomyolysis, and more than 50% of patients do not complain of muscle pain or weakness [42]. Because CK is considered to be the most sensitive indirect marker, the diagnosis of rhabdomyolysis relies essentially on serum or plasma CK values. The diagnostic cutoff value for CK varies widely, from 1000 or 2000 U/L to 10,000 U/L. In horses with recurrent exertional rhabdomyolysis, oral administration of dantrolene at 4 mg/kg provided measurable plasma concentrations and significantly decreased serum CK activity after exercise [43].
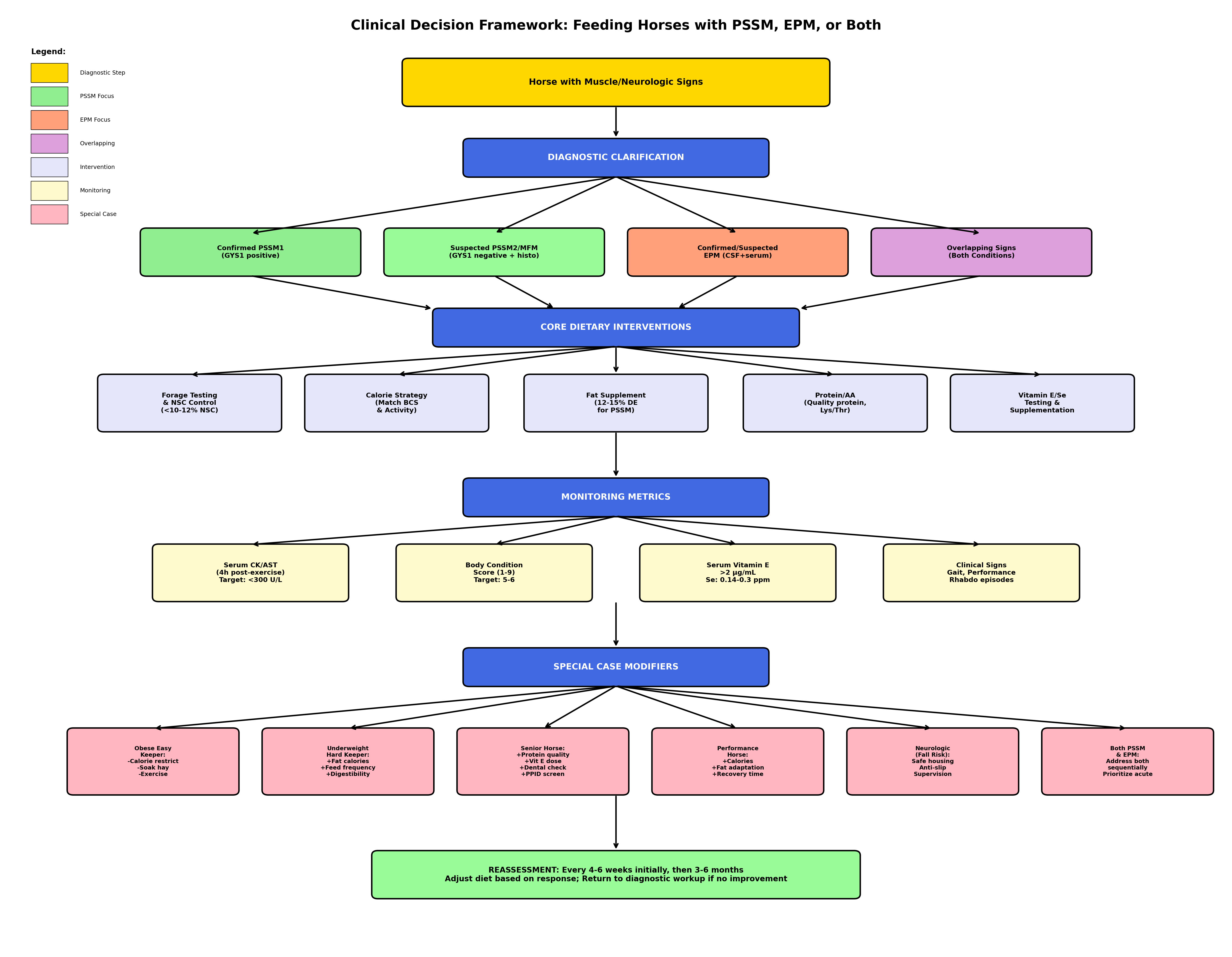
6.2 Evidence-Based Intervention Table
| Intervention | Evidence Level | Rationale | Contraindications | Monitoring Metric |
|---|---|---|---|---|
| NSC <10% DE (PSSM) | Strong (RCTs) | Reduces postprandial glucose/insulin response; decreases glycogen synthesis | Underweight horses requiring high calories | Serum CK 4h post-exercise; episode frequency |
| Fat 12-15% DE (PSSM) | Strong (RCTs) | Increases FFA availability; provides alternative energy substrate | Obesity without exercise program; hepatic disease | Body condition score; muscle enzyme recovery |
| Vitamin E 2000-5000 IU/day | Moderate (cohort) | Prevents oxidative damage; supports neuromuscular function | Rare: hypervitaminosis at extreme doses | Serum α-tocopherol (target >2 μg/mL) |
| Selenium supplementation | Moderate (cohort) | Supports GSH-Px activity; antioxidant defense | Se-adequate areas; toxicity risk | Whole blood Se; GSH-Px activity |
| Forage soaking/steaming | Moderate (lab studies) | Reduces WSC content 30-40% | May increase microbial contamination if improper | Hay analysis; body weight stability |
| Graduated exercise | Strong (clinical) | Promotes oxidative capacity; reduces insulin resistance | Acute rhabdomyolysis; severe weakness | CK trends; performance improvement |
| Low-calorie management (obese) | Moderate (observational) | Improves insulin sensitivity; reduces laminitis risk | Lactation; growth; high work demands | BCS; cresty neck score; insulin response |
| Amino acid supplementation | Limited (preliminary) | Supports muscle protein synthesis; reduces degradation | Renal compromise | Muscle mass maintenance; PUN |
6.3 Evidence Gap List
Several critical knowledge gaps remain in the clinical management of these conditions:
- PSSM2 genetic basis: The cause of PSSM2 in Quarter Horses remains unknown, and glycogen concentrations are not well-defined [1]. Of the 29 predicted high or moderate impact genetic variants identified in candidate genes, none were present only in PSSM2 horses and absent in controls.
- Optimal fat type and quantity: While long-chain fats are preferred, the optimal specific fatty acid profile and quantity for different PSSM phenotypes requires further investigation.
- EPM nutritional support: Limited evidence exists specifically evaluating nutritional interventions for EPM beyond antiprotozoal therapy. The role of vitamin E supplementation in EPM recovery remains inadequately characterized.
- Concurrent PSSM-EPM management: No controlled studies have evaluated dietary protocols specifically designed for horses with both conditions.
- Individual vitamin E response: Wide variability in vitamin E absorption efficiency exists, with some horses requiring high doses of supplementation to remain in the normal range, but genetic factors underlying this variability have not been evaluated in horses [19].
- Long-term outcomes: Most dietary intervention studies are short-term; long-term effects of strict NSC restriction and high-fat feeding on muscle health, performance, and longevity require further study.
- Breed-specific recommendations: While GYS1 mutation prevalence varies significantly among breeds, breed-specific dietary modifications beyond genetic testing guidance are not established.
- Exercise prescription specifics: The optimal intensity, duration, and frequency of exercise for different PSSM phenotypes and recovery stages from EPM lack standardized evidence-based protocols.
This clinical decision framework integrates current evidence for managing horses with PSSM, EPM, or both conditions. Diagnostic confirmation should guide the intensity of dietary modifications, with PSSM1-confirmed horses requiring the strictest NSC control and highest fat supplementation. EPM management prioritizes antiprotozoal treatment with nutritional support focusing on vitamin E adequacy and overall body condition maintenance. Regular reassessment of clinical response, serum enzyme activities, and body condition enables evidence-based adjustments to optimize outcomes for individual horses.
References
References
Numbered in order of first citation, IEEE style. Each entry links to the original source via DOI or publisher URL.
- S. Valberg, Z. J. Williams, C. Finno, A. Schultz, D. Velez-Irizarry, M. Henry, K. Gardner, and J. Petersen, “Type 2 polysaccharide storage myopathy in Quarter Horses is a novel glycogen storage disease causing exertional rhabdomyolysis.,” Equine Veterinary Journal, Aug. 2022. doi: 10.1111/evj.13876. View source
- S. Austin and S. Gray, “Equine protozoal myeloencephalitis—Presentation and progression,” Equine Veterinary Education, Oct. 2025. doi: 10.1111/eve.70016. View source
- R. Geor, “Role of dietary energy source in the expression of chronic exertional myopathies in horses,” Journal of Animal Science, Jun. 2005. doi: 10.2527/2005.8313_SUPPLE32X. View source
- N. Blohina, S. I. Sorokin, L. Khrabrova, A. Zelenchenkova, and B. Bazaron, “Biodiversity and functional variability of the allele pool of horse breed populations,” IOP Conference Series: Earth and Environment, Sep. 2021. doi: 10.1088/1755-1315/848/1/012229. View source
- M. Aleman, R. Scalco, J. Malvick, R. Grahn, A. True, and R. R. Bellone, “Prevalence of genetic mutations in horses with muscle disease from a neuromuscular disease laboratory.,” Journal of Equine Veterinary Science, Sep. 2022. doi: 10.2139/ssrn.4100989. View source
- Z. J. Williams, M. Bertels, and S. Valberg, “Muscle glycogen concentrations and response to diet and exercise regimes in Warmblood horses with type 2 Polysaccharide Storage Myopathy,” PLoS ONE, Sep. 2018. doi: 10.1371/journal.pone.0203467. View source
- R. MacKay and D. K. Howe, “Equine Protozoal Myeloencephalitis.,” The Veterinary clinics of North America. Equine practice, Jul. 2022. doi: 10.1016/j.cveq.2022.05.003. View source
- P. Bernardino, W. Smith, P. Conrad, A. Packham, E. Tamez-Trevino, S. Barnum, and N. Pusterla, “Molecular detection of Sarcocystis neurona in cerebrospinal fluid from 210 horses with suspected neurologic disease.,” Veterinary parasitology, Feb. 2021. doi: 10.1016/j.vetpar.2021.109372. View source
- W. P. Ribeiro, S. Valberg, J. D. Pagan, and B. Gustavsson, “The effect of varying dietary starch and fat content on serum creatine kinase activity and substrate availability in equine polysaccharide storage myopathy.,” Journal of Veterinary Internal Medicine, Nov. 2004. doi: 10.1111/j.1939-1676.2004.tb02637.x. View source
- M. Moore‐Colyer, K. Lumbis, A. C. Longland, and P. A. Harris, “The Effect of Five Different Wetting Treatments on the Nutrient Content and Microbial Concentration in Hay for Horses,” PLoS ONE, Nov. 2014. doi: 10.1371/journal.pone.0114079. View source
- J. R. Weinert-Nelson, A. Biddle, H. Sampath, and C. A. Williams, “Fecal Microbiota, Forage Nutrients, and Metabolic Responses of Horses Grazing Warm- and Cool-Season Grass Pastures,” Animals, Feb. 2023. doi: 10.3390/ani13050790. View source
- L. A. Borgia, S. J. Valberg, M. E. McCue, J. D. Pagan, and C. R. Roe, “Effect of dietary fats with odd or even numbers of carbon atoms on metabolic response and muscle damage with exercise in Quarter Horse–type horses with type 1 polysaccharide storage myopathy,” American Journal of Veterinary Research, Mar. 2010. doi: 10.2460/ajvr.71.3.326. View source
- K. C. Mowry, T. L. Thomson-Parker, C. Morales, K. Fikes, K. J. Stutts, J. L. Leatherwood, M. J. Anderson, R. X. Smith, and J. K. Suagee-Bedore, “Effects of Crude Rice Bran Oil and a Flaxseed Oil Blend in Young Horses Engaged in a Training Program,” Animals, Nov. 2022. doi: 10.3390/ani12213006. View source
- J. H. van der Kolk, S. Thomas, N. Mach, A. Ramseyer, D. Burger, V. Gerber, and J. Nuoffer, “Serum acylcarnitine profile in endurance horses with and without metabolic dysfunction.,” The Veterinary Journal, Jan. 2020. doi: 10.1016/j.tvjl.2019.105419. View source
- C. M. Latham, A. L. Wagner, and K. Urschel, “Effects of dietary amino acid supplementation on measures of whole‐body and muscle protein metabolism in aged horses,” Journal of animal physiology and animal nutrition, Oct. 2018. doi: 10.1111/jpn.12992. View source
- N. C. Galinelli, S. Bailey, N. J. Bamford, and P. A. Harris, “Nutritional considerations for the management of equine pituitary <i>pars intermedia</i> dysfunction,” Equine Veterinary Education, Nov. 2021. doi: 10.1111/eve.13593. View source
- D. Laflamme, “61 Life Stage Feeding: Is There Path Forward for Senior/Geriatric Nutrient recommendations?,” Journal of Animal Science, Nov. 2023. doi: 10.1093/jas/skad281.138. View source
- H. E. Bedford, S. J. Valberg, A. M. Firshman, M. Lucio, M. K. Boyce, and T. N. Trumble, “Histopathologic findings in the sacrocaudalis dorsalis medialis muscle of horses with vitamin E–responsive muscle atrophy and weakness,” Journal of the American Veterinary Medical Association, Apr. 2013. doi: 10.2460/javma.242.8.1127. View source
- C. J. Finno, “How Nutrigenomics Impacts Equine Health - A Case Study of Vitamin E.,” Journal of Equine Veterinary Science, Mar. 2025. doi: 10.1016/j.jevs.2025.105421. View source
- K. Vanschandevijl, H. Nollet, P. Deprez, C. Delesalle, L. Lefère, J. Dewulf, and G. V. Loon, “Variation in deficient serum vitamin E levels and impact on assessment of the vitamin E status in horses,” Vlaams Diergeneeskundig Tijdschrift, Feb. 2009. doi: 10.21825/vdt.87493. View source
- E. N. Hales, H. Habib, G. Favro, S. A. Katzman, R. Russell Sakai, S. A. Marquardt, M. H. Bordbari, B. Ming-Whitfield, J. Peterson, A. R. Dahlgren, V. N. Rivas, C. A. Ramirez, S. Peng, C. G. Donnelly, B. Dizmang, A. Kallenberg, R. A. Grahn, A. D. Miller, K. Woolard, B. C. Moeller, B. Puschner, and C. J. Finno, “Increased α-tocopherol metabolism in horses with equine neuroaxonal dystrophy,” Journal of Veterinary Internal Medicine, Jul. 2021. doi: 10.1111/jvim.16233. View source
- C. Kósa, K. Nagy, O. Szenci, B. Vincze, E. Andrásofszky, R. Szép, Á. Keresztesi, M. Mircean, M. Taulescu, and O. Kutasi, “The role of selenium and vitamin E in a Transylvanian enzootic equine recurrent rhabdomyolysis syndrome,” Acta Veterinaria Hungarica, Sep. 2021. doi: 10.1556/004.2021.00031. View source
- B. Hosnedlová, M. Kepinská, S. Skalíčková, C. Fernández, B. Ruttkay-Nedecký, T. D. Malevu, J. Sochor, M. Baroň, M. Melčová, J. Zídková, and R. Kizek, “A Summary of New Findings on the Biological Effects of Selenium in Selected Animal Species—A Critical Review,” International Journal of Molecular Sciences, Oct. 2017. doi: 10.3390/ijms18102209. View source
- C. Delesalle, M. D. Bruijn, S. Wilmink, H. Vandendriessche, G. C. Mol, B. Boshuizen, L. Plancke, and G. C. M. Grinwis, “White muscle disease in foals: focus on selenium soil content. A case series,” BMC Veterinary Research, May 2017. doi: 10.1186/s12917-017-1040-5. View source
- T. J. Divers, J. E. Cummings, A. D. Lahunta, H. F. Hintz, and H. O. Mohammed, “Evaluation of the risk of motor neuron disease in horses fed a diet low in vitamin E and high in copper and iron,” American Journal of Veterinary Research, Jan. 2006. doi: 10.2460/ajvr.67.1.120. View source
- W. Kędzierski, A. Chałabis‐Mazurek, Z. Bełkot, I. Janczarek, and S. Kowalik, “Zinc, copper and selenium deficiencies in broodmares in south-eastern Poland,” Polish Journal of Veterinary Sciences, Sep. 2025. doi: 10.24425/pjvs.2025.156074. View source
- M. G. Cappai, A. Taras, I. Cossu, R. Cherchi, C. Dimauro, F. Accioni, G. Boatto, M. Deroma, E. Spanu, D. Gatta, C. Dall’Aglio, and W. Pinna, “Effects of Dietary Zn/Se and α-Tocopherol Supplementation on Metabolic Milieu, Haemogram and Semen Traits of Breeding Stallions,” Biological Trace Element Research, Oct. 2020. doi: 10.1007/s12011-020-02447-7. View source
- A. Cristian, A. Dutulescu, and M. Codreanu, “Equine metabolic syndrome,” Practica Veterinara ro, 2024. doi: 10.26416/pv.43.1.2024.9642. View source
- C. D. Thatcher, R. Scott Pleasant, R. J. Geor, and F. Elvinger, “Prevalence of Overconditioning in Mature Horses in Southwest Virginia during the Summer,” Journal of Veterinary Internal Medicine, Sep. 2012. doi: 10.1111/j.1939-1676.2012.00995.x. View source
- N. Karikoski, J. R. Box, A. Mykkänen, V. V. Kotiranta, and M. Raekallio, “Variation in insulin response to oral sugar test in a cohort of horses throughout the year and evaluation of risk factors for insulin dysregulation,” Equine Veterinary Journal, May 2021. doi: 10.1111/evj.13529. View source
- K. Langner, D. Blaue, C. Schedlbauer, J. Starzonek, V. Julliand, and I. Vervuert, “Changes in the faecal microbiota of horses and ponies during a two-year body weight gain programme,” PLoS ONE, Mar. 2020. doi: 10.1371/journal.pone.0230015. View source
- A. Tomal, J. Szłapka-Kosarzewska, M. Mironiuk, I. Michalak, and K. Marycz, “Arthrospira platensis enriched with Cr(III), Mg(II), and Mn(II) ions improves insulin sensitivity and reduces systemic inflammation in equine metabolic affected horses,” Frontiers in Endocrinology, Apr. 2024. doi: 10.3389/fendo.2024.1382844. View source
- S. Pratt-Phillips, “Effect of Exercise Conditioning on Countering the Effects of Obesity and Insulin Resistance in Horses—A Review,” Animals, Feb. 2024. doi: 10.3390/ani14050727. View source
- J. M. MacLeay, S. J. Valberg, J. D. Pagan, J. Xue, F. D. D. L. Côrte, and J. M. Roberts, “Effect of ration and exercise on plasma creatine kinase activity and lactate concentration in Thoroughbred horses with recurrent exertional rhabdomyolysis,” American Journal of Veterinary Research, Nov. 2000. doi: 10.2460/ajvr.2000.61.1390. View source
- B. A. Valentine, H. F. Hintz, K. M. Freels, A. J. Reynolds, and K. Thompson, “Dietary control of exertional rhabdomyolysis in horses,” Journal of the American Veterinary Medical Association, May 1998. doi: 10.2460/javma.1998.212.10.1588. View source
- A. Jansson and J. Lindberg, “A forage-only diet alters the metabolic response of horses in training.,” Animal, Dec. 2012. doi: 10.1017/S1751731112000948. View source
- S. Hyyppä, “Post-exercise muscle glycogen repletion in horses,” Jukuri (Natural Resources Institute Finland (Luke)), Nov. 2007. doi: None. View source
- D. Freeman, D. Topliff, J. P. Baker, and W. Gill, “Nutrient Requirements of the Exercising Equine-A Review1,” Jun. 1987. doi: 10.15232/S1080-7446(15)32371-8. View source
- S. Reed, S. Reed, M. Furr, D. Howe, A. L. Johnson, R. MacKay, J. Morrow, N. Pusterla, and S. Witonsky, “Equine Protozoal Myeloencephalitis: An Updated Consensus Statement with a Focus on Parasite Biology, Diagnosis, Treatment, and Prevention,” Journal of Veterinary Internal Medicine, Feb. 2016. doi: 10.1111/jvim.13834. View source
- F. G. F. Padilha, L. A. G. Dimache, F. Q. D. Almeida, and A. M. R. Ferreira, “Blood biochemical parameters of Brazilian sport horses under training in tropical climate,” Revista Brasileira de Zootecnia, Aug. 2017. doi: 10.1590/s1806-92902017000800008. View source
- P. Brancaccio, F. Limongelli, and N. Maffulli, “Monitoring of serum enzymes in sport,” British Journal of Sports Medicine, Jan. 2006. doi: 10.1136/bjsm.2005.020719. View source
- M. Wernbom, G. Paulsen, T. Bjørnsen, K. Cumming, and T. Raastad, “Reply.,” Clinical Journal of Sports Medicine, Sep. 2020. doi: 10.1097/JSM.0000000000000854. View source
- E. C. McKenzie, S. J. Valberg, S. Godden, C. J. Finno, and M. J. Murphy, “Effect of oral administration of dantrolene sodium on serum creatine kinase activity after exercise in horses with recurrent exertional rhabdomyolysis,” American Journal of Veterinary Research, Jan. 2004. doi: 10.2460/ajvr.2004.65.74. View source
This review is one of 34 in the Foxwatch research corpus. Methodology: PRISMA 2020-style systematic literature review against PubMed, CAB Abstracts, Scopus, Web of Science, and equine veterinary conference proceedings.