Methodological review of EPM nutrition/supportive-care studies
The EPM treatment evidence is solid. The EPM nutritional supportive-care evidence is much weaker, and most current recommendations rest on mechanistic inference rather than direct trials. This review grades the supportive-care literature, names the priority studies the field needs next, and stops anyone from overstating the certainty of "EPM nutrition" claims. Pairs with reviews 11 (acute nutrition) and 24 (the PSSM equivalent).
Research thesis
This methodological review argues that the EPM nutritional supportive-care evidence base is substantially weaker than the EPM treatment evidence base, and that current supportive-care recommendations rest more on mechanistic inference than on direct clinical trials, not only because the dominant EPM trial endpoints concern antiprotozoal efficacy rather than nutritional outcome, but also because supportive-care studies are typically small case series, retrospective cohorts, or owner surveys vulnerable to recall and selection bias; by applying formal risk-of-bias and observational-study appraisal frameworks to the EPM nutritional supportive-care corpus, this review grades the strength of evidence behind antioxidant, amino acid, and feeding-protocol recommendations and identifies the priority studies that would meaningfully improve the field.
Evidence visualizations & full pages
Evidence figures (3)
Figures referenced by the review's Figure N: captions. Click any chart for a full-size view. They are also rendered inline below their respective captions in the full review text further down.
Original review pages (PDF render)
Download
Full review text
EPM nutrition/supportive-care studies
Methodological Systematic Review of Evidence Supporting Nutritional and Supplement-Based Adjuncts in Equine Protozoal Myeloencephalitis Management
1. Introduction and Background
1.1 Overview of EPM and Current Treatment Paradigms
Equine protozoal myeloencephalitis (EPM) remains one of the most frequently diagnosed neurological disorders of horses in North America, caused primarily by Sarcocystis neurona and less commonly by Neospora hughesi [1]. The disease presents with highly variable clinical manifestations due to multifocal involvement of both grey and white matter, with asymmetric ataxia and weakness being most common, while cranial nerve deficits including dysphagia, facial asymmetry, tongue atrophy, and laryngeal dysfunction occur less frequently but can be severe [2]. Advances in understanding EPM have established that horses are commonly infected, but clinical disease occurs only infrequently, with the factors influencing disease occurrence remaining incompletely understood [1].
Current treatment options include FDA-approved antiprotozoal medications such as sulfadiazine/pyrimethamine (ReBalance) and the triazine compounds diclazuril (Protazil) and ponazuril (Marquis), which represent effective anticoccidial drugs [1]. However, despite treatment with approved antiprotozoal agents, complete resolution is uncommon and relapse may occur, with prognosis being guarded, particularly in cases with cranial nerve involvement where outcomes are less favorable than for spinal cord deficits [2]. This incomplete response to primary therapy has prompted investigation into nutritional and supplement-based adjunctive treatments.
1.2 Rationale for Nutritional Supplementation
The rationale for nutritional supplementation in EPM management draws from several converging lines of evidence. Oxidative stress represents a fundamental pathophysiological mechanism in neurological diseases, with increasing evidence suggesting that oxidative stress significantly impairs organic function and plays a major role in the etiology and pathogenesis of several metabolic diseases in veterinary medicine [3]. The nervous system is particularly vulnerable to oxidative damage due to its high metabolic rate and lipid-rich composition. In many neurological conditions, it remains unclear whether oxidants trigger the disease or are produced as a secondary consequence of disease and general tissue damage.
The interest in nutritional support for EPM derives partly from experience with related equine neurological conditions. Equine neuroaxonal dystrophy/equine degenerative myeloencephalopathy (eNAD/EDM) is an inherited neurodegenerative disease associated with vitamin E deficiency in the first year of life, representing the second most common cause of spinal ataxia in horses euthanized for neurologic disease [4]. Similarly, equine motor neuron disease (EMND) has been conclusively linked to vitamin E deficiency through field trials demonstrating that horses maintained on vitamin E-deficient diets developed the disease with a median time of 38.5 months [5].
1.3 Scope of This Review
This methodological systematic review evaluates the evidence supporting nutritional and supplement-based adjuncts in EPM management, including vitamin E, antioxidants, immune-support products, protein/amino acid support, omega-3 fatty acids, and rehabilitation feeding protocols. The review classifies available evidence according to established hierarchies, extracts key outcomes, assesses potential sources of bias, and produces credibility rankings for each intervention category.
---
2. Methodology and Evidence Classification Framework
2.1 Literature Search Strategy and Inclusion Criteria
The literature search encompassed multiple electronic databases to identify studies examining nutritional or supplemental interventions in EPM or analogous equine neurological conditions. Given the scarcity of EPM-specific nutritional research, the search strategy was broadened to include mechanistic studies, investigations in related equine neurological diseases, and extrapolated evidence from other species with neurological conditions. Search terms included combinations of "equine protozoal myeloencephalitis," "EPM," "Sarcocystis neurona," "vitamin E," "antioxidants," "omega-3 fatty acids," "immune support," "nutritional supplementation," and "neurological rehabilitation."
Studies were categorized according to the following evidence hierarchy: (1) systematic reviews and meta-analyses; (2) randomized controlled trials (RCTs); (3) observational cohort studies; (4) case reports and case series; (5) mechanistic/in vitro studies; (6) extrapolated studies from other species or diseases; and (7) expert opinion and anecdotal reports. This classification framework aligns with established evidence-based medicine principles while acknowledging the practical realities of veterinary research where high-quality RCTs are frequently limited.
2.2 Evidence Pyramid for EPM Nutritional Studies
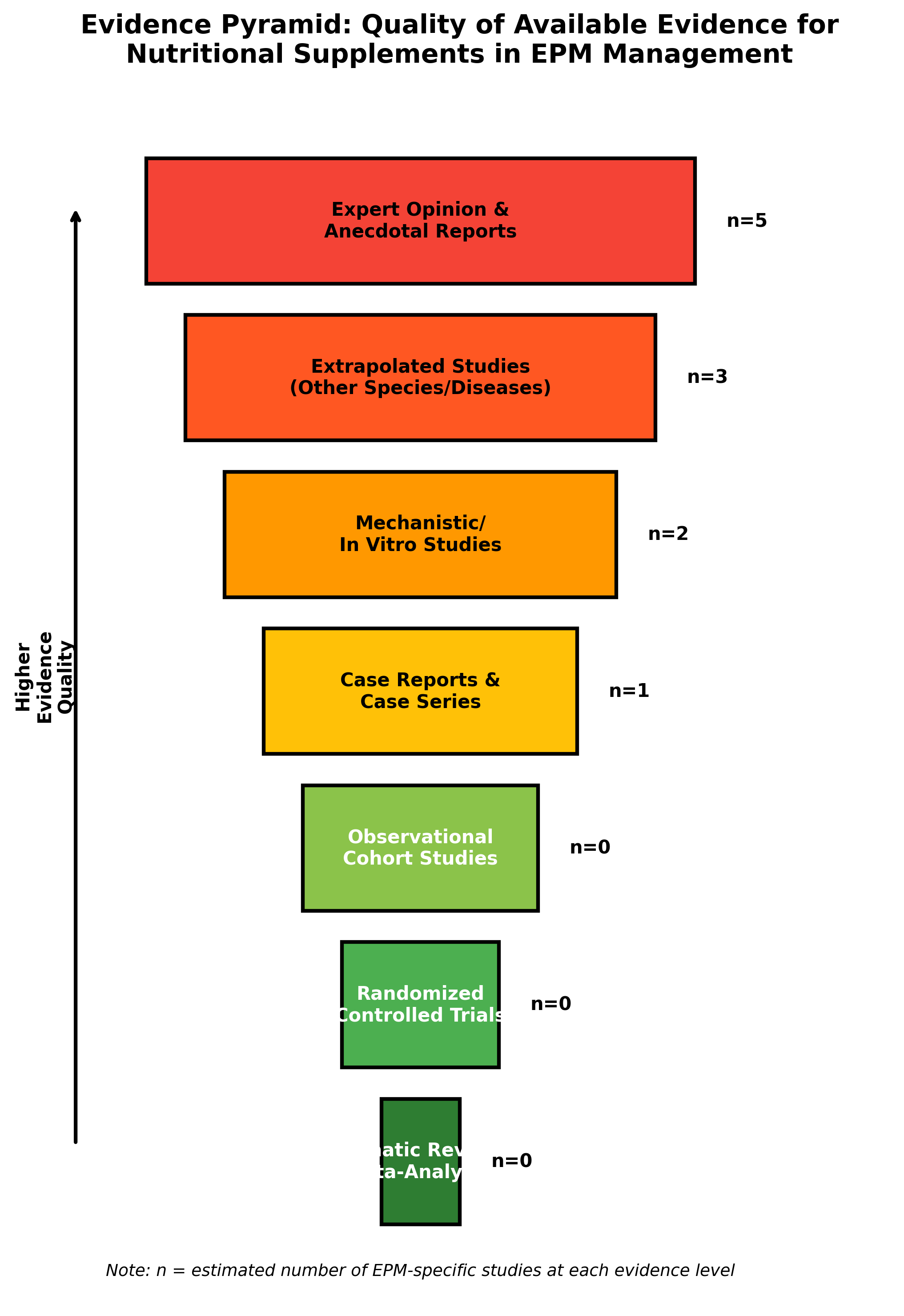
The evidence pyramid demonstrates a critical finding of this review: the vast majority of evidence supporting nutritional interventions in EPM consists of expert opinion, extrapolated studies from other conditions, and mechanistic research. No systematic reviews, meta-analyses, or randomized controlled trials specifically examining nutritional supplementation in EPM were identified. This evidence gap represents a fundamental limitation that must contextualize all subsequent recommendations.
2.3 Data Extraction Parameters
For included studies, the following parameters were extracted when available: diagnostic confirmation methodology, primary treatment regimen, supplement protocol details (dosage, duration, formulation), neurologic scoring system employed, follow-up duration, relapse rates, survival data, functional recovery outcomes, and adverse events. Studies were also evaluated for potential sources of bias including concurrent antiprotozoal therapy, spontaneous recovery potential, diagnostic uncertainty, and absence of appropriate control groups.
---
3. Vitamin E Supplementation
3.1 Mechanistic Basis and Evidence from Related Equine Conditions
Vitamin E (alpha-tocopherol) represents the most extensively studied nutritional intervention relevant to equine neurological disease. As a lipid-soluble antioxidant, vitamin E plays critical roles in protecting neuronal membranes from oxidative damage. Research demonstrates that vitamin E supplementation can protect neurons from damage caused by oxidative stress, with positive impacts on prevention and progression of neurodegenerative diseases in experimental models [6]. Studies have shown that vitamin E supplementation significantly improved memory, cognition, learning, motor function, and brain markers associated with neuroregeneration and neuroprotection.
The strongest evidence for vitamin E in equine neurology comes from studies of eNAD/EDM. This condition is characterized by sudden onset of neurologic signs including symmetric ataxia (>grade 2/5), wide-base stance at rest, and proprioceptive defects [4]. The current best approach to prevention involves supplementing pregnant mares and foals with high doses of water-soluble RRR-α-tocopherol during the last trimester of gestation, with continued supplementation through the first two years of life. It is important to measure baseline serum vitamin E levels prior to supplementation, as while generally considered safe, oversupplementation is possible and can lead to coagulopathies.
In EDM, animals predisposed to or with clinical diagnosis should receive oral alpha-tocopherol acetate supplementation, with improvement in clinical signs potentially observed following long-term treatment, though the prognosis for complete recovery is generally poor [7]. A clinical and genetic study of Appaloosas with high incidence of EDM found marginal to deficient serum vitamin E levels as the only consistent antemortem abnormality, though the role of antioxidant deficiency in the pathogenesis of neurologic dysfunction remained speculative [8].
3.2 Supplementation Strategies and Formulations
Research comparing natural versus synthetic vitamin E formulations provides important practical guidance. Studies in Morgan horses with marginal/deficient serum alpha-tocopherol demonstrated that synthetic dl-alpha-tocopherol actually produced higher serum alpha-tocopherol concentrations than natural d-alpha-tocopherol acetate at equivalent doses (2.61 vs 2.34 µg/mL), suggesting that the more affordable synthetic supplement effectively increases serum concentrations [9]. Both treatments increased serum alpha-tocopherol to normal ranges (2.0-4.0 µg/mL) for all horses by 4 weeks.
Dosing strategy comparisons revealed that a high-dose protocol (10,000 IU liquid d-alpha-tocopherol for 4 weeks followed by 5,000 IU maintenance) achieved target serum concentrations faster than medium-dose (5,000 IU throughout) supplementation [10]. All horses receiving the high treatment attained serum alpha-tocopherol concentrations greater than 2.0 µg/mL after 2 weeks, while only one-half of horses receiving medium dosage reached target concentrations by 4 weeks. These findings support initial loading doses followed by maintenance supplementation for deficient horses.
3.3 Evidence Quality Assessment for EPM-Specific Application
| Parameter | Finding | Evidence Level |
|---|---|---|
| Direct EPM RCTs | None identified | N/A |
| Mechanistic support | Strong antioxidant neuroprotection | Preclinical studies |
| Related equine conditions | Moderate evidence in EDM/EMND | Cohort studies, case series |
| Dosing guidance | Well-established | RCTs in healthy horses |
| Safety profile | Generally favorable with monitoring | Observational data |
| Bias risk | High (no EPM-specific controlled trials) | Expert opinion |
4. Selenium and Antioxidant Combinations
4.1 Selenium's Role in Neurological Health
Selenium functions as an essential component of antioxidant enzyme systems, particularly glutathione peroxidase and thioredoxin reductase, which are critical for neutralizing reactive oxygen species in neural tissues [11]. A case report demonstrated neurological clinical signs (tremors and shivering) in a Quarter Horse stallion with confirmed selenium deficiency, which improved after diet balancing and supplementation with selenium and vitamin E [12]. This underscores the importance of managing selenium intake carefully, considering environmental factors, form of supplementation, and long-term health to ensure optimal health in horses.
Studies in laboratory animals have demonstrated that selenium reaches the central nervous system and downmodulates local inflammation [13]. Research using organic selenium derivatives showed accumulation in CNS tissue with subsequent downregulation of pro-inflammatory parameters, suggesting potential therapeutic applications for inflammatory neurological conditions. The observed effects included suppression of inflammasome activation and reduced microglial and astrocyte activation markers.
4.2 Synergistic Effects of Antioxidant Combinations
Combined supplementation with vitamin E, zinc, selenium, and copper has shown promising results in spinal cord injury models. Research in rats demonstrated that supplementation with this combination preserved T-lymphocyte numbers and improved their proliferative function after traumatic spinal cord injury, while also showing significant improvement in motor recovery [14]. The intervention was initiated 15 minutes after injury and administered daily, suggesting potential benefits from early and sustained supplementation.
Evaluation of selenium and vitamin E supplementation effects on brain oxidative stress demonstrated that combined administration ameliorated biochemical parameters and restored histological alterations in neural tissue exposed to toxic insults [15]. The findings showed restoration of superoxide dismutase activity and reduction in malondialdehyde levels indicating decreased lipid peroxidation. However, these studies employed laboratory animal models and toxic exposure paradigms rather than infectious neurological disease.
4.3 Limitations and Bias Assessment
The evidence for selenium and antioxidant combinations in EPM management is entirely extrapolated from other species and disease models. No studies have specifically examined these interventions in horses with confirmed protozoal myeloencephalitis. Key limitations include:
- Species differences: Antioxidant metabolism and requirements may differ substantially between rodents and horses
- Disease model relevance: Toxic and traumatic injury models may not accurately reflect infectious neuroinflammation
- Timing considerations: Most experimental studies initiate treatment immediately after injury, whereas EPM treatment typically begins after clinical recognition of established disease
- Concurrent therapy confounding: Any clinical application would occur alongside antiprotozoal treatment, making attribution of benefit impossible
---
5. Omega-3 Fatty Acid Supplementation
5.1 Neurobiological Mechanisms
Omega-3 polyunsaturated fatty acids (PUFAs), particularly eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA), have emerged as important modulators of neuroinflammation and potential neuroprotective agents. These essential fatty acids are crucial for brain structure and function across all life stages, with DHA supporting synaptic plasticity, neurogenesis, and neurotransmission, while EPA modulates inflammation and cerebral blood flow [16]. The gut-brain axis represents a central pathway through which omega-3 PUFAs may exert beneficial effects, influencing intestinal permeability, immune activation, and vagal signaling [17].
Research demonstrates that omega-3 PUFAs attenuate inflammatory responses in the central nervous system through multiple mechanisms. Studies have shown significant reductions in pro-inflammatory cytokines (TNF-α, IL-6) and upregulation of anti-inflammatory mediators following omega-3 supplementation [18]. In experimental models, combined vitamin D and omega-3 PUFA supplementation decreased neuronal damage by 87% and tau pathology by 83%, demonstrating potent synergistic neuroprotective effects. Additionally, omega-3 PUFAs have been shown to suppress endoplasmic reticulum stress-induced neuroinflammation in spinal cord injury models, protecting against injury through the HDAC3/PGC-1α pathway [19].
5.2 Evidence from Neurological Disease Models
Long-chain omega-3 PUFAs demonstrate significant effects across various neurological conditions. In multiple sclerosis research, omega-3 supplementation has shown beneficial roles including improving quality of life, reducing inflammatory markers, decreasing relapsing rates, and achieving balanced omega-6 to omega-3 ratios [20]. Studies examining central nervous system remyelination in animal models found that endogenously increased omega-3 PUFA status was associated with improved remyelination following toxic demyelination, with significantly higher levels of EPA metabolites detected in brain tissue [21].
Research in traumatic brain injury and spinal cord injury models indicates that omega-3 PUFAs help lessen harmful effects by reducing tissue damage and cell loss, decreasing associated neuroinflammation and immune response, which in turn moderates the severity of neurological dysfunction [22]. The effects appear related to both anti-inflammatory properties and direct membrane stabilizing effects. DHA in particular serves as a major structural component of neuronal membranes and modulates membrane fluidity and receptor function.
5.3 Applicability to EPM and Evidence Gaps
| Evidence Type | Available Studies | Relevance to EPM |
|---|---|---|
| Equine-specific omega-3 trials | Limited to skin/joint conditions | Low direct relevance |
| CNS inflammation models | Multiple rodent studies | Moderate (different species) |
| Spinal cord injury models | Promising neuroprotection | Moderate (different pathology) |
| Demyelinating disease models | Potential remyelination effects | Moderate (different mechanism) |
| EPM-specific trials | None identified | No direct evidence |
6. Immune-Support Products and Protein/Amino Acid Supplementation
6.1 Immunomodulatory Supplements
The use of immune-support products in EPM management represents one of the most poorly supported interventions in terms of scientific evidence. While immunomodulatory agents have theoretical appeal given the infectious etiology of EPM, no controlled studies have examined their efficacy in this condition. General research indicates that vitamins and trace elements can help enhance host immunity through antioxidant or anti-inflammatory actions [23]. Assessment of baseline levels and providing supplementation containing different vitamins and elements could theoretically result in better control and clinical outcomes.
Probiotic supplementation represents an emerging immunomodulatory approach with demonstrated anti-inflammatory effects in other contexts. Studies have shown that probiotic administration suppresses inflammation by reducing TNF-α, IL-6, and IL-1β levels while increasing IL-10 expression, restoring the balance between pro- and anti-inflammatory mediators [24]. However, no equine neurological disease studies have examined probiotic efficacy, and translation to EPM management remains entirely speculative.
6.2 Protein and Branched-Chain Amino Acid Support
Nutritional support for muscle preservation during neurological illness represents an understudied area in equine medicine. Research in human stroke patients demonstrates that targeted nutritional intervention, particularly adequate protein and branched-chain amino acid (BCAA) supplementation, helps preserve muscle mass, enhance functional recovery, and optimize rehabilitation outcomes [25]. Dietary approaches including adequate protein intake significantly support neurological recovery by maintaining metabolic stability.
Studies in brain tumor patients showed that protein supplements had positive effects promoting muscle strength and physical performance, with significant increases in pinch grasp power and extended 6-minute walk test distances [26]. In malnourished patients, protein supplementation showed greater augmentation of muscle mass compared to those with adequate baseline nutrition. These findings suggest potential benefits for EPM patients experiencing neurogenic muscle atrophy, though direct evidence is absent.
6.3 Rehabilitation Feeding Considerations
Post-illness nutritional recovery has received some attention in equine colic research, which may inform EPM management. Studies demonstrated that horses with shorter post-surgical recovery consumed higher percentages of dry matter as forages in the first 24 hours and reached minimum dry matter intake more quickly [27]. Time to first feeding showed positive association with recovery length, supporting early nutritional intervention when clinically appropriate.
The integration of nutritional support with rehabilitation represents standard practice in human neurological care but lacks formal study in horses. For patients with severe neurological impairment, comprehensive care may require enteral feeding support, specialized diets, and careful attention to hydration status, though specific protocols for EPM patients have not been developed or validated.
---
7. Bias Assessment and Methodological Limitations
7.1 Comprehensive Bias Risk Evaluation
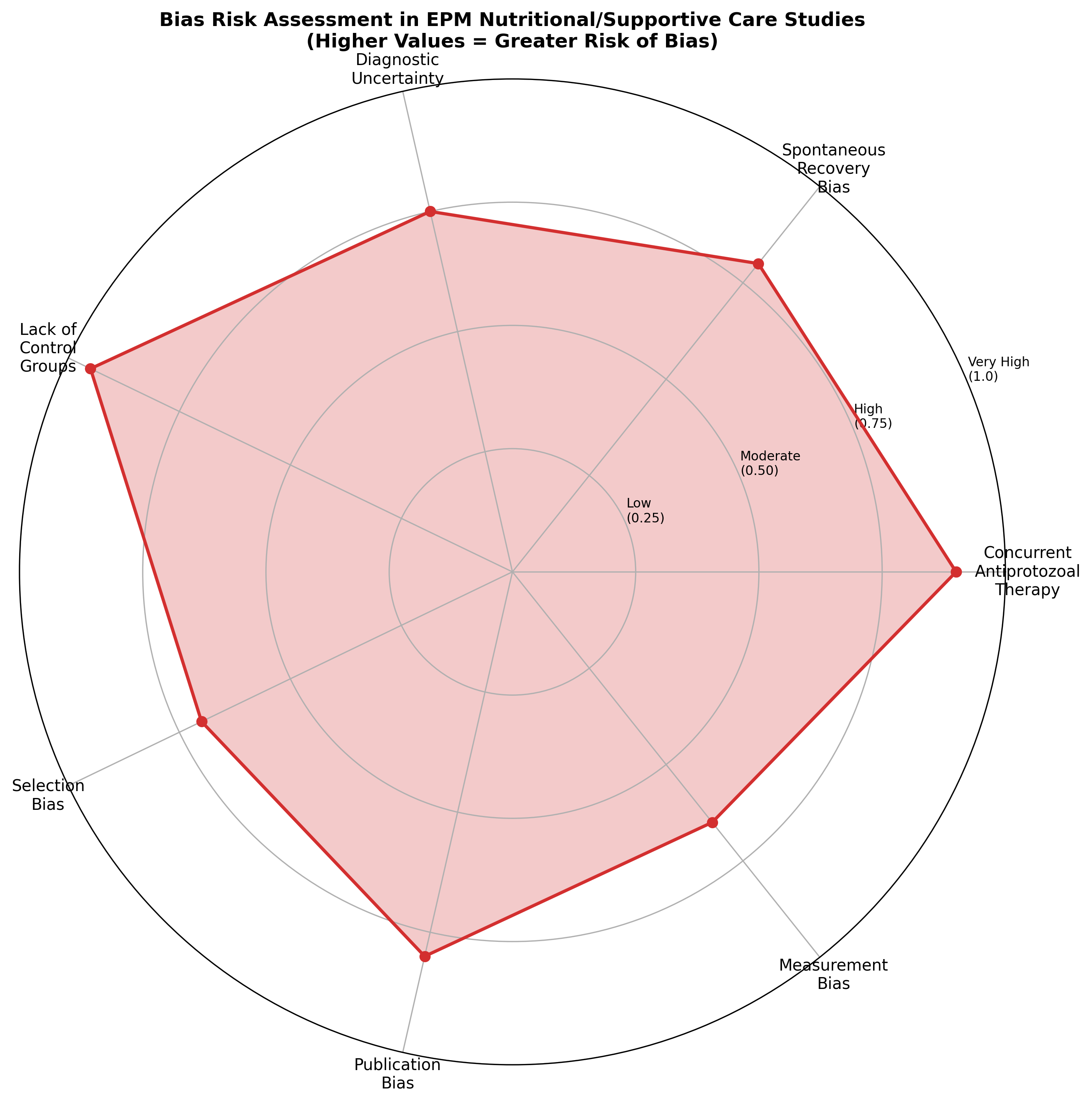
The radar chart illustrates the substantial bias risks inherent in evaluating nutritional interventions for EPM. The most critical sources of bias identified include:
Concurrent Antiprotozoal Therapy: Nearly all horses receiving nutritional supplements during EPM treatment also receive approved antiprotozoal medications. In the absence of placebo-controlled designs specifically examining supplements, it is impossible to attribute clinical improvement to nutritional intervention rather than primary therapy. Epizootic studies have demonstrated that treatment duration for EPM ranges from 45 to 211 days, with adverse effects including transient fever, anorexia, acute worsening of ataxia, mild anemia, and abortions reported with antiprotozoal therapy alone [28].
Spontaneous Recovery Potential: EPM presents a unique challenge for efficacy assessment because some degree of spontaneous recovery or stabilization occurs in many cases regardless of treatment. Treatment with approved antiprotozoal agents frequently leads to improvement, but complete resolution is uncommon, making determination of supplement contribution impossible without rigorous controlled designs [2].
Diagnostic Uncertainty: Definitive EPM diagnosis remains challenging, with paired serum and cerebrospinal fluid analysis being most reliable while PCR lacks sensitivity [2]. Empirical therapy is often pursued when owners decline testing, though clinical improvement alone cannot confirm the diagnosis. This diagnostic uncertainty means that some horses attributed as treatment successes may not have had EPM, confounding efficacy assessment.
Lack of Control Groups: The absence of controlled trials for nutritional interventions represents the fundamental limitation of current evidence. Without appropriate control groups receiving standard treatment without supplementation, efficacy cannot be established.
7.2 Quality of Extrapolated Evidence
Evidence extrapolated from other conditions carries inherent limitations:
| Source Condition | Key Limitations |
|---|---|
| Equine EDM/EMND | Different pathophysiology (degenerative vs. infectious) |
| Rodent SCI models | Species differences, acute injury vs. chronic infection |
| Human neurological diseases | Metabolic differences, different treatment contexts |
| In vitro/mechanistic studies | May not translate to clinical efficacy |
7.3 Publication and Reporting Bias
Studies demonstrating negative or null results for nutritional interventions are likely underrepresented in the literature due to publication bias. Additionally, case reports and anecdotal success stories are more likely to be shared than treatment failures, creating an artificially positive impression of supplement efficacy. The equine supplement industry's commercial interests may further bias available information toward favorable interpretations.
---
8. Credibility Ranking and Evidence Synthesis
8.1 Credibility Assessment by Intervention
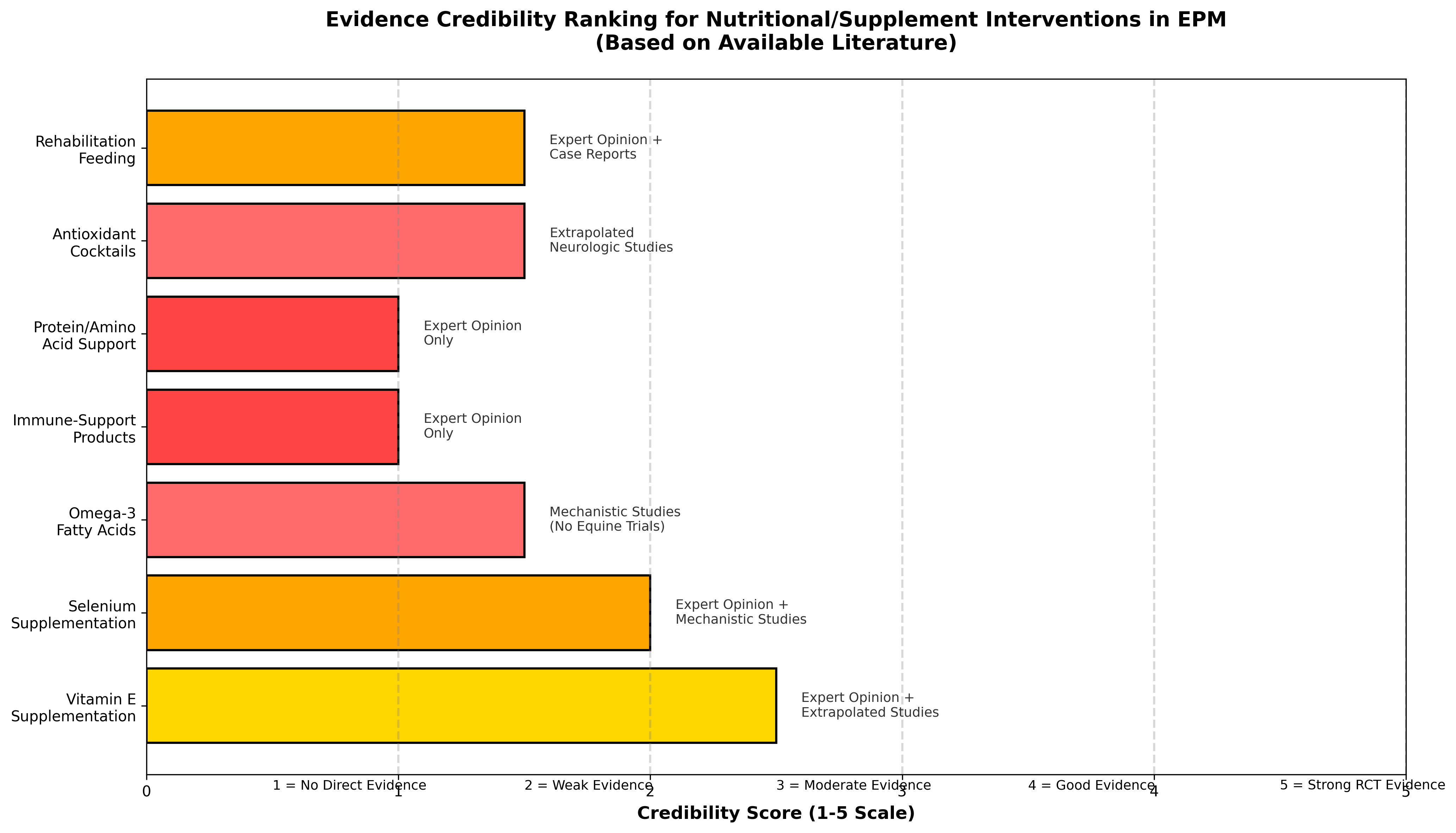
The credibility ranking synthesizes available evidence quality, mechanistic plausibility, and applicability to EPM management. Key findings include:
Vitamin E Supplementation (Score: 2.5/5): Represents the most credible intervention based on established deficiency-disease relationships in related equine conditions (EDM, EMND), well-characterized mechanistic pathways, defined dosing protocols, and reasonable safety profile. However, no direct EPM evidence exists, and the pathophysiology of infectious neuroinflammation differs from degenerative conditions where vitamin E has shown benefit.
Selenium Supplementation (Score: 2.0/5): Supported by mechanistic evidence for antioxidant enzyme function and case reports of deficiency-related neurological signs in horses. Combination with vitamin E may provide synergistic benefits based on laboratory animal research, but direct EPM evidence is absent.
Omega-3 Fatty Acids (Score: 1.5/5): Strong mechanistic rationale exists based on extensive research in neuroinflammation models, but no equine neurological disease trials have been conducted. Effects may vary based on formulation, dosage, and EPA:DHA ratio, none of which have been optimized for equine applications.
Antioxidant Cocktails (Score: 1.5/5): Multiple antioxidant combinations show promise in laboratory models but lack any equine-specific evidence. Risk of supplement interactions and appropriate dosing remain undefined.
Rehabilitation Feeding (Score: 1.5/5): Principles of nutritional support during recovery are well-established in human and companion animal medicine but have not been specifically studied in EPM patients.
Protein/Amino Acid Support (Score: 1.0/5): Theoretical benefit for maintaining muscle mass during neurological illness is reasonable but entirely unsupported by equine evidence.
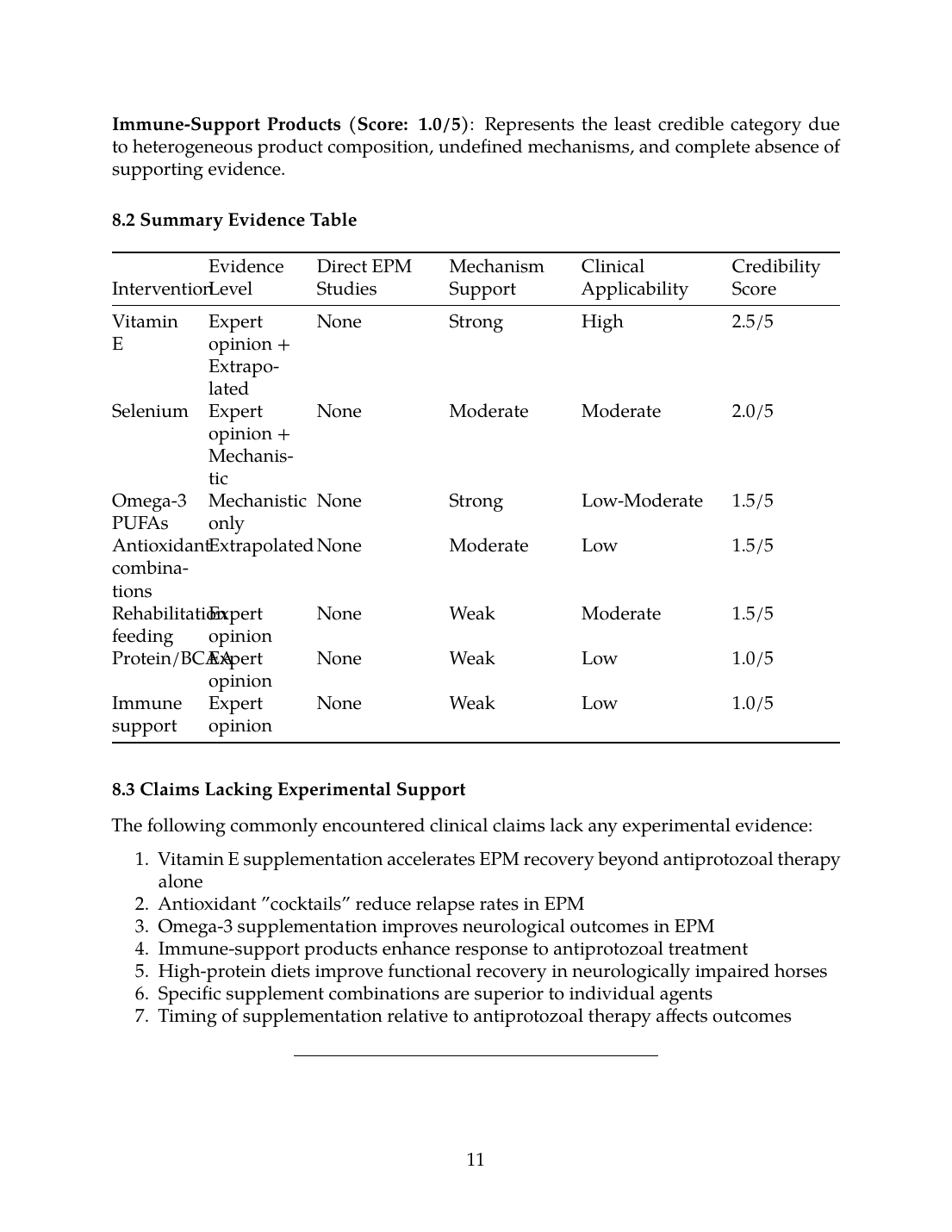
Immune-Support Products (Score: 1.0/5): Represents the least credible category due to heterogeneous product composition, undefined mechanisms, and complete absence of supporting evidence.
8.2 Summary Evidence Table
| Intervention | Evidence Level | Direct EPM Studies | Mechanism Support | Clinical Applicability | Credibility Score |
|---|---|---|---|---|---|
| Vitamin E | Expert opinion + Extrapolated | None | Strong | High | 2.5/5 |
| Selenium | Expert opinion + Mechanistic | None | Moderate | Moderate | 2.0/5 |
| Omega-3 PUFAs | Mechanistic only | None | Strong | Low-Moderate | 1.5/5 |
| Antioxidant combinations | Extrapolated | None | Moderate | Low | 1.5/5 |
| Rehabilitation feeding | Expert opinion | None | Weak | Moderate | 1.5/5 |
| Protein/BCAA | Expert opinion | None | Weak | Low | 1.0/5 |
| Immune support | Expert opinion | None | Weak | Low | 1.0/5 |
8.3 Claims Lacking Experimental Support
The following commonly encountered clinical claims lack any experimental evidence:
- Vitamin E supplementation accelerates EPM recovery beyond antiprotozoal therapy alone
- Antioxidant "cocktails" reduce relapse rates in EPM
- Omega-3 supplementation improves neurological outcomes in EPM
- Immune-support products enhance response to antiprotozoal treatment
- High-protein diets improve functional recovery in neurologically impaired horses
- Specific supplement combinations are superior to individual agents
- Timing of supplementation relative to antiprotozoal therapy affects outcomes
---
9. Conclusions and Recommendations
9.1 Summary of Findings
This methodological systematic review reveals a profound evidence gap regarding nutritional and supplement-based adjuncts in EPM management. No randomized controlled trials, systematic reviews, or prospective cohort studies specifically examining these interventions in EPM were identified. Available evidence consists primarily of expert opinion, mechanistic studies in other species, and extrapolation from related equine neurological conditions with different pathophysiology.
Vitamin E supplementation maintains the strongest theoretical basis given its established role in equine degenerative neurological diseases and well-characterized safety profile when appropriately monitored [4]. Omega-3 fatty acids demonstrate compelling neuroprotective mechanisms in laboratory models of neuroinflammation [29], but translation to equine clinical application remains speculative. All other interventions lack meaningful supporting evidence.
9.2 Implications for Clinical Practice
Given the current evidence landscape, clinicians should:
- Prioritize approved antiprotozoal therapy as the foundation of EPM treatment
- Consider vitamin E supplementation (after serum level assessment) as a low-risk adjunct, particularly in horses with marginal or deficient baseline status
- Avoid claims of efficacy for nutritional interventions that cannot be substantiated
- Counsel owners regarding the distinction between mechanistic plausibility and clinical evidence
- Monitor for adverse effects even with "natural" supplements, as toxicity is possible with excessive supplementation
9.3 Research Priorities
Future research should address the following critical gaps:
- Randomized controlled trials comparing antiprotozoal therapy with and without specific nutritional adjuncts
- Standardized neurological outcome measures to enable meaningful comparison across studies
- Investigation of supplement timing and duration relative to disease course and antiprotozoal treatment
- Pharmacokinetic studies establishing appropriate dosing for omega-3 PUFAs and other supplements in horses
- Long-term follow-up assessing relapse rates and functional outcomes with standardized protocols
Until rigorous clinical trials are conducted, the evidence supporting nutritional and supplement-based adjuncts in EPM management remains insufficient to justify definitive recommendations. Practitioners and horse owners should maintain realistic expectations and recognize that currently available supplements represent unproven interventions regardless of marketing claims.
References
References
Numbered in order of first citation, IEEE style. Each entry links to the original source via DOI or publisher URL.
- R. MacKay and D. K. Howe, “Equine Protozoal Myeloencephalitis.,” The Veterinary clinics of North America. Equine practice, Jul. 2022. doi: 10.1016/j.cveq.2022.05.003. View source
- S. Austin and S. Gray, “Equine protozoal myeloencephalitis—Presentation and progression,” Equine Veterinary Education, Oct. 2025. doi: 10.1111/eve.70016. View source
- C. Castillo Rodríguez, F. W. Menge, and J. Cerón, “Oxidative Stress in Veterinary Medicine,” Veterinary Medicine International, Aug. 2011. doi: 10.4061/2011/812086. View source
- A. E. Young and C. J. Finno, “Current insights into equine neuroaxonal dystrophy/equine degenerative myeloencephalopathy.,” The Veterinary Journal, May 2024. doi: 10.1016/j.tvjl.2024.106129. View source
- H. Mohammed, T. Divers, B. Summers, and A. de Lahunta, “Vitamin E deficiency and risk of equine motor neuron disease,” Acta Veterinaria Scandinavica, Jul. 2007. doi: 10.1186/1751-0147-49-17. View source
- B. C. D. C. Germano, L. C. C. D. Morais, F. I. Neta, A. Fernandes, F. I. Pinheiro, A. C. M. D. Rêgo, I. A. Filho, E. P. D. Azevedo, J. R. L. D. P. Cavalcanti, F. Guzen, and R. Cobucci, “Vitamin E and Its Molecular Effects in Experimental Models of Neurodegenerative Diseases,” International Journal of Molecular Sciences, Jul. 2023. doi: 10.3390/ijms241311191. View source
- M. M. Miller and C. Collatos, “Equine degenerative myeloencephalopathy.,” The Veterinary clinics of North America. Equine practice, Apr. 1997. doi: 10.1016/S0749-0739(17)30254-7. View source
- B. Ll, B. Hultgren, A. Craig, L. H. Appell, L. Ed, D. Mattson, and D. Duffield, “Clinical, viral, and genetic evaluation of equine degenerative myeloencephalopathy in a family of Appaloosas.,” Journal of the American Veterinary Medical Association, Mar. 1991. doi: 10.2460/javma.1991.198.06.1005. View source
- C. Ballard, E. Jensen-Vargas, K. Lassell, and S. Dobbins, “PSVII-13 The Evaluation of Natural Vs Synthetic Vitamin E Supplementation for the Management of Alpha-Tocopherol Serum Concentrations in Morgan Horses,” Journal of Animal Science, Nov. 2023. doi: 10.1093/jas/skad281.588. View source
- C. Ballard, E. Jensen-Vargas, K. Lassell, and S. Dobbins, “PSVII-12 The Effect of Two Supplemental Vitamin E Dosing Strategies in Morgan Horses with Marginal/Deficient Concentrations of Serum Alpha-Tocopherol,” Journal of Animal Science, Nov. 2023. doi: 10.1093/jas/skad281.587. View source
- M. Al‐zharani, E. Almuqri, M. Mubarak, H. Rudayni, N. H. Aljarba, K. Yaseen, S. Alkahtani, F. A. Nasr, A. Al-Doaiss, and M. S. Al-eissa, “Comparison between the Antioxidant Properties of Quercetin and Combined Vitamin E and Selenium to Ameliorate the Oxidative Stress Induced by Cadmium Toxicity,” Current Research in Nutrition and Food Science Journal, Dec. 2024. doi: 10.12944/crnfsj.12.3.9. View source
- T. Danese and E. Valle, “Dietary intervention in selenium imbalances: a case series,” UK-Vet Equine, Mar. 2026. doi: 10.12968/ukve.2025.0006. View source
- J. De Toledo, T. F. C. Fraga-Silva, P. Borim, L. R. C. D. Oliveira, E. D. S. Oliveira, L. L. Périco, C. Hiruma-Lima, A. A. D. de Souza, C. A. D. de Oliveira, P. Padilha, M. Pinatto-Botelho, A. D. dos Santos, A. Sartori, and S. Zorzella-Pezavento, “Organic Selenium Reaches the Central Nervous System and Downmodulates Local Inflammation: A Complementary Therapy for Multiple Sclerosis?,” Frontiers in Immunology, Oct. 2020. doi: 10.3389/fimmu.2020.571844. View source
- E. García, F. Hernández-Ayvar, R. Rodríguez-Barrera, A. Flores-Romero, C. Borlongan, and A. Ibarra, “Supplementation With Vitamin E, Zinc, Selenium, and Copper Re-Establishes T-Cell Function and Improves Motor Recovery in a Rat Model of Spinal Cord Injury,” Cell Transplantation, Jan. 2022. doi: 10.1177/09636897221109884. View source
- M. Gueroui and Z. Kechrid, “Evaluation of Some Biochemical Parameters and Brain Oxidative Stress in Experimental Rats Exposed Chronically to Silver Nitrate and the Protective Role of Vitamin E and Selenium,” Toxicological Research, Oct. 2016. doi: 10.5487/TR.2016.32.4.301. View source
- J. Adamiuk, K. Kopeć, A. Bartoszek, A. Marut, M. Biskup, J. Misiuk, A. Skuba, K. Załuska, A. Świdniak, and S. Nykiel, “The Role of Omega-3 Fatty Acids in Cognitive Health: From Development to Aging and Neurodegenerative Protection,” Quality in Sport, May 2025. doi: 10.12775/qs.2025.41.58753. View source
- A. Zinkow, W. Grodzicki, M. Czerwińska, and K. Dziendzikowska, “Molecular Mechanisms Linking Omega-3 Fatty Acids and the Gut–Brain Axis,” Molecules, Dec. 2024. doi: 10.3390/molecules30010071. View source
- R. Z. Alshahawy, S. M. Safwat, S. S. El-Kholy, S. F. El-Basiony, S. El-desouky, and S. M. Helmi, “Vitamin D and omega-3 fatty acids attenuate MSG-induced neurodegeneration by modulating tau pathology, neuroinflammation, and VDR expression in rats,” Scientific Reports, Jun. 2025. doi: 10.1038/s41598-025-06109-3. View source
- L. He, J. Ye, X. Zhuang, J. Shi, and W. Wu, “Omega-3 polyunsaturated fatty acids alleviate endoplasmic reticulum stress-induced neuroinflammation by protecting against traumatic spinal cord injury through the histone deacetylase 3/ peroxisome proliferator-activated receptor-γ coactivator pathway.,” Journal of Neuropathology and Experimental Neurology, Aug. 2024. doi: 10.1093/jnen/nlae094. View source
- W. A. Alammar, F. H. Albeesh, L. M. Ibrahim, Y. Y. Algindan, L. Yamani, and R. Khattab, “Effect of omega-3 fatty acids and fish oil supplementation on multiple sclerosis: a systematic review,” Nutritional neuroscience, Aug. 2019. doi: 10.1080/1028415X.2019.1659560. View source
- E. Siegert, F. Paul, M. Rothe, and K. Weylandt, “The effect of omega-3 fatty acids on central nervous system remyelination in fat-1 mice,” BMC Neuroscience, Jan. 2017. doi: 10.1186/s12868-016-0312-5. View source
- E. Valero-Hernández, J. L. Tremoleda, and A. T. Michael-Titus, “Omega-3 Fatty Acids and Traumatic Injury in the Adult and Immature Brain,” Nutrients, Nov. 2024. doi: 10.3390/nu16234175. View source
- H. Saeed, H. Osama, M. A. Abdelrahman, Y. M. Madney, H. S. Harb, M. Abdelrahim, and F. Ali, “Vitamins and other immune-supportive elements as cofactors for passing the COVID-19 pandemic,” Beni-Suef University Journal of Basic and Applied Sciences, Oct. 2021. doi: 10.1186/s43088-021-00163-2. View source
- S. A. Pamungkas, C. Prahasanti, S. Wardah, D. L. Kusriawati, M. J. Anggakusuma, M. G. Wijaya, M. Suhartono, W. Riawan, and Z. S. Ulhaq, “The effect of Lactobacillus rhamnosus administration against Porphyromonas gingivalis in vivo,” Journal of Oral Biology and Craniofacial Research, Nov. 2025. doi: 10.1016/j.jobcr.2025.10.025. View source
- A. C. R. Mauluddy and D. Sunardi, “The role of nutritional management in preventing stroke and improving clinical outcomes,” World Nutrition Journal, Aug. 2025. doi: 10.25220/wnj.v09.i1.0002. View source
- K. Cho, E. Han, M. Jung, C. M. Kang, J. C. Shin, and S. H. Im, “Effects of protein-enriched nutritional support on skeletal muscle mass and rehabilitative outcomes in brain tumor patients: a randomized controlled trial,” Scientific Reports, Jun. 2024. doi: 10.1038/s41598-024-63551-5. View source
- E. Valle, G. Giusto, L. Penazzi, M. Giribaldi, D. Bergero, M. J. Fradinho, L. P. Lamas, and M. Gandini, “Preliminary results on the association with feeding and recovery length in equine colic patients after laparotomy.,” Journal of animal physiology and animal nutrition, Apr. 2019. doi: 10.1111/jpn.13102. View source
- C. Fenger, D. Granstrom, J. Langemeier, and S. Stamper, “Epizootic of equine protozoal myeloencephalitis on a farm.,” Journal of the American Veterinary Medical Association, Apr. 1997. doi: 10.2460/javma.1997.210.07.923. View source
- Y. Chen, R. Touboul, Y. Chen, and C. L. Chang, “Strategic delivery of omega-3 fatty acids for modulating inflammatory neurodegenerative diseases,” Frontiers in Aging Neuroscience, Mar. 2025. doi: 10.3389/fnagi.2025.1535094. View source
This review is one of 34 in the Foxwatch research corpus. Methodology: PRISMA 2020-style systematic literature review against PubMed, CAB Abstracts, Scopus, Web of Science, and equine veterinary conference proceedings.