Micronutrients Beyond Vitamin E and Selenium
Beyond vitamin E and selenium, vitamin D, magnesium, B-complex, copper, zinc, iron, and chromium each carry small but real risk or benefit during muscle and neurologic recovery. This review tiers them by evidence strength so you can build a complete supplement plan instead of stacking three blends and hoping. Pairs with reviews 13 (antioxidants) and 23 (trace minerals in PSSM2).
Research thesis
This systematic review argues that micronutrient management in PSSM and EPM cannot stop at vitamin E and selenium, and that vitamin D, magnesium, B-complex vitamins, copper, zinc, iron, and chromium each carry measurable risk or benefit during muscle and neurologic recovery, not only because regional forage and manufactured feed gaps in these nutrients are common, but also because antiprotozoal treatment, prolonged stall rest, and fat-substituted diets each shift micronutrient demand; by reviewing the equine micronutrient, deficiency-state, and supplementation-trial literature across PSSM and EPM populations, this review builds a tiered micronutrient protocol stratified by disease severity and clinical context.
Evidence visualizations & full pages
Evidence figures (2)
Figures referenced by the review's Figure N: captions. Click any chart for a full-size view. They are also rendered inline below their respective captions in the full review text further down.
Original review pages (PDF render)
Download
Full review text
Micronutrients Beyond Vitamin E and Selenium
Systematic Review: Micronutrient Supplementation Beyond Vitamin E and Selenium in Polysaccharide Storage Myopathy (PSSM) and Equine Protozoal Myeloencephalitis (EPM)
1. Introduction and Background
1.1 Overview of PSSM and EPM as Nutritionally-Responsive Conditions
Polysaccharide storage myopathy (PSSM) represents a significant glycogen storage disorder affecting multiple horse breeds, particularly Quarter Horses and related breeds. Type 1 PSSM is an autosomal dominant disorder affecting more than 20 breeds of horses, presenting with exertional rhabdomyolysis and characterized by increased insulin sensitivity, excessive muscle glycogen storage, and accumulation of amylase-resistant polysaccharide in muscle tissue [1]. The condition responds well to dietary modification, specifically low nonstructural carbohydrate, high fat diets combined with regular exercise [2]. While vitamin E and selenium have been the traditional focus of nutritional therapy, emerging evidence suggests that additional micronutrients may play important roles in optimizing outcomes for affected horses.
Equine degenerative myeloencephalopathy (EDM) and equine protozoal myeloencephalitis (EPM) represent neurodegenerative conditions with distinct etiologies but similar clinical presentations including proprioception deficits and progressive ataxia [3]. EPM, caused by Sarcocystis neurona infection, requires both antimicrobial therapy and supportive care, while EDM appears associated with early vitamin E deficiency and suspected genetic inheritance. Both conditions may benefit from comprehensive micronutrient optimization beyond standard vitamin E and selenium supplementation.
1.2 Rationale for Expanded Micronutrient Assessment
The pathophysiology of muscle myopathies involves complex metabolic pathways requiring multiple cofactors and minerals for optimal function. In PSSM horses, gene expression profiling has revealed inflammation, glycogenesis inhibition, hypoxia, and mitochondrial dysfunctions [4]. These findings suggest that addressing oxidative stress, energy metabolism, and inflammatory cascades may require a broader nutritional approach. Furthermore, Quarter Horses diagnosed with EDM are 2.95 times more likely to present with disease compared to other breeds, indicating potential breed-specific nutritional vulnerabilities [3].
1.3 Scope of This Review
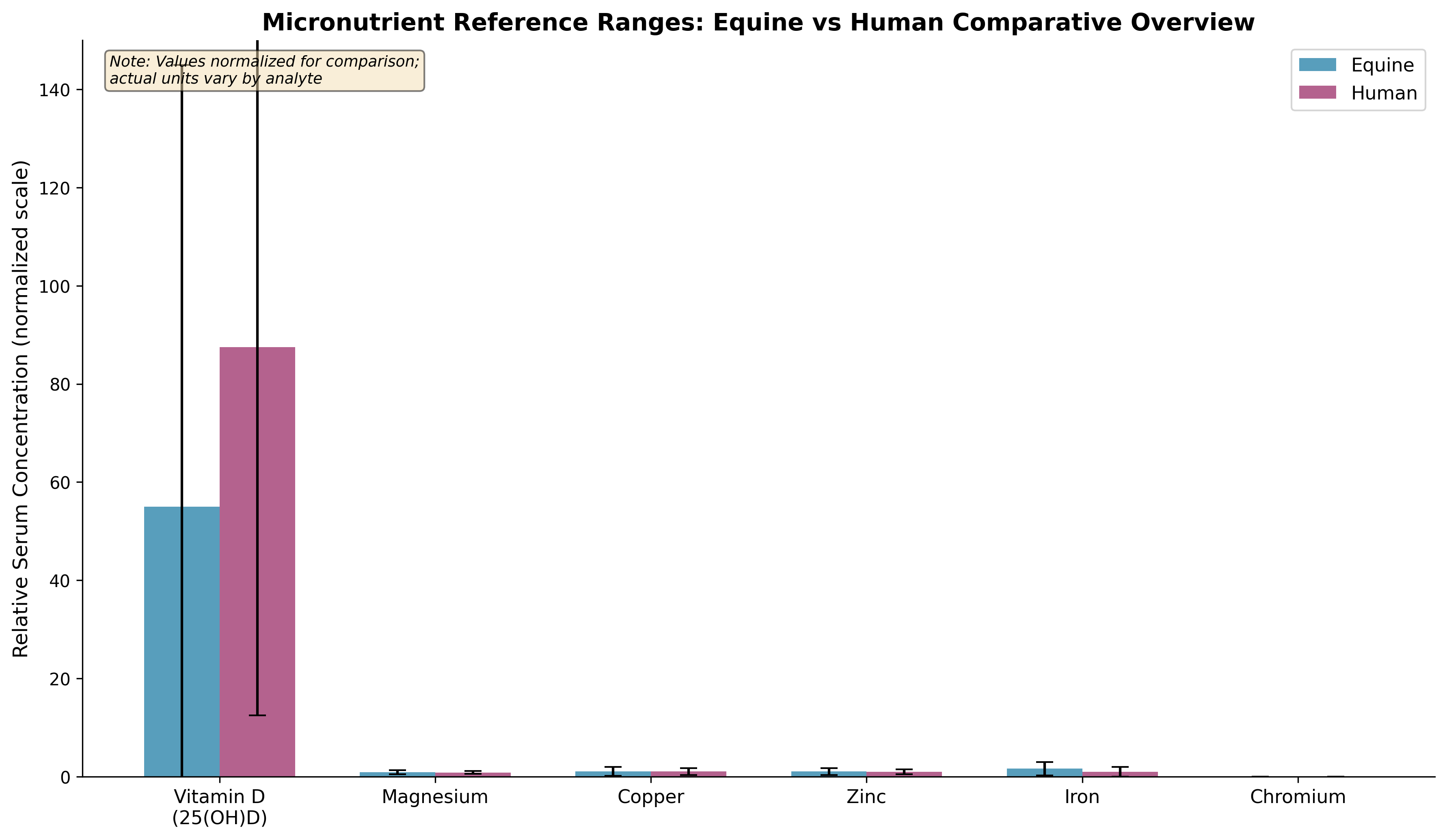
This systematic review examines the evidence for supplementation with vitamin D (25-OH-D testing), magnesium (oral, intravenous, and methylsulfonylmethane), B-complex vitamins (thiamine, riboflavin, cobalamin), and trace minerals (copper, zinc, iron, chromium) in horses with PSSM and EPM. We extract serum/plasma normal ranges, deficiency prevalence data, supplementation evidence with biomarker outcomes, dose-response relationships, and toxicity thresholds to generate a comprehensive micronutrient panel reference and tiered treatment protocols.
---
2. Vitamin D Status and Supplementation
2.1 Equine Vitamin D Metabolism and Reference Ranges
Vitamin D metabolism in horses differs substantially from humans and other species. Research demonstrates that in horses, 25-hydroxyvitamin D2 (25(OH)D2) is the predominant vitamin D metabolite, with an apparent lack of endogenous vitamin D3 production [5]. In healthy unsupplemented grazing ponies, the median 25(OH)D2 concentration was 10.7 nmol/L, with significant seasonal variation—concentrations were significantly higher on long days (>12 hours light) compared to short days (14.4 vs. 8.7 nmol/L, p<0.01). Stabled Thoroughbreds receiving D3 supplementation had lower 25(OH)D2 concentrations than grazing ponies (7.4 vs. 12.6 nmol/L), indicating the importance of dietary D2 sources in horses.
The relationship between vitamin D and endocrine disorders in horses does not reflect that of other species [5]. Increasing ACTH was associated with lower 25(OH)D2 concentrations (p<0.01), while measures of increased adiposity were paradoxically associated with higher 25(OH)D2 concentrations—opposite to the pattern observed in humans. This species-specific biology has important implications for interpreting vitamin D status in horses with metabolic dysfunction.
2.2 Vitamin D Deficiency Prevalence and Clinical Implications
In comparative medical literature, vitamin D deficiency affects nearly half of the global human population and remains a widespread public health challenge [6]. Deficiency in vitamin D has been implicated in musculoskeletal weakness, frailty, cognitive decline, autoimmune diseases, and respiratory infections. While there is reasonable agreement that serum 25(OH)D levels <50 nmol/L (20 ng/mL) constitute deficiency and severe deficiency states (<25–30 nmol/L) should be avoided, consensus on optimal thresholds remains elusive [7].
Studies in humans with chronic musculoskeletal pain demonstrate vitamin D deficiency was significantly more prevalent in affected patients (56.7%) compared to controls (22.5%), with a significant inverse relationship between vitamin D levels and pain severity (r = −0.61, p < 0.001) [8]. In collegiate athletes, 47.2% were vitamin D insufficient with 17.6% being deficient, and for each 5 ng/mL decrease in vitamin D level, there was a 13% increase in odds of injury [9]. These findings support the relevance of vitamin D optimization in athletic animals including performance horses.
2.3 Supplementation Protocols and Dose-Response
Guidelines for vitamin D supplementation vary between 200 and 2000 IU/day in older adults, aiming to achieve circulating 25-hydroxyvitamin D of at least 50 nmol/L [10]. Moderate-dose vitamin D (3200 IU/day) compared to placebo may improve vitamin D status with mean difference of 13.60 ng/mL (95% CI: 7.94-19.26) [11]. In patients with degenerative musculoskeletal disease and low vitamin D status, administration of 40,000 IU of vitamin D2 weekly for 6 months increased median 25(OH)D from 24.9 to 43.1 ng/mL with significant improvements in muscle mass, balance, gait speed, and physical performance [12].
Factors affecting 25(OH)D response to supplementation include baseline 25(OH)D levels, body mass index, ethnicity, type of vitamin D (D2 or D3), and genetics [13]. The response differs between individuals, and baseline vitamin D, BMI, and supplement form are among the most important predictors of response.
| Parameter | Deficient | Insufficient | Sufficient | Optimal |
|---|---|---|---|---|
| 25(OH)D (ng/mL) | <20 | 20-29 | 30-50 | 50-80 |
| 25(OH)D (nmol/L) | <50 | 50-72 | 75-125 | 125-200 |
| Clinical Risk | High | Moderate | Low | Minimal |
3. Magnesium: Oral, Intravenous, and MSM Formulations
3.1 Magnesium Physiology and Reference Ranges
Magnesium is an essential mineral involved in cell function, energy metabolism, and neuromuscular activity. The normal concentrations of total plasma magnesium range from 0.7 to 1.0 mM (1.7-2.5 mg/dL), though it is difficult to diagnose hypomagnesemia based solely on plasma concentrations because only 1% of total body magnesium resides in extracellular fluids and merely 0.3% in plasma [14]. The long half-life (41-181 days for radio-labeled magnesium) required to equalize concentrations in body tissues makes accurate assessment challenging.
Magnesium blocks, reduces, or enhances the flow of ions by controlling several ion channels in cell membranes and is involved in cell division, metabolism, and genetic expression through activation of enzymes via ATP-Mg complex formation [14]. It acts as an anticonvulsant by blocking N-methyl-D-aspartate (NMDA) glutamate receptors and reduces acetylcholine release from presynaptic endings by inhibiting calcium entry, playing an antagonistic role against calcium at the neuromuscular junction.
3.2 Magnesium Supplementation Evidence
A Cochrane systematic review examining magnesium supplementation for skeletal muscle cramps identified 11 trials enrolling 735 individuals [15]. For idiopathic cramps in older adults, differences in cramp frequency when comparing magnesium to placebo were small and not statistically significant (MD -0.18 cramps/week, 95% CI -0.84 to 0.49; 5 studies, 307 participants; moderate-certainty evidence). The percentage of individuals experiencing ≥25% reduction in cramp rate was also not different (RR 1.04, 95% CI 0.84-1.29; high-certainty evidence).
Oral magnesium was associated primarily with gastrointestinal adverse events (e.g., diarrhea), experienced by 11-37% of participants [15]. The review concluded that magnesium supplementation is unlikely to provide clinically meaningful cramp prophylaxis to older adults with idiopathic skeletal muscle cramps, though results for pregnancy-associated cramps and disease-state-associated cramps remain inconclusive.
3.3 Methylsulfonylmethane (MSM) as Sulfur/Magnesium Alternative
Methylsulfonylmethane (MSM) has become a popular dietary supplement with anti-inflammatory applications. Research demonstrates that MSM supplementation improves various health-specific outcomes including inflammation, joint/muscle pain, oxidative stress, and antioxidant capacity [16]. As a Generally Recognized As Safe (GRAS) approved substance, MSM is well-tolerated at dosages up to four grams daily with few known and mild side effects. While MSM provides organic sulfur rather than magnesium directly, its role in reducing inflammation and oxidative stress may complement magnesium supplementation in neuromuscular conditions.
---
4. B-Complex Vitamins: Thiamine, Riboflavin, and Cobalamin
4.1 B-Vitamin Functions in Neuromuscular Metabolism
B-complex vitamins serve as essential cofactors in energy metabolism, neurotransmitter synthesis, and cellular function. Thiamine (B1) deficiency can result in profound neurological dysfunction and developmental delay, as demonstrated in cases of malnutrition where red cell transketolase activity (a marker of thiamine status) was severely reduced (0.36 vs. reference range 0.6-1.3) [17]. Vitamin B12 (cobalamin) deficiency presents with neurological manifestations and elevated urinary methylmalonic acid, with reference ranges typically 160-810 pmol/L in plasma.
In pregnant women with fetal neural tube defects, lower vitamin B12 levels and lower serum concentrations of inorganic phosphorus (which correlated directly with zinc levels) were observed compared to other pregnancy complications [18]. This highlights the interaction between B vitamins and trace minerals in neurological development and function.
4.2 B-Vitamin Status Assessment and Deficiency Prevalence
Studies in patients with phenylketonuria receiving amino acid formulas demonstrated that vitamin B12 deficiency occurred in 15.2% of PKU patients versus 30.6% of healthy controls [19]. The phenylalanine-free amino acid formulas resulted in adequate vitamin A and zinc levels but excess folic acid, vitamin B12, copper, and vitamin E values higher than required levels. These findings emphasize the importance of balanced supplementation rather than indiscriminate high-dose therapy.
In clinical trials examining micronutrient supplementation in children with ADHD, substantial nutrient deficiencies pre-treatment were observed for vitamin D (13%) and copper (15%), but most children entered trials with B12, folate, and other nutrient levels within expected ranges [20]. Lower pre-treatment folate and B12 levels predicted greater improvement with micronutrient therapy for some outcome measures, suggesting that those with relative deficiency may benefit most from supplementation.
4.3 Supplementation Protocols and Dose Considerations
For severe deficiency states, intramuscular B12 administration along with iron and thiamine supplementation has been shown to normalize levels [17]. The evidence supports targeted supplementation in confirmed deficiency rather than universal high-dose therapy. In the context of equine neuromuscular disease, thiamine requirements may increase during periods of high metabolic demand, strenuous exercise, or when carbohydrate metabolism is altered as in PSSM.
| B-Vitamin | Serum Reference | Deficiency Threshold | Typical Oral Dose (Equine) |
|---|---|---|---|
| Thiamine (B1) | Variable by assay | Transketolase <0.6 | 100-1000 mg/day |
| Riboflavin (B2) | 106-638 nmol/L | <106 nmol/L | 20-50 mg/day |
| Cobalamin (B12) | 160-810 pmol/L | <160 pmol/L | 10-100 µg/day |
5. Trace Minerals: Copper, Zinc, Iron, and Chromium
5.1 Reference Ranges in Equine Species
Serum trace element analysis in horses reveals significant inter-individual and breed variation. In Arabian horses and Hassawi donkeys, atomic absorption spectrometry demonstrated measurable concentrations of selenium, manganese, chromium, copper, iron, and zinc with significant differences between horses and donkeys in Se, Mn, and Cr [21]. Gender differences were also observed, with statistically significant differences in serum Se and Cr between male and female horses. These species and gender variations are important considerations when interpreting laboratory data.
In a study of horses aged 2 months to 26 years, correlations were detected between age and whole blood iron (r=0.62), selenium (r=0.76), and zinc (r=0.47), similar to patterns reported in horses using serum or plasma [22]. Trace elements including chromium and lead that were below detection levels in blood were detectable in mane hair, suggesting hair analysis may be useful for investigating suspected exposure to excessive trace minerals or heavy metals.
5.2 Copper and Zinc: Interactions and Clinical Significance
Copper deficiency can cause cytopenias presenting as anemia, neutropenia, and malaise, with bone marrow demonstrating cytoplasmic vacuolization of precursors [23]. Importantly, zinc supplementation can induce copper deficiency through competitive absorption inhibition. The interaction between copper and zinc requires careful consideration when supplementing either mineral.
Zinc and copper are important elements in regulating gene expression of metallothioneins, and zinc deficiency may be a risk factor for various pathological conditions [24]. In cattle studies, strong negative correlations were observed between parasitic burden (EPG) and serum concentrations of zinc, manganese, iron, and copper, with zinc identified as the strongest predictor of parasitic resistance [25]. This suggests that adequate zinc status may support immune function against parasitic challenges including protozoal infections relevant to EPM.
5.3 Iron Assessment and Supplementation Considerations
Iron status assessment in animals reveals complex patterns. Excess serum iron and nickel, combined with deficiency of copper, zinc, arsenic, cobalt, and chromium, can lead to impairments of mineral status with systemic effects on respiratory and hematopoietic systems [26]. The imbalance in ratios of Fe-Cu-Zn and Fe-Cu-Co negatively affects erythropoiesis, immune system formation, and antioxidant status.
Iron supplementation may be beneficial when functional anemia is present, but must be balanced against the risks of iron excess [27]. The relationship between body weight and iron levels remains contradictory in research, with some studies correlating obesity with iron deficiency while others indicate excess body mass may coexist with iron excess [28].
5.4 Chromium and Glucose Metabolism
Chromium plays an essential role in carbohydrate, protein, and lipid metabolism, characterized as a component of the amplification mechanism of insulin cellular signaling and a contributing factor to increased sensitivity of insulin receptors [29]. In cardiac and skeletal muscle, the chromium-influenced IGF-1 pathway stimulates glucose uptake and transportation as well as lactate production, while also stimulating RNA and protein synthesis from an anabolic perspective.
Given that Quarter Horses with PSSM have enhanced cellular uptake of glucose that may be caused by increased sensitivity to insulin [30], chromium supplementation warrants careful consideration. While chromium typically enhances insulin action, in already insulin-sensitive PSSM horses, additional chromium could theoretically exacerbate glycogen accumulation. Decreased chromium levels are associated with metabolic syndrome in humans, but growth responses to chromium supplementation have been inconsistent among different sources and inclusion rates [29].
---
6. Comprehensive Micronutrient Panel and Tiered Protocol
6.1 Recommended Diagnostic Panel
Based on the evidence reviewed, a comprehensive micronutrient assessment panel for horses with PSSM and EPM should include:
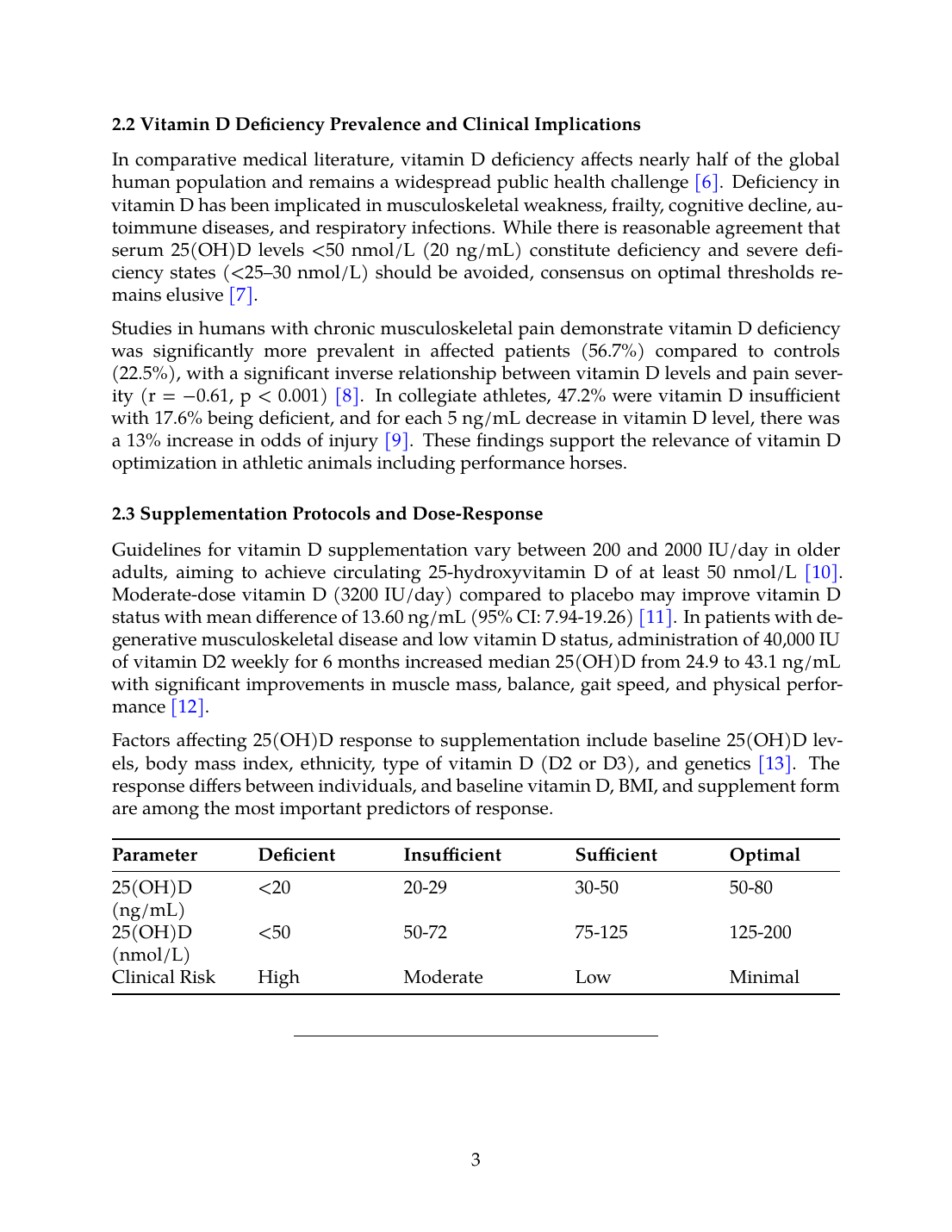
| Analyte | Sample Type | Normal Range (Equine) | Deficiency Threshold | Clinical Notes |
|---|---|---|---|---|
| 25(OH)D2 | Plasma | 8-15 nmol/L | <8 nmol/L | Seasonal variation; increases with pasture access |
| 25(OH)D3 | Plasma | Varies with supplementation | N/A | Only detectable with D3 supplementation |
| Magnesium (total) | Serum | 1.7-2.5 mg/dL | <1.7 mg/dL | Poor reflection of intracellular status |
| Copper | Serum | 0.6-1.5 µg/mL | <0.6 µg/mL | Consider ceruloplasmin assay |
| Zinc | Serum | 0.7-1.4 µg/mL | <0.7 µg/mL | Ensure Cu:Zn ratio 1:3 to 1:5 |
| Iron | Serum | 0.9-2.3 µg/mL | <0.9 µg/mL | Include TIBC, ferritin if available |
| Chromium | Whole blood/Hair | Below detection-0.01 µg/mL | N/A | Hair analysis more sensitive |
| Vitamin B12 | Serum | 200-500 pg/mL | <200 pg/mL | Consider methylmalonic acid |
| Thiamine | Red cell transketolase | 0.6-1.3 activity ratio | <0.6 | Functional assay preferred |
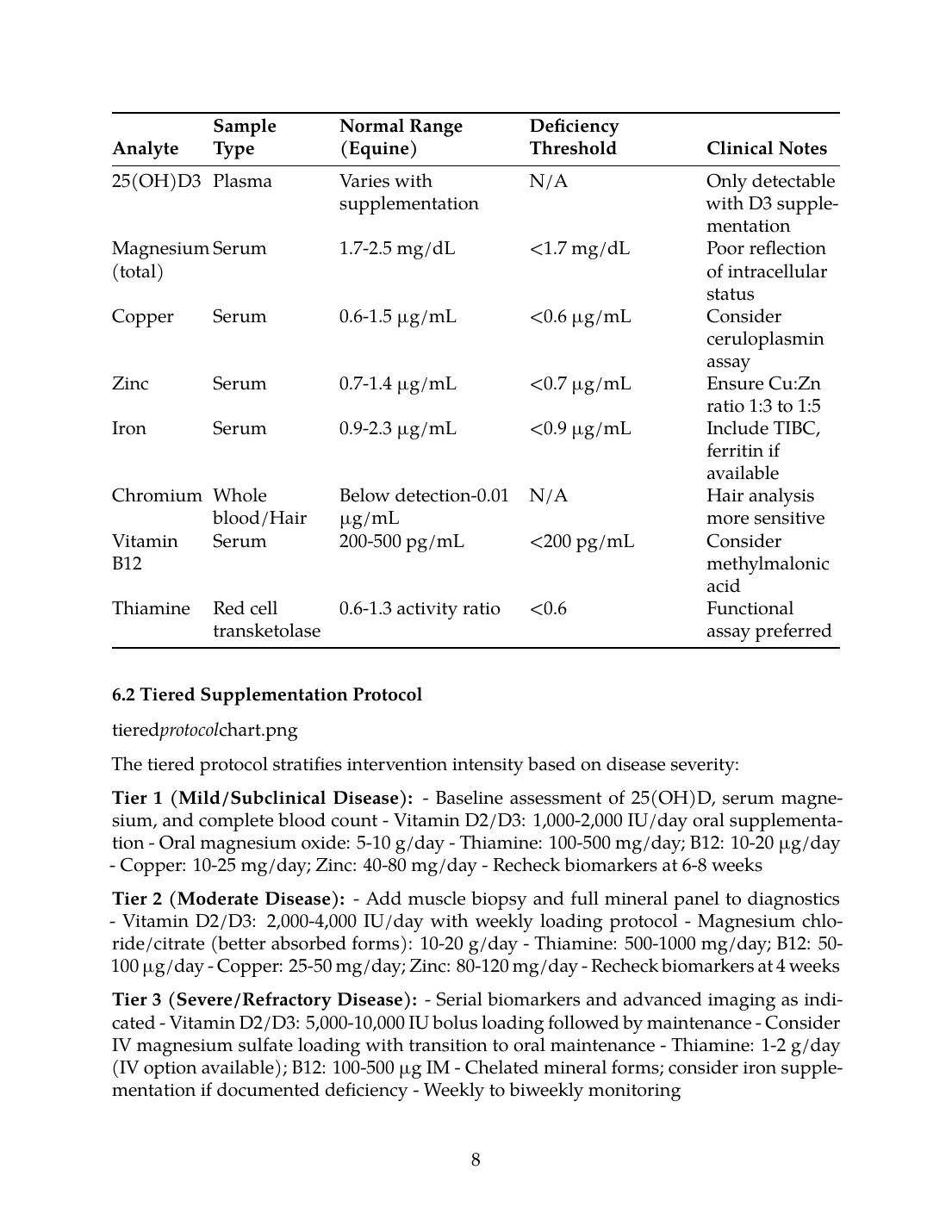
6.2 Tiered Supplementation Protocol
The tiered protocol stratifies intervention intensity based on disease severity:
Tier 1 (Mild/Subclinical Disease):
- Baseline assessment of 25(OH)D, serum magnesium, and complete blood count
- Vitamin D2/D3: 1,000-2,000 IU/day oral supplementation
- Oral magnesium oxide: 5-10 g/day
- Thiamine: 100-500 mg/day; B12: 10-20 µg/day
- Copper: 10-25 mg/day; Zinc: 40-80 mg/day
- Recheck biomarkers at 6-8 weeks
Tier 2 (Moderate Disease):
- Add muscle biopsy and full mineral panel to diagnostics
- Vitamin D2/D3: 2,000-4,000 IU/day with weekly loading protocol
- Magnesium chloride/citrate (better absorbed forms): 10-20 g/day
- Thiamine: 500-1000 mg/day; B12: 50-100 µg/day
- Copper: 25-50 mg/day; Zinc: 80-120 mg/day
- Recheck biomarkers at 4 weeks
Tier 3 (Severe/Refractory Disease):
- Serial biomarkers and advanced imaging as indicated
- Vitamin D2/D3: 5,000-10,000 IU bolus loading followed by maintenance
- Consider IV magnesium sulfate loading with transition to oral maintenance
- Thiamine: 1-2 g/day (IV option available); B12: 100-500 µg IM
- Chelated mineral forms; consider iron supplementation if documented deficiency
- Weekly to biweekly monitoring
6.3 Special Considerations for PSSM Management
For horses with PSSM specifically, dietary management remains the cornerstone of therapy. Research demonstrates that diets with <5% digestible energy from starch and >12% digestible energy from fat can reduce exertional rhabdomyolysis by increasing availability of free fatty acids for muscle metabolism [31]. The dietary modification is effective because PSSM horses have increased rate of glucose clearance in response to insulin secretion, making low soluble carbohydrate diets the most effective approach to decrease glycogen accumulation [30].
With the recommended diet and exercise regime, 80% of PSSM Warmblood owners reported overall improvement with significant decreases in decline in performance and rhabdomyolysis [32]. However, 53% of horses were still not advancing as expected, with reluctance to go forward and collect persisting in approximately one-third of horses. This incomplete response suggests that additional factors—potentially including micronutrient status—may influence outcomes.
6.4 Toxicity Thresholds and Safety Monitoring
| Micronutrient | Upper Tolerance Level | Toxicity Signs | Monitoring Recommendation |
|---|---|---|---|
| Vitamin D | ~10,000 IU/day (horses) | Hypercalcemia, soft tissue calcification | Serum calcium, 25(OH)D at 4-6 weeks |
| Magnesium (IV) | Variable | Respiratory depression, cardiac effects | Monitor deep tendon reflexes during infusion |
| Copper | ~100 mg/day (horses) | Hepatotoxicity, hemolysis | Liver enzymes periodically |
| Zinc | ~500 mg/day (horses) | Copper deficiency, GI upset | Monitor Cu:Zn ratio |
| Iron | Avoid excess | Oxidative stress, hepatotoxicity | TIBC, ferritin; avoid in iron replete animals |
| Chromium | Limited toxicity data | Unknown in horses | Generally low concern with organic forms |
---
7. Conclusions and Future Directions
7.1 Summary of Evidence
This systematic review reveals that while vitamin E and selenium remain central to nutritional management of equine neuromuscular diseases, other micronutrients deserve clinical attention. Vitamin D metabolism in horses uniquely favors D2 metabolites, with seasonal and management influences requiring consideration when assessing status and planning supplementation. Magnesium supplementation evidence for muscle cramps remains equivocal, though its role in neuromuscular function is well-established physiologically. B-complex vitamins serve critical roles as metabolic cofactors, with thiamine and cobalamin most relevant to neuromuscular pathology. Trace mineral interactions, particularly the Cu-Zn relationship and Fe-Cu-Zn ratios, require balanced supplementation approaches.
7.2 Limitations and Gaps in Evidence
The literature reveals significant gaps in equine-specific research for most micronutrients beyond vitamin E and selenium. Direct evidence for supplementation efficacy in PSSM and EPM cohorts is sparse, necessitating extrapolation from human and other veterinary species data. Reference ranges for horses vary by breed, age, gender, and methodology, complicating interpretation. The genetic basis of Type 2 PSSM remains unknown, limiting targeted nutritional intervention strategies [32].
7.3 Research Priorities
Future research should prioritize: (1) establishing equine-specific reference ranges for vitamin D metabolites, B-vitamins, and trace minerals across breeds and management conditions; (2) prospective clinical trials examining micronutrient supplementation as adjunctive therapy in PSSM and EPM; (3) investigation of biomarker responses to tiered supplementation protocols; and (4) pharmacokinetic studies of vitamin D, magnesium, and B-vitamins in horses to optimize dosing strategies.
7.4 Clinical Recommendations
Based on available evidence, clinicians managing horses with PSSM and EPM should:
- Implement comprehensive micronutrient assessment beyond vitamin E and selenium
- Apply species-specific interpretation of vitamin D status, recognizing D2 predominance in horses
- Balance trace mineral supplementation with attention to Cu:Zn ratios and iron status
- Utilize tiered intervention protocols stratified by disease severity
- Monitor biomarker responses to guide dose adjustments
- Maintain dietary modification as the foundation of PSSM management while optimizing micronutrient status
The integration of expanded micronutrient assessment and supplementation into clinical protocols offers potential for improved outcomes in these challenging conditions, though definitive evidence from controlled equine trials remains a priority for future investigation.
References
References
Numbered in order of first citation, IEEE style. Each entry links to the original source via DOI or publisher URL.
- A. Firshman and S. Valberg, “Polysaccharide Storage Myopathy.,” The Veterinary clinics of North America. Equine practice, Jan. 2025. doi: 10.1016/j.cveq.2024.11.004. View source
- R. Geor, “Role of dietary energy source in the expression of chronic exertional myopathies in horses,” Journal of Animal Science, Jun. 2005. doi: 10.2527/2005.8313_SUPPLE32X. View source
- B. D. F. Dallo, L. X. R. D. Silva, L. A. O. D. Paula, N. F. P. E. Silva, C. Smaniotto, G. M. Pagliosa, A. D. M. Viott, and M. D. C. S. Lopes, “Mieloencefalopatia Degenerativa Equina: Características Clínicas e Histopatológicas e Diagnósticos Diferenciais,” Acta Scientiae Veterinariae, Nov. 2023. doi: 10.22456/1679-9216.130299. View source
- E. Barrey, E. Mucher, N. Jeansoule, T. Larcher, L. Guigand, B. Herszberg, S. Chaffaux, G. Guérin, X. Mata, P. Benech, M. Canale, O. Alibert, P. Maltere, and X. Gidrol, “Gene expression profiling in equine polysaccharide storage myopathy revealed inflammation, glycogenesis inhibition, hypoxia and mitochondrial dysfunctions,” BMC Veterinary Research, Aug. 2009. doi: 10.1186/1746-6148-5-29. View source
- M. Dosi, B. McGorum, R. D. Kirton, E. Cillán-García, R. Mellanby, J. Keen, E. Hurst, and R. Morgan, “The effect of season, management and endocrinopathies on vitamin D status in horses.,” Equine Veterinary Journal, Aug. 2022. doi: 10.1111/evj.13873. View source
- D. E. Mudiyanselage, C. Ouyang, R. D. Jin, S. Velmurugan, Y. Jiang, J. Sun, and D. Ma, “Vitamin D deficiency and disease conditions relevant to: Orthopaedic translation.,” Journal of Orthopaedic Translation, Mar. 2026. doi: 10.1016/j.jot.2026.101061. View source
- E. Casado, E. Costa, P. Mezquita-Raya, R. Andújar-Espinosa, and J. Neyro, “Calcifediol in the management of vitamin D deficiency-related skeletal and extraskeletal diseases: overview and clinical cases,” Drugs in Context, Sep. 2023. doi: 10.7573/dic.2023-5-4. View source
- D. Kumar and V. Vijh, “Vitamin D Deficiency and Its Association with Musculoskeletal Pain,” Acta Medica International, Dec. 2025. doi: 10.21276/amit.2025.v12.i3.256. View source
- A. Frank, M. R. Joachim, J. L. Sanfilippo, C. J. Call, S. Hetzel, M. A. Brooks, and A. M. Spiker, “Low Vitamin D Levels Are Associated With Increased Risk of Musculoskeletal Injuries in Collegiate Athletes.,” Sports health, Mar. 2026. doi: 10.1177/19417381251407674. View source
- T. Chevalley, M. Brandi, K. Cashman, E. Cavalier, N. Harvey, S. Maggi, C. Cooper, N. Al-Daghri, O. Bock, O. Bruyère, M. Rosa, B. Cortet, A. Cruz-Jentoft, A. Cherubini, B. Dawson-Hughes, R. Fielding, N. Fuggle, P. Halbout, J. Kanis, J. Kaufman, O. Lamy, A. Laslop, M. C. P. Yerro, R. Radermecker, J. Thiyagarajan, T. Thomas, N. Veronese, M. D. Wit, J. Reginster, and R. Rizzoli, “Role of vitamin D supplementation in the management of musculoskeletal diseases: update from an European Society of Clinical and Economical Aspects of Osteoporosis, Osteoarthritis and Musculoskeletal Diseases (ESCEO) working group,” Aging Clinical and Experimental Research, Oct. 2022. doi: 10.1007/s40520-022-02279-6. View source
- M. Chakhtoura, N. Nakhoul, E. Akl, B. Safadi, C. S. Mantzoros, M. Metzendorf, and G. E. Fuleihan, “Oral vitamin D supplementation for adults with obesity undergoing bariatric surgery.,” Cochrane Database of Systematic Reviews, Oct. 2024. doi: 10.1002/14651858.CD011800.pub2. View source
- S. Dechsupa, W. Yingsakmongkol, W. Limthongkul, W. Singhatanadgige, S. Assawakosri, and S. Honsawek, “Vitamin D Supplementation Improves Muscle Mass, Physical Function, and Quality of Life in Patients With Degenerative Lumbar Disease,” Clinical and Translational Science, Aug. 2025. doi: 10.1111/cts.70315. View source
- H. Mazahery and P. V. von Hurst, “Factors Affecting 25-Hydroxyvitamin D Concentration in Response to Vitamin D Supplementation,” Nutrients, Jun. 2015. doi: 10.3390/nu7075111. View source
- J. Han, “About uses of magnesium during perioperative period,” Korean Journal of Anesthesiology, Jun. 2012. doi: 10.4097/kjae.2012.62.6.509. View source
- S. R. Garrison, C. Korownyk, M. Kolber, G. Allan, V. Musini, R. K. Sekhon, and N. Dugré, “Magnesium for skeletal muscle cramps.,” Cochrane Database of Systematic Reviews, Sep. 2020. doi: 10.1002/14651858.CD009402.pub3. View source
- M. Butawan, R. Benjamin, and R. Bloomer, “Methylsulfonylmethane: Applications and Safety of a Novel Dietary Supplement,” Nutrients, Mar. 2017. doi: 10.3390/nu9030290. View source
- E. Mcintyre, J. Wilcox, J. McGill, and P. Lewindon, “Silver Toxicity in an Infant of Strict Vegan Parents,” Journal of Pediatric Gastroenterology and Nutrition - JPGN, Oct. 2001. doi: 10.1097/00005176-200110000-00016. View source
- Y. Milyutina, M. O. Shengelia, A. P. Sazonova, O. Bespalova, and A. Korenevsky, “The relationship between mineral and vitamin statuses in the blood serum of pregnant women with fetal congenital malformations,” Journal of obstetrics and women''s diseases, Jan. 2025. doi: 10.17816/jowd639031. View source
- E. Kose and N. Arslan, “Vitamin/mineral and micronutrient status in patients with classical phenylketonuria.,” Clinical Nutrition, Feb. 2018. doi: 10.1016/j.clnu.2018.01.034. View source
- J. Rucklidge, M. J. F. Eggleston, K. A. Darling, A. Stevens, M. Kennedy, and C. Frampton, “Can we predict treatment response in children with ADHD to a vitamin‐mineral supplement? An investigation into pre‐treatment nutrient serum levels, MTHFR status, clinical correlates and demographic variables,” Progress in Neuro-psychopharmacology and Biological Psychiatry, Mar. 2019. doi: 10.1016/j.pnpbp.2018.09.007. View source
- T. Shawaf, F. Almathen, A. Meligy, W. El-Deeb, and S. Al-Bulushi, “Biochemical analysis of some serum trace elements in donkeys and horses in Eastern region of Kingdom of Saudi Arabia,” Veterinary World, Oct. 2017. doi: 10.14202/vetworld.2017.1269-1274. View source
- M. Brummer-Holder, B. Cassill, and S. Hayes, “Interrelationships Between Age and Trace Element Concentration in Horse Mane Hair and Whole Blood.,” Journal of Equine Veterinary Science, Apr. 2020. doi: 10.1016/j.jevs.2020.102922. View source
- S. Morales, A. Nael, and D. Buchbinder, “Copper Deficiency and Cytopenias.,” Journal of pediatric hematology/oncology, Jan. 2021. doi: 10.1097/MPH.0000000000002047. View source
- S. M. Awadh, Z. Yaseen, and M. S. Al-Suwaiyan, “The role of environmental trace element toxicants on autism: A medical biogeochemistry perspective.,” Ecotoxicology and Environmental Safety, Jan. 2023. doi: 10.1016/j.ecoenv.2023.114561. View source
- A. Miresa, T. Tolemariam, B. Duguma, E. S. Dierenfeld, A. Nigussie, and F. Begna, “Association between serum mineral concentrations and gastrointestinal parasite burden in zebu cattle accessing ‘hora’ mineral water in southwestern Ethiopia,” PLoS ONE, Aug. 2025. doi: 10.1371/journal.pone.0329812. View source
- E. Kalaeva, V. Kalaev, A. Chernitskiy, M. Alhamed, and V. Safonov, “Incidence risk of bronchopneumonia in newborn calves associated with intrauterine diselementosis,” Veterinary World, May 2020. doi: 10.14202/vetworld.2020.987-995. View source
- C. V. D. Venter, C. Konn, M. Doherty, and R. Allan Purdy, “The Role of Minerals in COVID-19: An Umbrella Review,” CAND Journal, Dec. 2023. doi: 10.54434/candj.164. View source
- W. Banach, K. Nitschke, N. Krajewska, W. Mongiałło, O. Matuszak, J. Muszyński, and D. Skrypnik, “The Association between Excess Body Mass and Disturbances in Somatic Mineral Levels,” International Journal of Molecular Sciences, Oct. 2020. doi: 10.3390/ijms21197306. View source
- P. S. A. Moreira, C. Palhari, and R. C. Berber, “Dietary chromium and growth performance animals: a review,” Jul. 2020. doi: 10.36560/13720201151. View source
- F. D. De La Côrte, S. Valberg, J. MacLeay, S. Williamson, and J. R. Mickelson, “Glucose uptake in horses with polysaccharide storage myopathy.,” American Journal of Veterinary Research, Apr. 1999. doi: 10.2460/ajvr.1999.60.04.458. View source
- W. P. Ribeiro, S. Valberg, J. D. Pagan, and B. Gustavsson, “The effect of varying dietary starch and fat content on serum creatine kinase activity and substrate availability in equine polysaccharide storage myopathy.,” Journal of Veterinary Internal Medicine, Nov. 2004. doi: 10.1111/j.1939-1676.2004.tb02637.x. View source
- Z. J. Williams, M. Bertels, and S. Valberg, “Muscle glycogen concentrations and response to diet and exercise regimes in Warmblood horses with type 2 Polysaccharide Storage Myopathy,” PLoS ONE, Sep. 2018. doi: 10.1371/journal.pone.0203467. View source
This review is one of 34 in the Foxwatch research corpus. Methodology: PRISMA 2020-style systematic literature review against PubMed, CAB Abstracts, Scopus, Web of Science, and equine veterinary conference proceedings.