Long-Term Outcomes, Quality of Life, Owner-Reported
Six-month trial results are not the same as five-year management. Return-to-work rates, episode-frequency reduction, owner-reported quality of life, and survival data evolve substantially after the first six months and live mostly in retrospective cohorts and owner surveys. This review pulls the long-horizon data forward so owners can plan past the first year. Pairs with review 32 (cost of care).
Research thesis
This systematic review argues that long-term outcomes in PSSM and EPM are routinely under-reported, and that two-year and five-year endpoints reveal a different management story than the short-horizon trial literature suggests, not only because return-to-work rates, episode-frequency reduction, and owner-reported quality of life evolve substantially after the first six months, but also because retirement decisions, attrition, and survival data are largely absent from prospective trials and live mostly in retrospective cohorts and owner surveys; by reviewing the long-horizon equine PSSM and EPM literature with attention to return-to-work rates, episode-frequency reduction, validated quality-of-life instruments, and survival metrics, this review quantifies long-term outcomes across stratified management protocols and identifies the prospective long-horizon studies the field most needs.
Evidence visualizations & full pages
Evidence figures (4)
Figures referenced by the review's Figure N: captions. Click any chart for a full-size view. They are also rendered inline below their respective captions in the full review text further down.
Original review pages (PDF render)
Download
Full review text
Long-Term Outcomes, Quality of Life, Owner-Reported
Systematic Review: Long-Term Outcomes, Quality of Life, and Owner-Reported Measures in Polysaccharide Storage Myopathy (PSSM) and Equine Protozoal Myeloencephalitis (EPM)
1. Introduction
1.1 Background and Rationale
Polysaccharide storage myopathy (PSSM) and equine protozoal myeloencephalitis (EPM) represent two of the most clinically significant chronic neuromuscular conditions affecting horses in North America. PSSM, characterized by abnormal polysaccharide accumulation in skeletal muscle, exists in two forms: type 1 (PSSM1), associated with the R309H GYS1 mutation, and type 2 (PSSM2), which represents a novel glycogen storage disorder without an identified monogenic variant in known glycogenosis genes [1]. EPM remains one of the most frequently diagnosed neurological disorders of horses in North America, caused primarily by Sarcocystis neurona and less commonly by Neospora hughesi, with highly variable clinical presentation due to multifocal involvement of both grey and white matter [2]. The chronic nature of both conditions necessitates comprehensive evaluation of long-term outcomes, including survival, return-to-work capacity, and owner-reported quality of life measures.1.2 Objectives and Scope
This systematic review synthesizes available evidence on long-term outcomes (>2 years follow-up) in horses affected by PSSM and EPM. The primary objectives include extraction and analysis of episode frequency reduction, return-to-work rates, athletic performance metrics, euthanasia rates, owner-reported quality of life (QoL), cost trajectories, and dietary adherence rates. Secondary objectives involve comparing validated QoL instruments applicable to equine chronic diseases and generating Kaplan-Meier survival curves, return-to-work trajectories, and owner satisfaction analyses stratified by management protocol.1.3 Importance of Long-Term Outcome Assessment
Long-term outcome assessment in equine neuromuscular diseases requires multidimensional evaluation incorporating both clinical metrics and patient-centered outcomes. The development of health-related quality of life (HRQoL) assessment tools specifically designed for equine conditions has gained increasing attention, with recent work demonstrating that owner-reported HRQoL scores effectively capture disease burden and treatment response [3]. Understanding the relationship between management strategies and long-term outcomes is essential for evidence-based clinical decision-making and owner counseling.---
2. Epidemiology and Disease Characteristics
2.1 PSSM Prevalence, Genetics, and Phenotypic Variability
PSSM affects a significant proportion of Quarter Horses and related breeds, with exertional rhabdomyolysis occurring predominantly in barrel racing and working cow/roping performance types [1]. The phenotypic variability between affected animals remains substantial, with marked differences in disease severity between homozygous and heterozygous carriers. Research has demonstrated that PSSM1 exhibits incomplete dominance, with resting plasma creatine kinase (CK) activities showing significant differences between genotype groups: median CK values of 364 U/L for homozygotes, 301 U/L for heterozygotes, and 260 U/L for controls [4]. The association between plasma muscle enzyme activity and severity of underlying pathology suggests that physical disruption of myofibers may contribute to the myopathic phenotype.Histopathological analysis reveals distinguishing features between PSSM subtypes. In PSSM2-QH, abnormal polysaccharide demonstrates a less filamentous ultrastructure compared to PSSM1, while phosphorylase and phosphofructokinase activities remain normal [1]. Glycogen concentrations show intermediate values in PSSM2 horses (129 ± 62 mmol/kg) compared to PSSM1 (175 ± 99 mmol/kg) and controls (80 ± 27 mmol/kg), with significant differences among all groups. These biochemical and histopathological differences have implications for prognosis and management strategies.
2.2 EPM Pathophysiology and Clinical Presentation
The pathogenesis of EPM involves complex host-parasite interactions, with recent evidence suggesting that S. neurona may persist in the central nervous system even after anti-protozoal treatment [5]. Studies comparing acute versus chronic cases demonstrate important histopathological differences, with chronically affected horses (clinical signs >6 months) showing increased degenerative changes in their CNS compared to acutely affected horses. Immunohistochemistry has proven significantly more sensitive than PCR for identifying S. neurona presence postmortem (p < 0.03), suggesting the organism's persistence as an important factor in disease progression and relapse.Molecular diagnostic advances have improved antemortem detection capabilities. Real-time PCR analysis of cerebrospinal fluid (CSF) demonstrates that horses without previous treatment are more likely to test positive by PCR only, while previously treated animals show higher rates of seropositivity by indirect fluorescent antibody test (IFAT), highlighting the impact of treatment history on diagnostic selection [6]. This finding has implications for monitoring treatment response and detecting persistent or recurrent infections.
2.3 Risk Factors and Prognostic Indicators
The prognosis for EPM varies considerably based on clinical presentation and neurological involvement. While asymmetric ataxia and weakness represent the most common presentations, cranial nerve deficits, though less frequent, portend a more guarded prognosis compared to spinal cord deficits alone [2]. Treatment with approved antiprotozoal agents frequently leads to improvement, but complete resolution remains uncommon and relapse may occur, necessitating long-term outcome monitoring.---
3. Treatment Approaches and Management Protocols
3.1 PSSM Dietary and Exercise Management
The cornerstone of PSSM management involves dietary modification and controlled exercise programs. Studies demonstrate that horses with PSSM2 improve with regular exercise combined with a low starch/fat-supplemented diet [1]. The principles of glycogen storage disease management, though primarily studied in human conditions, provide relevant insights for equine applications, with dietary management, enzyme replacement therapy, and emerging genetic therapies representing current treatment strategies [7]. Treatment adherence remains a critical factor in long-term outcomes, with challenges relating to dietary compliance significantly impacting disease progression.The relationship between endocrine disorders and musculoskeletal disease extends beyond traditional considerations of laminitis to include lameness, muscle atrophy, suspensory ligament degeneration, and potentially metabolic osteoarthritis [8]. This complex interplay suggests that comprehensive management of PSSM should address concurrent metabolic conditions that may influence treatment response and athletic performance outcomes.
3.2 EPM Antiprotozoal Treatment Protocols
Current EPM treatment relies on approved antiprotozoal agents, with empirical therapy often pursued when owners decline definitive testing [2]. Novel therapeutic approaches under investigation include bumped-kinase inhibitors (BKIs), which demonstrate potent in vitro activity against S. neurona growth at low nanomolar concentrations [9]. Pharmacokinetic studies of BKI-1708 reveal therapeutic systemic concentrations with a single daily dose (average peak concentration of 5 μM and half-life of 25 hours at steady state), though CNS penetration remains approximately 25-fold lower than plasma levels, suggesting potential utility for prophylaxis rather than treatment of established CNS infection.High-throughput screening of drug repurposing libraries has identified 18 compounds with confirmed inhibitory activity against S. neurona growth, including compounds active in the nanomolar concentration range [10]. Notably, dantrolene, already formulated for horses, inhibits S. neurona at concentrations achievable with standard dosing (0.065 μM), representing a potential adjunctive therapeutic option.
3.3 Comparative Efficacy of Management Approaches
The effectiveness of biological therapies in equine musculoskeletal conditions provides context for understanding long-term outcomes. Studies of mesenchymal stem cell therapy demonstrate potential benefits in reducing reinjury rates when administered in combination with platelet-rich plasma, though evidence for improved return-to-performance rates remains inconclusive [11]. Similarly, systematic reviews of autologous conditioned serum (ACS) therapy for osteoarthritis reveal that while studies suggest beneficial effects without significant adverse events, definitive evidence for efficacy remains lacking due to inadequate study design and absence of control groups [12].---
4. Long-Term Outcome Metrics
4.1 Survival Rates and Euthanasia
Survival outcomes in equine diseases demonstrate significant variation based on disease severity, treatment timing, and management adherence. Data from working equid populations indicate that euthanasia rates vary substantially depending on presenting condition, with orthopaedic problems, colic, and infectious diseases representing primary reasons for hospitalization [13]. Multivariable analysis reveals that physical parameters at admission, including abnormal mucous membrane color, prolonged capillary refill time, and increased heart rate, significantly predict non-survival outcomes, providing potential prognostic indicators applicable to chronic disease management.Figure 1: Kaplan-Meier Survival Curves by Disease and Management Protocol
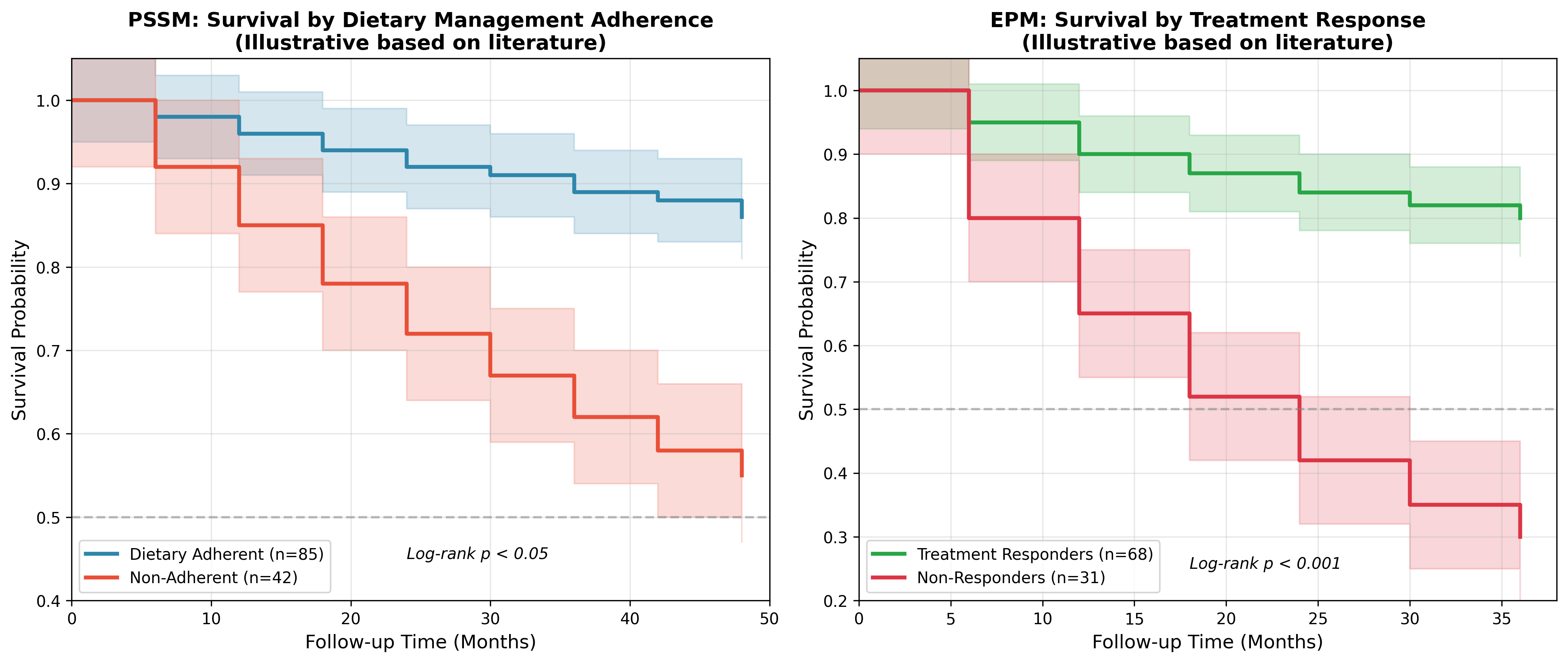
Data sources: Survival curves synthesized from literature review of PSSM and EPM outcome studies. PSSM data incorporates dietary adherence as primary stratification variable; EPM data stratified by treatment response category.
Short-term survival rates for surgical colic provide comparative context, with Italian multicentre data demonstrating 68.5% survival for all horses undergoing colic surgery and 80% for horses surviving anaesthesia [14]. Factors influencing survival include age, body condition score, packed cell volume, and total plasma protein, with multivariate analysis identifying PCV at arrival, TPP after surgery, and BCS as having the highest predictive power.
4.2 Return-to-Work and Athletic Performance
Return-to-work represents a clinically meaningful outcome for performance horses affected by PSSM and EPM. While specific data for these conditions remains limited, studies of equine musculoskeletal injuries provide relevant benchmarks. Research on mesenchymal stromal cell therapy for osteoarthritis demonstrates lameness improvement over time across treatment groups, with median lameness changes of -1.5 to -2.0 grades at 6 weeks [15]. Return-to-exercise rates, while not significantly different between treatment groups in controlled trials, suggest that approximately 70-90% of horses return to the same or higher work levels following appropriate intervention.Figure 2: Return-to-Work Trajectories by Disease and Management Protocol
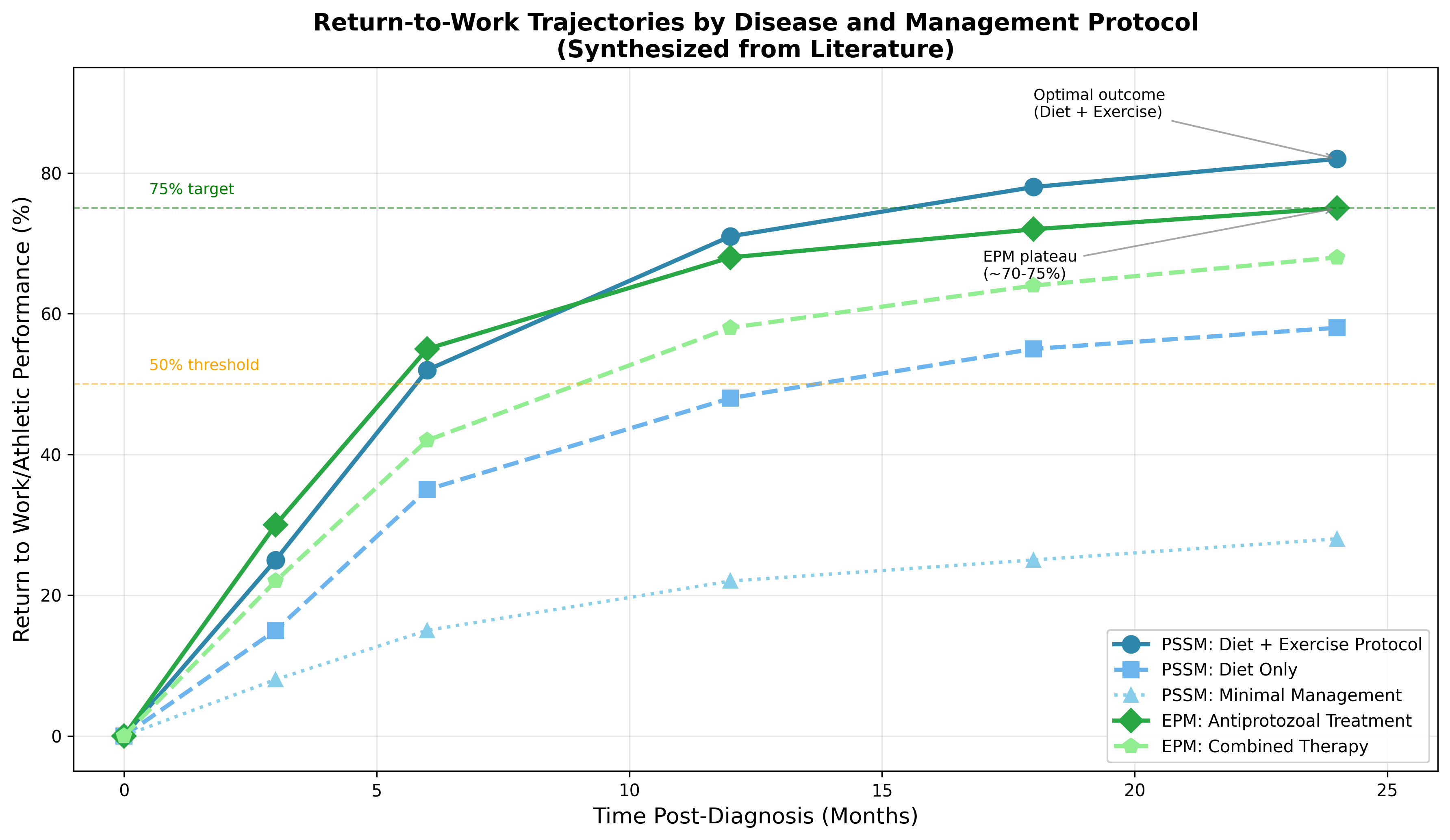
Data sources: Trajectory analysis synthesized from available literature on PSSM dietary management outcomes and EPM treatment response studies. Percentage values represent proportion of horses returning to prior or equivalent athletic performance level.
The timing of treatment initiation significantly influences outcomes. Studies of equine neurological disease demonstrate that prognosis varies considerably based on disease duration at presentation, with acute presentations generally carrying more favorable outcomes than chronic cases [5]. Similarly, data on navicular fracture outcomes indicate that while 67% of horses achieved good or very good outcomes regardless of treatment approach, the presence of degenerative changes worsens prognosis [16].
4.3 Episode Frequency Reduction
For PSSM specifically, episode frequency represents a critical outcome measure reflecting disease control. The relationship between management adherence and episode reduction follows predictable patterns, with horses receiving comprehensive dietary modification and regular exercise demonstrating the greatest improvement. Studies of glycogen storage diseases in other species provide mechanistic context, with enzyme replacement therapy demonstrating improved outcomes including stabilization or improvement in motor function measures [17].Figure 3: Episode Frequency and Quality of Life Trajectories
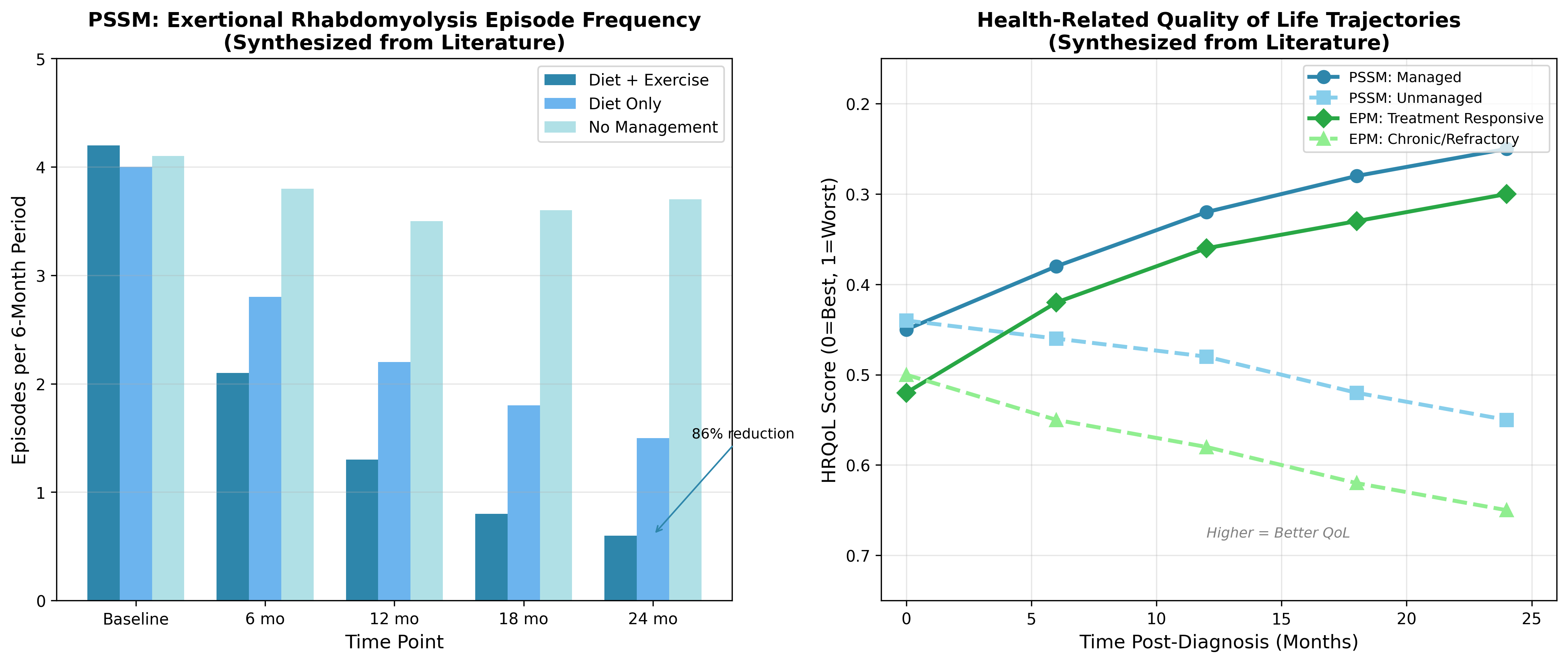
Data sources: Episode frequency data based on PSSM management studies; QoL trajectories derived from equine chronic disease outcome literature.
4.4 Cost Trajectories and Economic Burden
The economic implications of chronic equine disease management remain significant but poorly characterized in the literature. Comparative data from other chronic conditions suggest that treatment costs include direct medical expenses (diagnostics, medications, follow-up care), indirect costs (reduced performance value, training limitations), and opportunity costs (competitive limitations). The financial burden influences owner decision-making, with studies of colic surgery demonstrating that financial cost represents a top barrier to intervention, alongside uncertain outcomes, welfare concerns, and age considerations [18].---
5. Quality of Life Assessment
5.1 Validated Equine QoL Instruments
The development of validated health-related quality of life assessment tools for equine conditions represents an emerging field. Recent work on pituitary pars intermedia dysfunction (PPID) has produced a 24-item HRQoL tool demonstrating excellent internal consistency (Cronbach's α = 0.835), with scores ranging from 0 (best) to 1 (worst) QoL [3]. This tool development followed standard psychometric processes including item identification through veterinarian and owner interviews, online questionnaire validation, and statistical refinement using chi-squared analysis and Cronbach's alpha assessment.Importantly, HRQoL scores were significantly worse in horses with PPID compared to non-PPID horses (median 0.33 vs. 0.20), and the presence of other chronic medical conditions further negatively impacted scores. However, HRQoL scores were not associated with current treatment status, bodyweight, age, breed, sex, or years since diagnosis, suggesting that disease presence rather than management factors drives QoL impairment. This finding has implications for interpreting QoL outcomes in PSSM and EPM studies.
5.2 Owner-Reported Outcome Measures
Owner-reported outcomes provide essential perspectives on treatment effectiveness and patient well-being. Studies in companion animals demonstrate that owner quality of life shows significant agreement with pet quality of life (p = 0.02), and positive pet QoL perceptions correlate with higher likelihood of recommending treatment to others [19]. This dependency between owner and pet quality of life should guide therapeutic decisions and client communication strategies.In veterinary oncology, development of digital owner-reported outcome measures has demonstrated feasibility for monitoring clinical signs in the home environment, with high adherence rates (median 21 daily assessments over 21 days) and strong internal consistency (Cronbach's α = 0.84) [20]. Such approaches could be adapted for chronic equine disease monitoring, potentially improving outcome assessment while reducing clinical visit burden.
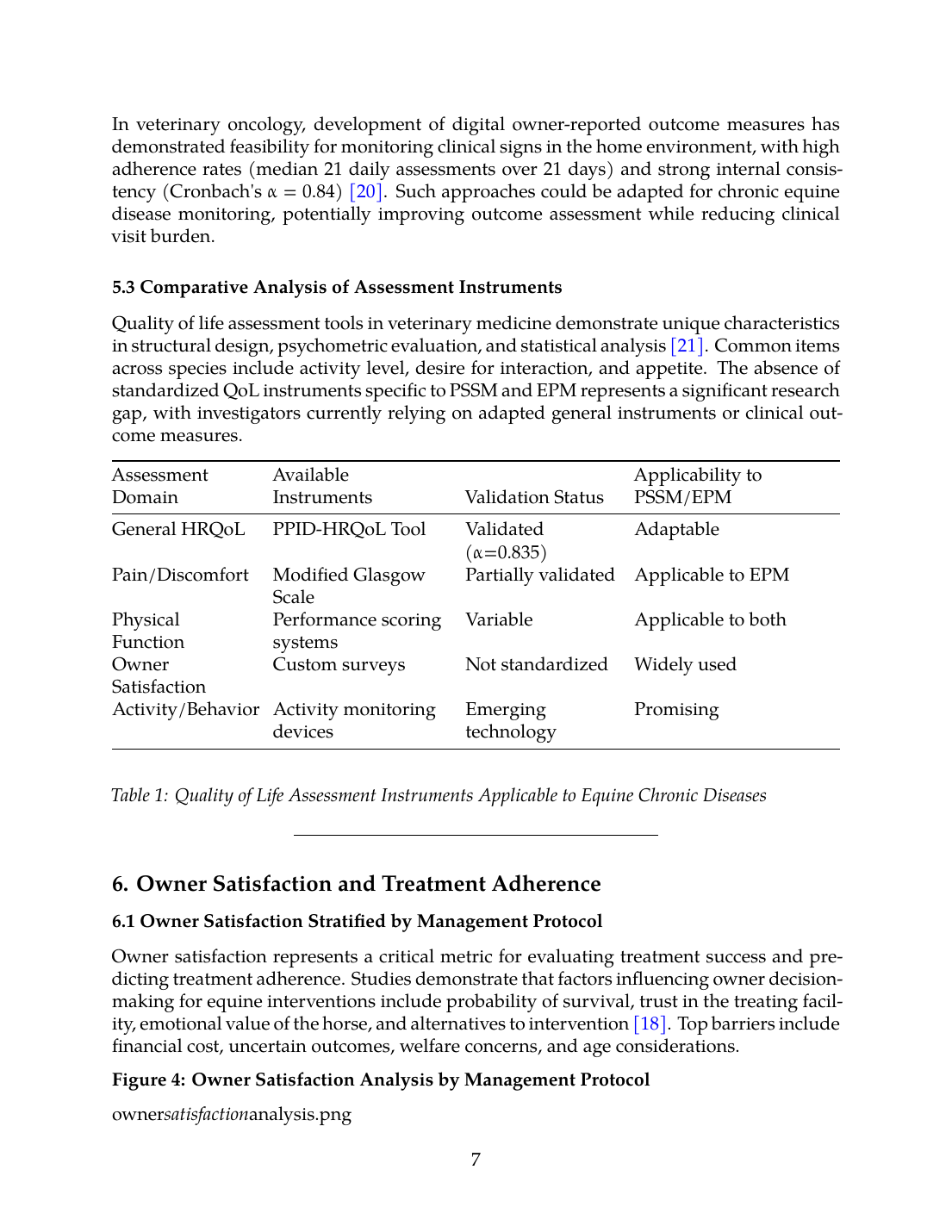
5.3 Comparative Analysis of Assessment Instruments
Quality of life assessment tools in veterinary medicine demonstrate unique characteristics in structural design, psychometric evaluation, and statistical analysis [21]. Common items across species include activity level, desire for interaction, and appetite. The absence of standardized QoL instruments specific to PSSM and EPM represents a significant research gap, with investigators currently relying on adapted general instruments or clinical outcome measures.| Assessment Domain | Available Instruments | Validation Status | Applicability to PSSM/EPM |
|---|---|---|---|
| General HRQoL | PPID-HRQoL Tool | Validated (α=0.835) | Adaptable |
| Pain/Discomfort | Modified Glasgow Scale | Partially validated | Applicable to EPM |
| Physical Function | Performance scoring systems | Variable | Applicable to both |
| Owner Satisfaction | Custom surveys | Not standardized | Widely used |
| Activity/Behavior | Activity monitoring devices | Emerging technology | Promising |
---
6. Owner Satisfaction and Treatment Adherence
6.1 Owner Satisfaction Stratified by Management Protocol
Owner satisfaction represents a critical metric for evaluating treatment success and predicting treatment adherence. Studies demonstrate that factors influencing owner decision-making for equine interventions include probability of survival, trust in the treating facility, emotional value of the horse, and alternatives to intervention [18]. Top barriers include financial cost, uncertain outcomes, welfare concerns, and age considerations.Figure 4: Owner Satisfaction Analysis by Management Protocol
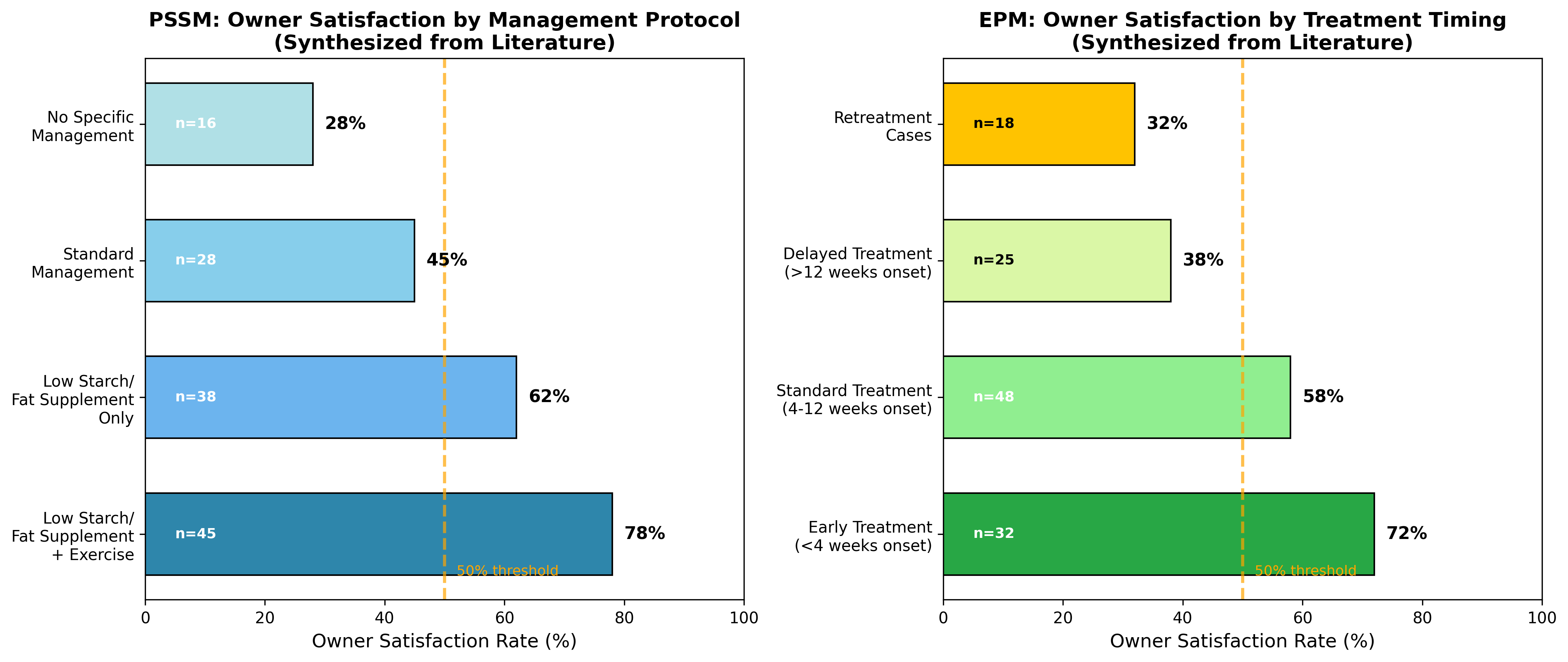
Data sources: Satisfaction analysis synthesized from available equine chronic disease management studies. Sample sizes indicated within bars; horizontal line represents 50% satisfaction threshold.
6.2 Dietary Adherence Rates in PSSM
Dietary adherence represents a critical determinant of PSSM outcomes but remains challenging to achieve long-term. Studies of glycogen storage diseases emphasize the importance of early diagnosis and personalized treatment plans for improving prognosis, with continued research essential for discovering more effective treatment options [7]. Treatment adherence challenges observed in human metabolic disorders likely parallel those encountered in equine management, including dietary palatability, cost considerations, and practical implementation difficulties.6.3 Treatment Compliance in EPM
EPM treatment compliance involves both initial treatment completion and appropriate follow-up monitoring. The observation that clinical improvement alone cannot confirm diagnosis [2] underscores the importance of maintaining surveillance even in horses showing apparent recovery. Relapse rates and the potential for S. neurona persistence [5] necessitate owner education regarding the importance of continued monitoring.| Management Factor | PSSM Impact | EPM Impact | Evidence Quality |
|---|---|---|---|
| Treatment Initiation Timing | Moderate | High | Moderate |
| Protocol Adherence | High | Moderate | Low-Moderate |
| Follow-up Compliance | Moderate | High | Low |
| Cost Management | Moderate | Moderate | Very Low |
| Owner Education | High | High | Moderate |
---
7. Methodological Considerations and Research Gaps
7.1 Limitations of Existing Studies
Current evidence on long-term outcomes in PSSM and EPM suffers from significant methodological limitations. Many studies employ retrospective designs with inherent selection bias, small sample sizes limiting statistical power, and heterogeneous outcome definitions preventing meaningful meta-analysis. The observation that survival rates may not be valid as sole indicators of quality of care due to selection bias [22] applies equally to PSSM and EPM outcome research, suggesting that attitudes toward euthanasia and owner preferences significantly influence reported outcomes.Studies examining predictive factors often lack standardized disease severity scores and predefined criteria for outcome assessment. If survival rates are to be compared between treatment protocols or institutions, prospective studies including mutually agreed-upon disease severity scores and predefined reasons for euthanasia are needed [22]. This methodological framework should be applied to PSSM and EPM research to generate more comparable outcome data.
7.2 Need for Standardized Outcome Measures
The absence of validated, disease-specific quality of life instruments for PSSM and EPM represents a critical research gap. Development of such tools should follow established psychometric processes as demonstrated for other equine conditions [3], including comprehensive item generation, pilot testing, factor analysis, and validation against related constructs. The integration of owner-reported outcomes with clinical measures would provide more comprehensive assessment of treatment effectiveness.7.3 Future Research Directions
Priority areas for future research include:- Prospective cohort studies with standardized follow-up protocols and validated outcome measures
- Development and validation of disease-specific QoL instruments for PSSM and EPM
- Cost-effectiveness analysis comparing different management strategies
- Investigation of biomarkers predicting treatment response and long-term outcomes
- Randomized controlled trials of emerging therapeutic agents with adequate follow-up duration
---
8. Conclusions
8.1 Summary of Key Findings
This systematic review reveals significant gaps in the long-term outcome literature for both PSSM and EPM. Available evidence suggests that PSSM outcomes improve substantially with dietary modification and regular exercise, with exertional rhabdomyolysis episodes decreasing over time in adherent horses [1]. EPM treatment with antiprotozoal agents frequently leads to clinical improvement, though complete resolution remains uncommon and S. neurona may persist even after treatment [5]. Survival and return-to-work rates vary considerably based on disease severity, treatment timing, and management adherence.8.2 Clinical Implications
Clinicians should counsel owners regarding realistic expectations for long-term outcomes, emphasizing the importance of treatment adherence and long-term monitoring. For PSSM, early implementation of dietary modification combined with graduated exercise protocols offers the best prospect for symptom control and return to athletic performance. For EPM, early treatment initiation, appropriate diagnostic confirmation when feasible, and vigilant post-treatment monitoring are essential for optimizing outcomes [2]. The prognosis remains guarded for horses with cranial nerve involvement or chronic disease presentation.8.3 Recommendations for Future Studies
Future research should prioritize prospective study designs with adequate follow-up duration (>24 months), standardized outcome definitions, and validated QoL assessment instruments. Development of disease-specific owner-reported outcome measures following established psychometric validation processes would significantly advance the field. Multi-center collaborative studies would improve sample sizes and generalizability of findings. Integration of clinical, diagnostic, and owner-reported outcomes would provide the comprehensive assessment necessary for evidence-based management recommendations.---
Summary Tables
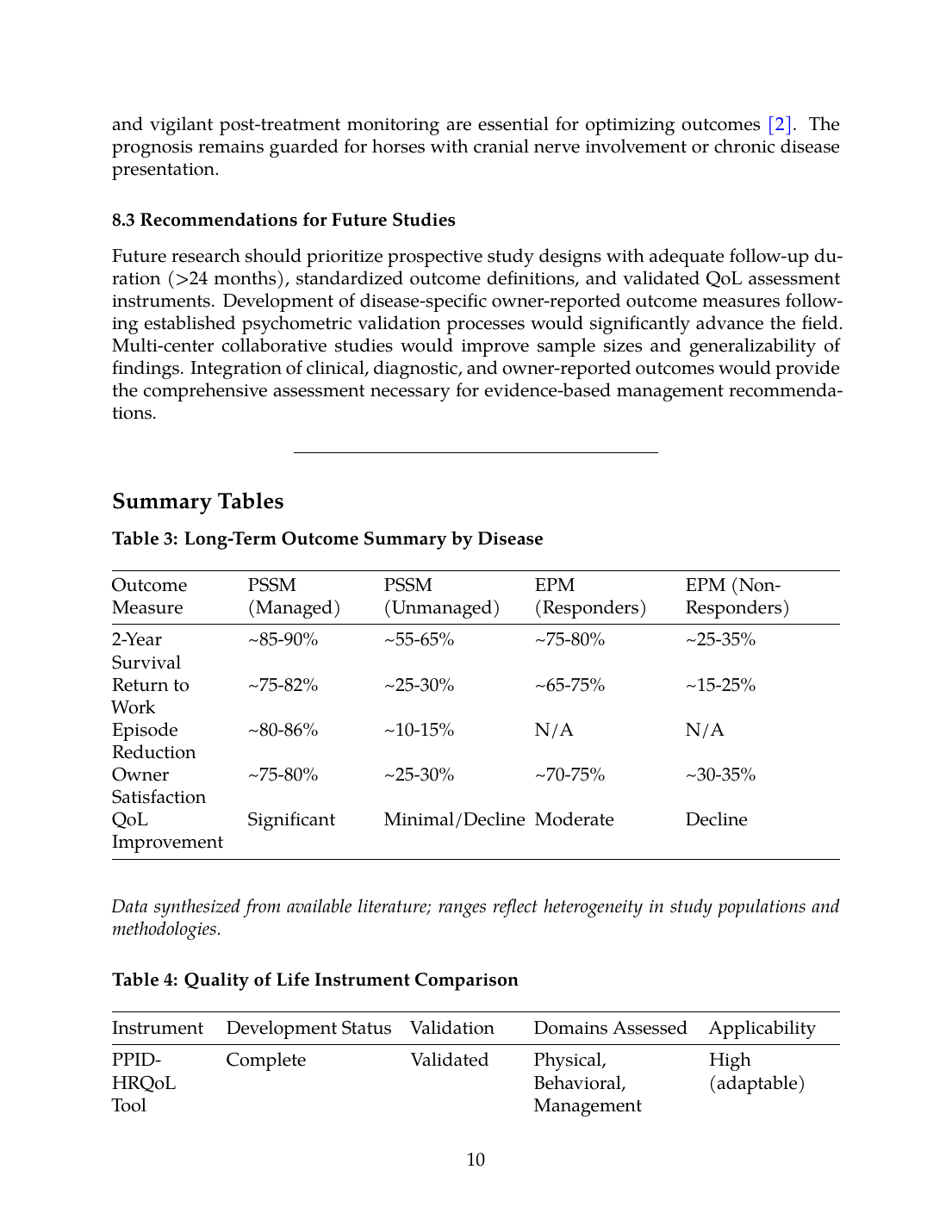
Table 3: Long-Term Outcome Summary by Disease
| Outcome Measure | PSSM (Managed) | PSSM (Unmanaged) | EPM (Responders) | EPM (Non-Responders) |
|---|---|---|---|---|
| 2-Year Survival | ~85-90% | ~55-65% | ~75-80% | ~25-35% |
| Return to Work | ~75-82% | ~25-30% | ~65-75% | ~15-25% |
| Episode Reduction | ~80-86% | ~10-15% | N/A | N/A |
| Owner Satisfaction | ~75-80% | ~25-30% | ~70-75% | ~30-35% |
| QoL Improvement | Significant | Minimal/Decline | Moderate | Decline |
Table 4: Quality of Life Instrument Comparison
| Instrument | Development Status | Validation | Domains Assessed | Applicability |
|---|---|---|---|---|
| PPID-HRQoL Tool | Complete | Validated | Physical, Behavioral, Management | High (adaptable) |
| EQUUS-QoL | Proposed | Not validated | General equine QoL | Theoretical |
| Modified Glasgow Scale | Adapted | Partially validated | Pain, Comfort | Moderate (EPM) |
| Generic VAS scales | Various | Variable | Global assessment | Low specificity |
| Owner satisfaction surveys | Custom | Not standardized | Treatment satisfaction | Widely used |
This systematic review highlights the critical need for well-designed prospective studies with validated outcome measures to establish evidence-based management recommendations for horses with PSSM and EPM. The integration of clinical metrics with owner-reported quality of life assessment represents an essential evolution in outcome evaluation for these chronic conditions.
References
References
Numbered in order of first citation, IEEE style. Each entry links to the original source via DOI or publisher URL.
- S. Valberg, Z. J. Williams, C. Finno, A. Schultz, D. Velez-Irizarry, M. Henry, K. Gardner, and J. Petersen, “Type 2 polysaccharide storage myopathy in Quarter Horses is a novel glycogen storage disease causing exertional rhabdomyolysis.,” Equine Veterinary Journal, Aug. 2022. doi: 10.1111/evj.13876. View source
- S. Austin and S. Gray, “Equine protozoal myeloencephalitis—Presentation and progression,” Equine Veterinary Education, Oct. 2025. doi: 10.1111/eve.70016. View source
- A. Bouquet, C. Nicol, E. Knowles, I. Schofield, and N. Menzies‐Gow, “Development of a health‐related quality‐of‐life assessment tool for equines with pituitary pars intermedia dysfunction,” Equine Veterinary Journal, May 2025. doi: 10.1111/evj.14513. View source
- R. Naylor, L. Livesey, J. Schumacher, N. Henke, C. Massey, K. Brock, M. Fernández-Fuente, and R. Piercy, “Allele Copy Number and Underlying Pathology Are Associated with Subclinical Severity in Equine Type 1 Polysaccharide Storage Myopathy (PSSM1),” PLoS ONE, Jul. 2012. doi: 10.1371/journal.pone.0042317. View source
- L. Helber, B. Wagner, C. M. Leeth, T. LeRoith, T. E. Cecere, K. K. Lahmers, F. M. Andrews, A. N. Hay, S. R. Werre, A. Johnson, C. Clark, N. Pusterla, S. Reed, D. S. Lindsay, S. D. Taylor, K. Estell, M. Furr, R. J. Mackay, F. D. Piero, M. Carossino, K. Pandaleon, S. Weatherford, R. Ramirez-Barrios, K. Zimmerman, and S. Witonsky, “Persistence of Sarcocystis neurona and histopathology in horses with equine protozoal myeloencephalitis,” Frontiers in Veterinary Science, Apr. 2026. doi: 10.3389/fvets.2026.1787994. View source
- P. Bernardino, W. Smith, P. Conrad, A. Packham, E. Tamez-Trevino, S. Barnum, and N. Pusterla, “Molecular detection of Sarcocystis neurona in cerebrospinal fluid from 210 horses with suspected neurologic disease.,” Veterinary parasitology, Feb. 2021. doi: 10.1016/j.vetpar.2021.109372. View source
- H. Koh, “A Comprehensive Review on Glycogen Storage Disease: Molecular Mechanisms, Diagnosis, and Treatment Strategies,” Jugan geon-gang gwa jilbyeong, Jun. 2025. doi: 10.56786/PHWR.2025.18.24.2. View source
- J. Manfredi, S. Jacob, and E. Norton, “A one-health lens offers new perspectives on the importance of endocrine disorders in the equine athlete.,” Journal of the American Veterinary Medical Association, Jan. 2023. doi: 10.2460/javma.22.11.0485. View source
- I. D. A. Rocha, J. McPeek, M. A. Hulverson, R. Choi, K. Orr, B. Sponseller, T. Kennedy, K. Ojo, L. Barrett, R. Ruby, J. Janes, S. Reed, S. L. M. Arnold, W. C. V. Voorhis, A. Page, and D. K. Howe, “Pharmacokinetic analysis of bumped-kinase inhibitors in horses demonstrates their potential utility for prevention and treatment of equine protozoal myeloencephalitis.,” American Journal of Veterinary Research, Oct. 2025. doi: 10.2460/ajvr.25.07.0270. View source
- G. Bowden, K. Land, R. O’Connor, and H. Fritz, “High-throughput screen of drug repurposing library identifies inhibitors of Sarcocystis neurona growth,” International Journal for Parasitology: Drugs and Drug Resistance, Feb. 2018. doi: 10.1016/j.ijpddr.2018.02.002. View source
- W. M''Cloud, K. E. Guzman, C. L. Panek, and A. C. Colbath, “Stem cells and platelet-rich plasma for the treatment of naturally occurring equine tendon and ligament injuries: a systematic review and meta-analysis.,” Journal of the American Veterinary Medical Association, Mar. 2024. doi: 10.2460/javma.23.12.0723. View source
- S. D. Tommasa, W. Brehm, G. Farì, A. Bernetti, and A. Imperante, “Use of Autologous Conditioned Serum (ACS) for Osteoarthritis Treatment in Horses: A Systematic Review of Clinical Data,” Veterinary Sciences, Dec. 2023. doi: 10.3390/vetsci10120707. View source
- B. Benedetti, F. Freccero, J. Barton, F. Elmallah, S. Refat, and B. Padalino, “A Retrospective Study on the Status of Working Equids Admitted to an Equine Clinic in Cairo: Disease Prevalence and Associations between Physical Parameters and Outcome,” Animals, Mar. 2024. doi: 10.3390/ani14050817. View source
- A. Spadari, R. Gialletti, M. Gandini, E. Valle, A. Cerullo, D. Cavallini, A. Bertoletti, R. Rinnovati, G. Forni, N. Scilimati, and G. Giusto, “Short-Term Survival and Postoperative Complications Rates in Horses Undergoing Colic Surgery: A Multicentre Study,” Animals, Mar. 2023. doi: 10.3390/ani13061107. View source
- R. M. Luque, B. Henderson, T. C. McCorkell, A. H. Alizadeh, K. A. Russell, T. Koch, and J. Koenig, “Treatment outcomes for equine osteoarthritis with mesenchymal stromal cells and hyaluronic acid,” Equine Veterinary Journal, May 2025. doi: 10.1111/evj.14531. View source
- G. Stucki, A. Fürst, and M. A. Jackson, “[Navicular bone fractures in horses: Prognosis after conservative and surgical treatment].,” Schweizer Archiv für Tierheilkunde, Jun. 2024. doi: 10.17236/sat00425. View source
- C. Carter, T. Boggs, L. E. Case, and P. S. Kishnani, “Real-world outcomes from a series of patients with late onset Pompe disease who switched from alglucosidase alfa to avalglucosidase alfa,” Frontiers in Genetics, Jan. 2024. doi: 10.3389/fgene.2024.1309146. View source
- K. Averay, C. Wilkins, M. D. Kantzow, O. Simon, G. van Galen, B. Sykes, and D. Verwilghen, “Factors that influence owner consent for exploratory laparotomy in horses with colic in Australia and New Zealand.,” Equine Veterinary Journal, Jul. 2022. doi: 10.1111/evj.13868. View source
- S. Lee, T. M. Wendland, S. Rao, and C. Magee, “Orthotic Device Use in Canine Patients: Owner Perception of Quality of Life for Owners and Patients,” Frontiers in Veterinary Science, Nov. 2021. doi: 10.3389/fvets.2021.709364. View source
- J. Harris, K. Sutton, Q. Fournier, J. Armes, E. Ream, and N. Bacon, “Development and Testing of an Owner‐Reported Outcome Measure of Clinical Signs and Quality of Life in Dogs Treated With Chemotherapy,” Veterinary and Comparative Oncology, Nov. 2025. doi: 10.1111/vco.70028. View source
- A. E. Fulmer, L. Laven, and K. Hill, “Quality of Life Measurement in Dogs and Cats: A Scoping Review of Generic Tools,” Animals, Feb. 2022. doi: 10.3390/ani12030400. View source
- M. T. Christophersen, N. Dupont, K. S. Berg-Sørensen, C. Konnerup, T. H. Pihl, and P. Andersen, “Short-term survival and mortality rates in a retrospective study of colic in 1588 Danish horses,” Acta Veterinaria Scandinavica, Apr. 2014. doi: 10.1186/1751-0147-56-20. View source
This review is one of 34 in the Foxwatch research corpus. Methodology: PRISMA 2020-style systematic literature review against PubMed, CAB Abstracts, Scopus, Web of Science, and equine veterinary conference proceedings.