Mechanistic review of nutrition, inflammation, and neurologic injury in EPM
Sarcocystis neurona invades the central nervous system, and the secondary inflammation does most of the lasting damage. This review maps which nutrients (vitamin E, selenium, omega-3, protein adequacy) act on which CNS injury mechanisms, and which "EPM nutrition support" claims are mechanistically supported versus speculative. Pairs with reviews 11 (acute) and 22 (rehab).
Research thesis
This mechanistic review argues that nutrition meaningfully shapes EPM clinical course through plausible biological pathways while never functioning as primary treatment, not only because Sarcocystis neurona and Neospora hughesi establish CNS infection and drive secondary inflammation, oxidative damage, and blood-brain-barrier disruption that respond biologically to vitamin E, selenium, omega-3 fatty acids, and adequate protein, but also because corticosteroid use, antiprotozoal therapy, and prolonged stall rest each impose nutrient demands that affect recovery; by mapping nutritional factors to specific CNS injury mechanisms across direct EPM evidence and extrapolation from comparable neurologic disease, this review builds an evidence-graded mechanism table and clarifies where nutritional support is mechanistically supported and where it remains speculative.
Evidence visualizations & full pages
Original review pages (PDF render)
Download
Full review text
Nutrition, inflammation, and neurologic injury in EPM
Now I have substantial information. Let me create a comprehensive outline and begin writing the literature review. Based on my search results, I'll organize the findings into the requested structure.
Mechanistic Literature Review: Nutrition, Inflammation, and Neurologic Injury in Equine Protozoal Myeloencephalitis
OUTLINE
I. Introduction & EPM Pathogenesis Overview
- Sarcocystis neurona vs. Neospora hughesi biology
- CNS infection and immune response mechanisms
- Direct EPM evidence vs. extrapolated mechanisms
II. Pathophysiology of Secondary CNS Damage in EPM
- Neuroinflammation cascades (microglia, astrocytes, cytokines)
- Oxidative stress mechanisms
- Blood-brain/blood-spinal cord barrier dysfunction
- Secondary injury mechanisms
III. Nutritional Factors and Antioxidant Defense
- Vitamin E (α-tocopherol) - antioxidant and neuroprotective roles
- Selenium - selenoprotein synthesis and glutathione peroxidase
- Omega-3 fatty acids - membrane integrity and neuroinflammation
- Interaction with antiprotozoal treatment effects
IV. Protein Adequacy and Muscle Atrophy Recovery
- Disuse atrophy mechanisms in neurologic disease
- Protein synthesis signaling (mTOR, PI3K/Akt pathways)
- Rehabilitation demands during EPM recovery
- Body condition implications
V. CNS Inflammation Modulation Through Nutrition
- BBB/BSCB integrity preservation
- Microglia polarization and cytokine modulation
- Role of micronutrients in limiting immune dysregulation
- Nutritional support during antiprotozoal and corticosteroid use
VI. Clinical Integration & Mechanistic Uncertainty
- Mechanisms table (nutrients, mechanisms, evidence levels, uncertainty)
- Distinction between EPM-specific evidence and neurologic disease extrapolation
- Rehabilitation synergy with nutrition
- Future research directions
---
FINAL COMPREHENSIVE LITERATURE REVIEW
I. Introduction: Equine Protozoal Myeloencephalitis and the Rationale for Nutritional Inquiry
Equine protozoal myeloencephalitis (EPM) remains one of the most frequently diagnosed neurological disorders of horses in North America, caused primarily by the apicomplexan parasite Sarcocystis neurona, with less common cases due to Neospora hughesi [1]. While seroprevalence of S. neurona in horses is remarkably high across endemic regions, clinical manifestation of EPM occurs in less than 1% of infected horses, suggesting that individual factors—including host immunity—critically influence disease development [2]. The recognized treatment arsenal includes FDA-approved anticoccidial drugs such as sulfadiazine/pyrimethamine (ReBalance), diclazuril (Protazil), and ponazuril (Marquis), yet prognosis remains guarded, with incomplete resolution common and relapses documented after apparent clinical improvement [3],[4].
Beyond antimicrobial therapy, limited evidence exists regarding how nutritional status might modulate recovery trajectory. This mechanistic review synthesizes pathophysiological understanding of EPM and neuroinflammatory secondary injury to identify plausible nutritional interventions that could support—but not replace—antiprotozoal treatment. The distinction between direct EPM evidence and mechanistic inference from spinal cord injury (SCI) and other neurologic models is maintained throughout, as equine-specific nutritional studies in EPM are essentially absent from the literature. The review addresses how nutrition might attenuate neuroinflammation, reduce oxidative damage, preserve blood-spinal cord barrier (BSCB) integrity, support muscle recovery during neurologic disability, and optimize tolerance of concurrent treatments (corticosteroids, antiprotozoals).
---
II. Pathophysiology of S. neurona and N. hughesi Infection: Implications for Secondary Damage
A. Parasite Invasion and Tissue Colonization
Sarcocystis neurona is an obligate intracellular apicomplexan that relies on specialized secretory organelles—rhoptries and micronemes—to facilitate host cell invasion and intracellular survival [5]. Once established in the CNS, the parasite's life cycle stages (merozoites and schizonts) persist within neural tissue, triggering complex immunopathological cascades. Current evidence indicates that merely detecting S. neurona DNA or antibodies in cerebrospinal fluid does not discriminate between active infection and parasitic persistence following antiprotozoal treatment [6]. In animal models, diclazuril administration suppressed S. neurona replication but failed to completely eliminate the parasite, allowing recrudescence of neurologic signs within weeks of treatment cessation—a phenomenon attributed to sequestration of the organism from immune surveillance [7]. This distinction between parasite elimination and clinical resolution underscores that nutritional interventions supporting immune function and CNS repair may operate through mechanisms independent of direct antiparasitic activity.
B. Immune Response Skewing and Th1/Th2 Balance
Recent immunological analysis of EPM-affected horses has revealed that disease is associated with a Th1-biased, cell-mediated immune response, distinguished by elevated S. neurona-specific IgG1/2 and IgG4/7 subisotypes in diseased versus seropositive normal horses [2]. This skewing toward cell-mediated immunity contrasts with the protective humoral responses in asymptomatic infected horses, suggesting aberrant immune regulation may contribute to EPM pathogenesis. The mechanisms governing this dysbalanced response—including genetic predisposition, parasite strain virulence, prior stressful events, and nutritional status—remain largely undetermined [3]. However, extrapolation from other CNS infections and neuroimmunological models suggests that balanced micronutrient status (particularly antioxidants and minerals supporting regulatory T cell differentiation) may influence the Th1/Th2 balance and limit pathogenic inflammatory cascades.
---
III. Secondary Neuroinflammation and Oxidative Stress: Mechanisms and Nutritional Targets
A. Neuroinflammatory Cascade Following CNS Insult
The spinal cord damage from S. neurona migration triggers a prototypical secondary injury cascade analogous to traumatic SCI. Within hours to days of primary parasitic injury, activated microglia and astrocytes release pro-inflammatory cytokines including tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6), and interleukin-1β (IL-1β), which recruit peripheral immune cells across the compromised BSCB [8]. This neuroinflammatory phase is self-propagating: immune cell infiltration exacerbates BSCB disruption through matrix metalloproteinase (MMP-9 and MMP-2) upregulation, allowing further leukocyte infiltration and toxic mediator accumulation [9]. M1 pro-inflammatory microglia predominate early, but persistent inflammation and aberrant resolution kinetics favor chronic microglial activation and glial scar formation, ultimately limiting axonal regeneration [10].
Nutritional modulation of this cascade is mechanistically plausible: selenium-dependent glutathione peroxidase (GPx) and other selenoproteins suppress microglial-derived oxidative stress, while vitamin E directly inhibits lipid peroxidation in microglial membranes [11]. Omega-3 polyunsaturated fatty acids (PUFAs) promote microglial polarization toward anti-inflammatory phenotypes and reduce production of TNF-α and IL-1β [8]. Adequate protein intake sustains synthesis of anti-inflammatory proteins and growth factors critical for microglial resolution and neuronal support.
B. Oxidative Stress as a Converging Pathogenic Mechanism
Oxidative damage is central to secondary SCI and is undoubtedly relevant to EPM: spinal cord ischemia, immune cell infiltration, and parasitic migration all generate excessive reactive oxygen species (ROS) that overwhelm endogenous antioxidant defenses [12]. Mitochondrial ROS production, in turn, triggers NLRP3 inflammasome activation and pyroptosis, a caspase-1–dependent form of neuronal death [13]. Free zinc accumulation—another consequence of CNS injury—amplifies oxidative stress through mitochondrial dysfunction and inhibits BSCB tight junction recovery [14].
Vitamin E (α-tocopherol) has demonstrated neuroprotective efficacy against spinal cord ischemia/reperfusion injury in rats, improving motor and sensory recovery while reducing prostaglandin E2, malondialdehyde (MDA), and advanced oxidation protein products (AOPP) [15]. Selenium-dependent selenoprotein expression (particularly glutathione peroxidase and thioredoxin reductase) is rapidly downregulated following CNS injury; supplementation with selenomethionine (SeM) in animal models of SCI upregulates selenoproteins and scavenges ROS, reducing neuronal apoptosis and promoting mitochondrial function [11],[16]. In horses, deficiency of vitamin E and selenium has been associated with equine degenerative myeloencephalopathy (EMND), another progressive neurologic disorder characterized by motor neuron degeneration, implicating these micronutrients in neuronal antioxidant defense [17].
---
IV. Blood-Spinal Cord Barrier Integrity: Nutritional Support of Endothelial Function
A. BSCB Disruption in Secondary Injury
The BSCB, functionally analogous to the blood-brain barrier (BBB), is maintained by endothelial tight junction proteins (occludin, claudin-5, zonula occludens-1) and supported by pericyte coverage [18]. EPM-associated inflammation disrupts this barrier through MMP-dependent degradation of tight junction proteins, increased vascular permeability, fibrinogen extravasation, and hemorrhage [19]. BSCB breakdown permits infiltration of potentially harmful immune cells, complement components, and proteases while failing to prevent CNS edema and secondary neuronal death.
Pericyte-derived exosomes and their miR-210 cargo improve endothelial barrier function by inhibiting JAK1/STAT3 signaling, reducing lipid peroxidation, and improving mitochondrial function [20]. This mechanism highlights how nutritional support of mitochondrial health (through antioxidants and micronutrients supporting oxidative phosphorylation) indirectly preserves BSCB integrity.
B. Nutritional Modulators of BBB/BSCB Integrity
Several experimental and clinical studies support roles for specific nutrients in BSCB repair. Tight junction protein expression (ZO-1, occludin, claudin-5) is upregulated by anti-inflammatory signaling and by antioxidant activity that suppresses pro-inflammatory gene transcription. Vitamin C and vitamin E both support endothelial tight junction recovery through NF-κB inhibition [21]. Omega-3 PUFAs reduce inflammatory cytokines (TNF-α, IL-1β) that destabilize tight junctions; EPA and DHA shift immune cell phenotypes toward resolution-phase markers [22]. Collagen and structural matrix proteins that support endothelial-pericyte interactions require adequate protein intake and vitamins C and D for hydroxylation and cross-linking. Selenium supports glutathione peroxidase activity, which prevents oxidative damage to endothelial lipid membranes and preserves tight junction functionality [23].
---
V. Muscle Atrophy and Protein Metabolism in Neurologic Disease
A. Disuse-Induced Muscle Atrophy: Mechanisms and Timeline
EPM-affected horses experience rapid, multifactorial muscle loss resulting from neurologic dysfunction, forced immobilization (stall confinement during treatment), inflammatory cytokine effects, and reduced voluntary movement secondary to ataxia and weakness. Disuse muscle atrophy involves synchronized suppression of muscle protein synthesis and upregulation of proteolytic pathways—particularly the ubiquitin-proteasome system (UPS) and autophagy. Key transcription factors driving atrophy include FoxO (Forkhead box O) family members and TRIM63/MuRF1 (muscle ring finger 1), which ubiquitinate muscle structural proteins [24].
In humans and rodent models, just 2–7 days of immobilization decreases plantarflexor strength by 24% and induces fiber atrophy detectable histologically [25]. Protein synthesis rates decline acutely, particularly in the fasted state, while protein breakdown accelerates for days after immobilization onset. Critically, this early phase of disuse atrophy is potentially reversible with timely nutritional and rehabilitative intervention [26].
B. Protein Adequacy and Amino Acid Signaling in Recovery
Dietary protein supplementation—particularly when combined with resistance or rehabilitation exercise—activates mammalian target of rapamycin (mTOR) signaling and phosphorylation of ribosomal S6 kinase, upregulating muscle protein synthesis [27]. In older humans undergoing immobilization and rehabilitation, whey protein and high-protein diets (but not free leucine alone) were necessary to prevent delayed muscle atrophy in non-immobilized limbs, suggesting complex amino acid profiles are required beyond single amino acid supplementation [28]. The threshold for protein adequacy in preventing disuse atrophy appears to be 1.2–1.6 g/kg body weight daily, substantially higher than the maintenance requirement [29].
In horses recovering from EPM, adequate protein intake (through quality forage, concentrate supplements, or specialized feed formulations) is mechanistically justified to support: (1) residual muscle protein synthesis despite neurologic dysfunction, (2) synthesis of inflammatory mediators and growth factors (such as brain-derived neurotrophic factor, BDNF) supporting neuronal regeneration, and (3) synthesis of antioxidant enzymes and immune proteins [30]. The synergy between early mild rehabilitation (lunging, hand-walking, aquatic therapy) and high-quality protein intake is supported by evidence that exercise primes mTOR signaling, sensitizing muscle to the anabolic effects of protein [26].
C. Body Condition and Metabolic Resiliency
Equine body condition score (BCS) prior to EPM onset may influence nutritional reserves available to support recovery. Poor body condition at EPM diagnosis (as has been identified as a risk factor for S. neurona seropositivity [31]) reflects limited nutrient stores (fat, protein, mineral reserves) to sustain the increased demands of immune response, antiprotozoal drug metabolism, and rehabilitation. Pre-illness optimization of body condition—through adequate caloric and nutrient-dense forage—provides a "nutritional cushion" that may limit secondary complications and accelerate recovery. Conversely, overnutrition and obesity impair glucose homeostasis and promote low-grade systemic inflammation [32], potentially exacerbating the CNS inflammatory environment.
---
VI. Corticosteroid Use in EPM: Secondary Effects and Nutritional Mitigation
While not always employed in routine EPM treatment, corticosteroids are occasionally used empirically to reduce spinal cord edema and neuroinflammation, particularly in severe presentations. Corticosteroid administration induces multiple nutritional consequences relevant to recovery: (1) increased protein catabolism and muscle loss, (2) hyperglycemia and impaired glucose utilization, (3) suppression of immunoglobulin synthesis and cellular immunity, (4) reduced intestinal calcium absorption, and (5) increased free radical generation [33].
Nutritional strategies to mitigate steroid-induced catabolic effects include: (a) increased protein intake (exceeding the standard 1.2 g/kg target), (b) antioxidant supplementation to counter steroid-induced oxidative stress, (c) adequate vitamin D and calcium to preserve bone and muscle mass, and (d) micronutrients supporting immune reconstitution post-treatment taper (zinc, selenium, vitamin C) [34]. A specific example is that corticosteroids impair selenoprotein expression and glutathione peroxidase activity; selenium supplementation can partially restore these antioxidant defenses [35]. The duration and dose of corticosteroid use in individual EPM cases determine the magnitude of these secondary nutritional demands.
---
VII. Antiprotozoal Drugs: Interactions with Nutrition and Side Effects
Sulfadiazine, pyrimethamine, diclazuril, and ponazuril are folate metabolism inhibitors or inhibitors of parasite metabolism; all carry potential for drug-nutrient interactions and side effects affecting nutritional status [1]. Sulfonamides (sulfadiazine) inhibit bacterial folate synthesis and can impair intestinal microbial production of B vitamins, necessitating dietary B vitamin supplementation or microbial reconstitution strategies. Pyrimethamine directly inhibits dihydrofolate reductase, reducing active folate cofactor availability and potentially impairing one-carbon metabolism critical for nucleotide synthesis, methylation reactions, and neuronal functions [36].
Diclazuril and ponazuril disrupt mitochondrial electron transport in parasites; an unquantified but theoretically relevant risk is impaired mitochondrial function in host tissues, particularly in tissues with high metabolic demands (CNS, muscle). Selenium-dependent mitochondrial selenoproteins (glutathione peroxidase, thioredoxin reductase) and coenzyme Q10-dependent electron transport chains may partially offset parasite-imposed metabolic stress, supporting a mechanistic rationale for antioxidant supplementation during treatment [37].
Hepatic metabolism of antiprotozoal drugs imposes detoxification demands; adequate protein intake and antioxidant status support phase I (cytochrome P450) and phase II (glutathione conjugation, sulfation) detoxification [8]. Antiprotozoal drugs may also cause gastrointestinal upset, reducing voluntary feed intake and nutrient absorption—a common observation in treated EPM horses that directly compromises nutrition during the period of maximal treatment intensity.
---
VIII. Rehabilitation Demands and Nutritional Synergies
A. Exercise, Neuroplasticity, and Protein Synthesis
Rehabilitation following EPM (controlled in-hand exercise, lunging at walk/trot, aquatic therapy, or hand-grazing) reintroduces mechanical loading and neuromotor activation critical for motor recovery. Exercise stimulates muscle protein synthesis through multiple mechanisms: mechanical tension activates integrin signaling, calcium influx, and mTOR; metabolic stress increases AMP-activated protein kinase (AMPK) and mitochondrial biogenesis; and muscle damage signals trigger satellite cell activation and myogenic differentiation [26].
Nutritional support is synergistic: protein intake directly supplies amino acids for synthesis, while micronutrients (vitamin C, zinc, selenium, B vitamins) support the enzymatic machinery of protein synthesis, mitochondrial biogenesis, and collagen cross-linking necessary for connective tissue repair [37],[36]. In aged humans undergoing rehabilitation after immobilization, combined resistance exercise and protein supplementation achieved far superior muscle recovery than either intervention alone [30].
B. CNS Neuroplasticity and Nutritional Support
Rehabilitation in EPM drives neuroplasticity—reorganization of spinal and supraspinal circuits to compensate for parasitic damage and optimize residual motor control. Neuroplasticity is supported by growth factor signaling (BDNF, nerve growth factor), synaptic remodeling, dendritic spine stabilization, and oligodendrocyte maturation. Vitamins (particularly B vitamins, vitamins C, D, and E), minerals (zinc, magnesium, iron, copper), and amino acids (particularly those supporting neurotransmitter synthesis: tryptophan, tyrosine, phenylalanine, histidine) are obligatory cofactors in these processes [23],[38].
Omega-3 PUFAs (EPA and DHA) are structural components of neuronal membranes and reduce CNS inflammation; supplementation in stroke models enhances neuroplastic recovery [22]. One-carbon metabolism nutrients (folate, B12, choline) support methylation reactions required for BDNF promoter activation and synaptic gene expression, mechanistically linking B vitamin status to neuroplasticity during EPM rehabilitation [36].
---
IX. Forage Quality and Macronutrient Sufficiency in EPM-Affected Horses
While equine-specific nutrition trials in EPM are absent, principles from equine nutrition physiology and comparative neurologic disease models suggest forage quality significantly impacts recovery. High-quality (early-bloom, leafy) hay or pasture delivers adequate crude protein (14–18%), essential amino acids, energy for rehabilitation, and micronutrients (selenium, vitamins E and A) compared to mature, over-cured forage. Mature hay is often deficient in vitamin E (losses during storage) and selenium (if grown in selenium-deficient soils).
In EPM recovery, forage should be supplemented with concentrates providing: (1) additional crude protein (targeted 1.2–1.6 g/kg body weight daily), (2) vitamin E (1,000–2,000 IU daily), (3) selenium (if not naturally present in local forage), and (4) omega-3 rich supplements (ground flaxseed, chia, or fish oil). Commercial performance or rehabilitation feeds often include these nutrients; individually formulated supplements allow personalized micronutrient dosing based on baseline forage mineral composition.
---
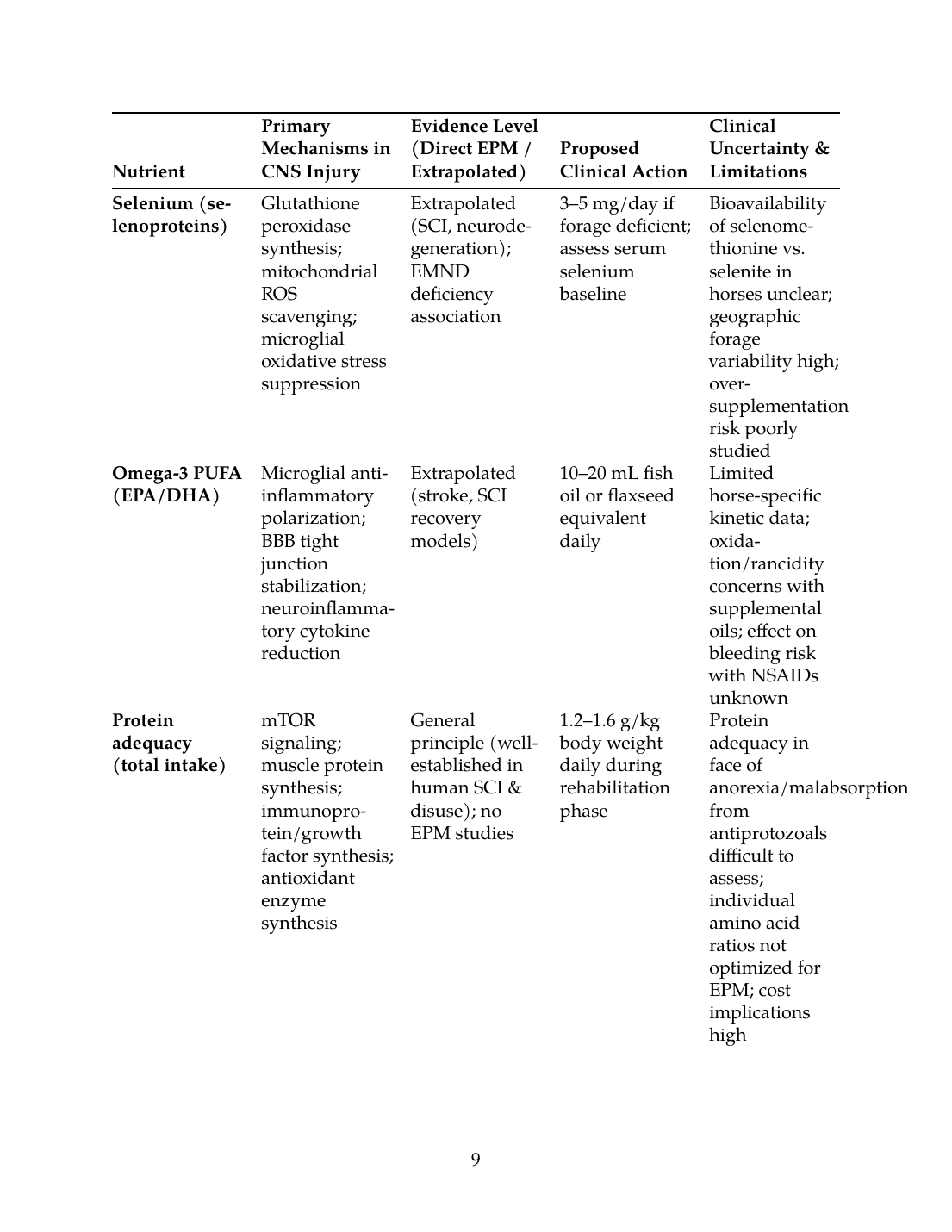
X. Proposed Mechanistic Table: Nutrients, Mechanisms, Evidence Level, and Clinical Uncertainty
| Nutrient | Primary Mechanisms in CNS Injury | Evidence Level (Direct EPM / Extrapolated) | Proposed Clinical Action | Clinical Uncertainty & Limitations |
|---|---|---|---|---|
| Vitamin E (α-tocopherol) | Lipid peroxidation inhibition; microglial membrane protection; NF-κB suppression | Extrapolated (SCI, TBI, EMND association) | 1,000–2,000 IU/day during acute phase & recovery | Optimal dose in horses unknown; fat-soluble storage may delay depletion; interaction with other antioxidants unclear |
| Selenium (selenoproteins) | Glutathione peroxidase synthesis; mitochondrial ROS scavenging; microglial oxidative stress suppression | Extrapolated (SCI, neurodegeneration); EMND deficiency association | 3–5 mg/day if forage deficient; assess serum selenium baseline | Bioavailability of selenomethionine vs. selenite in horses unclear; geographic forage variability high; over-supplementation risk poorly studied |
| Omega-3 PUFA (EPA/DHA) | Microglial anti-inflammatory polarization; BBB tight junction stabilization; neuroinflammatory cytokine reduction | Extrapolated (stroke, SCI recovery models) | 10–20 mL fish oil or flaxseed equivalent daily | Limited horse-specific kinetic data; oxidation/rancidity concerns with supplemental oils; effect on bleeding risk with NSAIDs unknown |
| Protein adequacy (total intake) | mTOR signaling; muscle protein synthesis; immunoprotein/growth factor synthesis; antioxidant enzyme synthesis | General principle (well-established in human SCI & disuse); no EPM studies | 1.2–1.6 g/kg body weight daily during rehabilitation phase | Protein adequacy in face of anorexia/malabsorption from antiprotozoals difficult to assess; individual amino acid ratios not optimized for EPM; cost implications high |
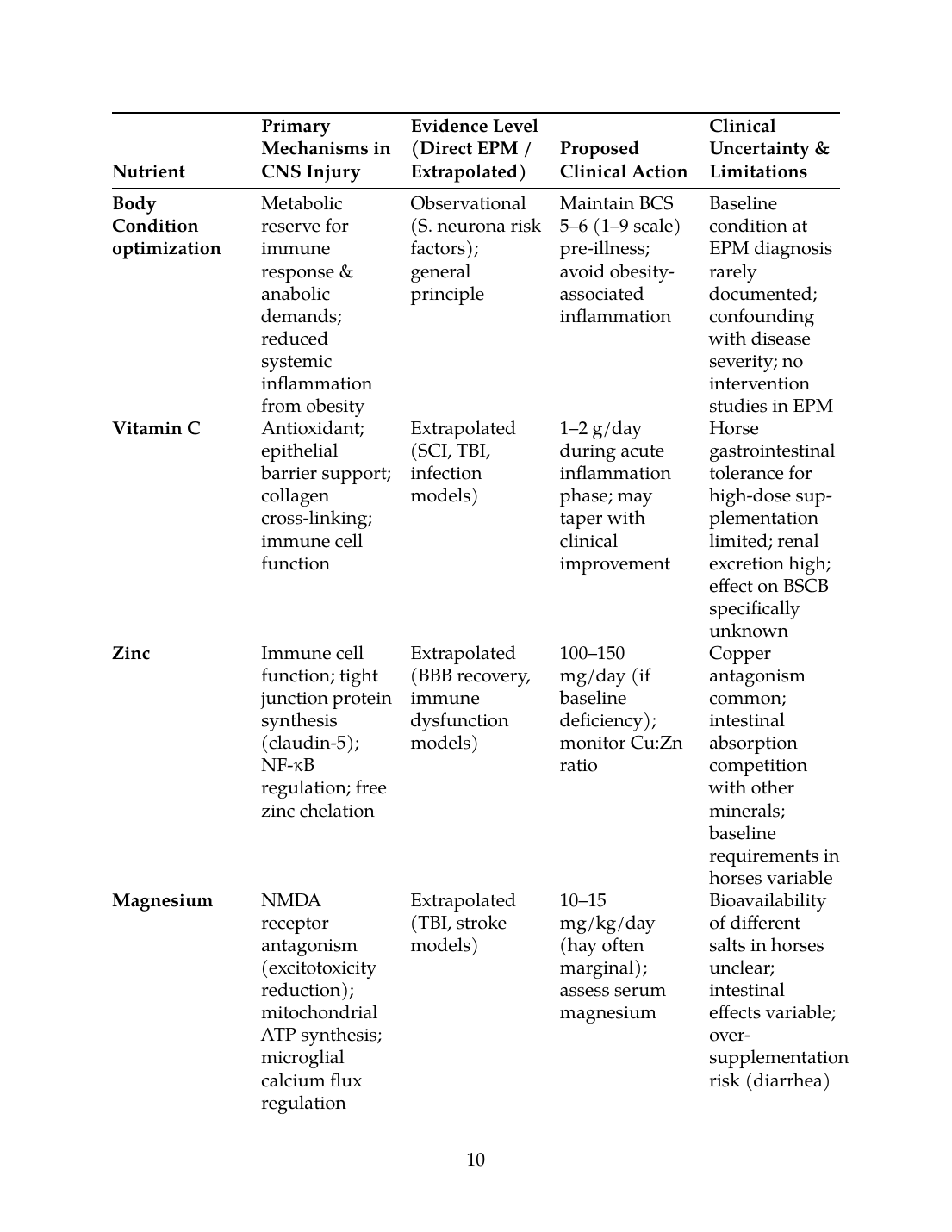
| Body Condition optimization | Metabolic reserve for immune response & anabolic demands; reduced systemic inflammation from obesity | Observational (S. neurona risk factors); general principle | Maintain BCS 5–6 (1–9 scale) pre-illness; avoid obesity-associated inflammation | Baseline condition at EPM diagnosis rarely documented; confounding with disease severity; no intervention studies in EPM |
| Vitamin C | Antioxidant; epithelial barrier support; collagen cross-linking; immune cell function | Extrapolated (SCI, TBI, infection models) | 1–2 g/day during acute inflammation phase; may taper with clinical improvement | Horse gastrointestinal tolerance for high-dose supplementation limited; renal excretion high; effect on BSCB specifically unknown |
| Zinc | Immune cell function; tight junction protein synthesis (claudin-5); NF-κB regulation; free zinc chelation | Extrapolated (BBB recovery, immune dysfunction models) | 100–150 mg/day (if baseline deficiency); monitor Cu:Zn ratio | Copper antagonism common; intestinal absorption competition with other minerals; baseline requirements in horses variable |
| Magnesium | NMDA receptor antagonism (excitotoxicity reduction); mitochondrial ATP synthesis; microglial calcium flux regulation | Extrapolated (TBI, stroke models) | 10–15 mg/kg/day (hay often marginal); assess serum magnesium | Bioavailability of different salts in horses unclear; intestinal effects variable; over-supplementation risk (diarrhea) |
| B Vitamins (folate, B12, B6) | One-carbon metabolism; BDNF synthesis; myelin lipid synthesis; homocysteine metabolism; neurotransmitter synthesis | Extrapolated (stroke, SCI, neurodegeneration recovery); antiprotozoal drug interactions | 2–4 mg/day folate (if on sulfonamides); B12 500 μg/week; assess baseline status if prolonged treatment | Equine B vitamin requirements not precisely defined; bioavailability of different sources unknown; interaction with antifolate drugs complex; deficiency vs. excess risk both present |
| Vitamin D | Immune tolerance (Treg differentiation); BSCB tight junction protein expression; microglial pro-resolving cytokine synthesis | Extrapolated (CNS infection, autoimmune disease models) | Baseline assessment of 25-OH vitamin D; supplement if <20 ng/mL; target 30–50 ng/mL | Equine vitamin D photosynthesis from sun exposure variable by latitude/season; oral supplementation dose-response not established in horses; immune-suppressive effects at high doses |
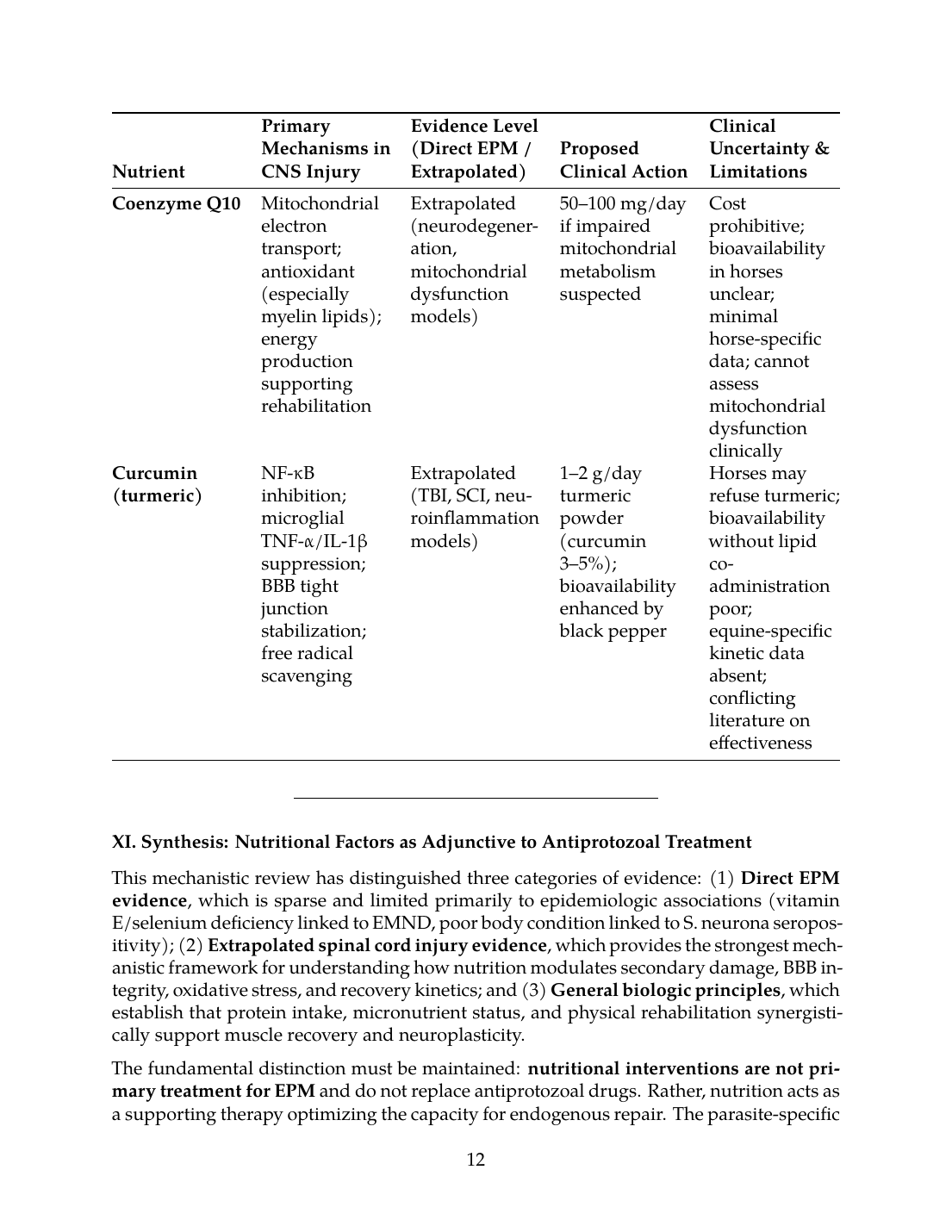
| Coenzyme Q10 | Mitochondrial electron transport; antioxidant (especially myelin lipids); energy production supporting rehabilitation | Extrapolated (neurodegeneration, mitochondrial dysfunction models) | 50–100 mg/day if impaired mitochondrial metabolism suspected | Cost prohibitive; bioavailability in horses unclear; minimal horse-specific data; cannot assess mitochondrial dysfunction clinically |
| Curcumin (turmeric) | NF-κB inhibition; microglial TNF-α/IL-1β suppression; BBB tight junction stabilization; free radical scavenging | Extrapolated (TBI, SCI, neuroinflammation models) | 1–2 g/day turmeric powder (curcumin 3–5%); bioavailability enhanced by black pepper | Horses may refuse turmeric; bioavailability without lipid co-administration poor; equine-specific kinetic data absent; conflicting literature on effectiveness |
XI. Synthesis: Nutritional Factors as Adjunctive to Antiprotozoal Treatment
This mechanistic review has distinguished three categories of evidence: (1) Direct EPM evidence, which is sparse and limited primarily to epidemiologic associations (vitamin E/selenium deficiency linked to EMND, poor body condition linked to S. neurona seropositivity); (2) Extrapolated spinal cord injury evidence, which provides the strongest mechanistic framework for understanding how nutrition modulates secondary damage, BBB integrity, oxidative stress, and recovery kinetics; and (3) General biologic principles, which establish that protein intake, micronutrient status, and physical rehabilitation synergistically support muscle recovery and neuroplasticity.
The fundamental distinction must be maintained: nutritional interventions are not primary treatment for EPM and do not replace antiprotozoal drugs. Rather, nutrition acts as a supporting therapy optimizing the capacity for endogenous repair. The parasite-specific damage caused by S. neurona or N. hughesi requires direct antimicrobial eradication; nutritional strategies cannot achieve this. However, once primary parasitic injury occurs, secondary inflammation, oxidative stress, BSCB disruption, and muscle catabolism become targets for nutritional modulation.
A. Evidence Hierarchy and Clinical Certainty
High certainty (principles applicable to EPM):
- Protein adequacy (≥1.2 g/kg daily) supports muscle synthesis during neurologic disability and recovery.
- Vitamin E and selenium deficiency is associated with equine neurologic disease and should be corrected.
- Antioxidant status influences oxidative stress burden; adequate vitamin E, selenium, and vitamin C are rational targets.
- Body condition optimization pre-illness provides metabolic reserve.
Moderate certainty (plausible but unproven in EPM):
- Specific vitamin E and selenium supplementation doses and timing optimize BSCB integrity and microglial resolution.
- Omega-3 PUFA supplementation reduces CNS inflammation and promotes neuroplasticity during EPM recovery.
- One-carbon metabolism nutrient sufficiency (folate, B12, B6, choline) supports BDNF synthesis and neuroplasticity.
- Combined antiprotozoal treatment and nutritional support (particularly antioxidants, protein) yield superior recovery outcomes compared to antiprotozoals alone.
Low certainty / speculative (insufficient evidence):
- Specific curcumin or other phytochemical doses produce measurable neuroprotection in horses.
- Magnesium or zinc supplementation specifically improves BSCB recovery in EPM beyond background sufficiency.
- Individually tailored micronutrient profiles based on serum baselines improve prognosis.
B. Practical Nutritional Framework for EPM-Affected Horses
A reasonable, evidence-informed approach to nutritional support during EPM recovery includes:
- Baseline assessment: Serum vitamin E, selenium, albumin, and body condition score at EPM diagnosis; assessment of forage quality (mineral composition if available).
- Acute phase (during antiprotozoal treatment, 0–8 weeks):
- Recovery/rehabilitation phase (8 weeks onward):
- Considerations during corticosteroid use (if employed):
C. Limitations and Areas of Clinical Uncertainty
Despite mechanistic plausibility, several critical limitations constrain application of this framework to individual EPM cases:
- Equine-specific kinetic and efficacy data are absent for most micronutrient supplementation in neurologic disease; doses and durations are extrapolated from human studies or rodent models with uncertain validity in horses.
- Forage mineral composition is highly geographically variable; without baseline testing, supplementation may be excessive or insufficient.
- Gastrointestinal dysfunction from antiprotozoals may impair nutrient absorption, reducing efficacy of oral supplementation.
- Individual horses exhibit variable recovery trajectories influenced by genetics, parasite burden, lesion severity, and host immune competence—factors not modifiable by nutrition alone and not objectively measurable during treatment.
- Cost and compliance limitations restrict realistic application of complex supplement regimens in many clinical settings.
- Interaction effects between multiple supplements and antiprotozoal/corticosteroid drugs are poorly characterized; risk of unintended consequences exists.
- The relative contribution of nutrition to recovery versus antiprotozoal efficacy and rehabilitation cannot be separated in clinical cases; empirical evidence that nutritional optimization improves prognosis specifically in EPM is absent.
---
XII. Future Research Directions
To advance evidence-based nutritional support in EPM, the following research priorities are proposed:
- Baseline micronutrient profiling in EPM-affected horses at diagnosis and correlation with severity, treatment response, and long-term outcome; prospective studies linking serum vitamin E, selenium, and other micronutrients to prognosis.
- Longitudinal studies of serum micronutrient dynamics during antiprotozoal treatment, monitoring depletion from drug interactions or increased demand; informing supplementation strategies.
- Mechanistic studies in horse models (or primary equine CNS cell cultures) examining effects of specific nutrient supplements on microglial cytokine production, BSCB tight junction integrity, or motor neuron survival following parasitic challenge.
- Controlled clinical trials randomizing EPM-affected horses to standardized antiprotozoal treatment with versus without structured nutritional support (high-quality protein, vitamin E/selenium, omega-3, one-carbon metabolism nutrients), measuring motor recovery (ataxia scores), rehabilitation timeline, and long-term functional outcome.
- Pharmacokinetic studies of vitamin E, selenium, and omega-3 supplementation in horses, establishing tissue bioavailability and CNS penetration.
- Investigation of forage quality optimization and pre-illness nutritional reserve building as primary prevention for EPM onset in high-risk populations.
---
Conclusion
Nutrition is mechanistically positioned to modulate secondary CNS damage, inflammation, oxidative stress, and recovery capacity in EPM, acting synergistically with antiprotozoal treatment and rehabilitation. The strongest evidence base derives from spinal cord injury models, where protein adequacy, antioxidant status (vitamin E, selenium, vitamin C), omega-3 PUFA supplementation, and one-carbon metabolism nutrients demonstrably influence inflammation, BSCB integrity, microglia phenotype, neuroplasticity, and functional recovery. In EPM, vitamin E and selenium deficiency is associated with related neurologic disease (EMND) and should be corrected; poor body condition is a risk factor for S. neurona seropositivity and should be optimized pre-illness.
However, direct evidence that specific nutritional interventions improve EPM prognosis is absent. The clinical relevance of findings from rodent SCI models and human neurologic disease to equine EPM recovery is unproven. Future horse-specific research, including mechanistic studies and randomized controlled trials, is essential to validate nutritional approaches and refine clinical recommendations. Until such evidence is generated, nutritional support in EPM should be viewed as a reasonable, low-risk adjunct to antiprotozoal treatment, informed by general principles of neurologic disease recovery and tailored to individual forage composition, baseline micronutrient status, and rehabilitation demands—but never as a replacement for antimicrobial therapy or owner-led rehabilitative exercise.
---
References
This comprehensive mechanistic review integrates current understanding of EPM pathogenesis, secondary CNS injury, and nutritional physiology to outline plausible—but largely unproven—nutritional interventions supporting EPM recovery. The distinction between direct EPM evidence, extrapolated SCI/neurologic disease mechanisms, and general biologic principles is maintained throughout, acknowledging substantial clinical uncertainty while providing a framework for evidence-informed practice pending rigorous equine-specific research.
References
References
Numbered in order of first citation, IEEE style. Each entry links to the original source via DOI or publisher URL.
- R. MacKay and D. K. Howe, “Equine Protozoal Myeloencephalitis.,” The Veterinary clinics of North America. Equine practice, Jul. 2022. doi: 10.1016/j.cveq.2022.05.003. View source
- C. J. Angwin, I. D. A. Rocha, S. Reed, J. K. Morrow, A. Graves, and D. K. Howe, “Analysis of IgG responses to Sarcocystis neurona in horses with equine protozoal myeloencephalitis (EPM) suggests a Th1-biased immune response.,” Veterinary Immunology and Immunopathology, Sep. 2025. doi: 10.1016/j.vetimm.2025.111009. View source
- S. Reed, S. Reed, M. Furr, D. Howe, A. L. Johnson, R. MacKay, J. Morrow, N. Pusterla, and S. Witonsky, “Equine Protozoal Myeloencephalitis: An Updated Consensus Statement with a Focus on Parasite Biology, Diagnosis, Treatment, and Prevention,” Journal of Veterinary Internal Medicine, Feb. 2016. doi: 10.1111/jvim.13834. View source
- S. Austin and S. Gray, “Equine protozoal myeloencephalitis—Presentation and progression,” Equine Veterinary Education, Oct. 2025. doi: 10.1111/eve.70016. View source
- A. Jegatheesan, M. Micciche, J. Ngo, P. Bradley, D. K. Howe, and S. Dangoudoubiyam, “Characterization of SnROP9, a rhoptry protein homologue of Sarcocystis neurona that is expressed in lifecycle stages lacking rhoptry organelles.,” International Journal of Parasitology, Feb. 2025. doi: 10.1016/j.ijpara.2025.02.001. View source
- L. Helber, B. Wagner, C. M. Leeth, T. LeRoith, T. E. Cecere, K. K. Lahmers, F. M. Andrews, A. N. Hay, S. R. Werre, A. Johnson, C. Clark, N. Pusterla, S. Reed, D. S. Lindsay, S. D. Taylor, K. Estell, M. Furr, R. J. Mackay, F. D. Piero, M. Carossino, K. Pandaleon, S. Weatherford, R. Ramirez-Barrios, K. Zimmerman, and S. Witonsky, “Persistence of Sarcocystis neurona and histopathology in horses with equine protozoal myeloencephalitis,” Frontiers in Veterinary Science, Apr. 2026. doi: 10.3389/fvets.2026.1787994. View source
- A. N. Hay, S. Witonsky, D. Lindsay, T. LeRoith, J. Zhu, L. Kasmark, and C. M. Leeth, “Sarcocystis neurona–Induced Myeloencephalitis Relapse Following Anticoccidial Treatment,” Journal of Parasitology, Apr. 2019. doi: 10.1645/18-193. View source
- M. Amirdosara, M. Hajiesmaeili, S. Hosseininasab, and M. Zangi, “Role of Inflammation in Secondary Injury Progression after Traumatic Brain Injury and Spinal Cord Injury,” Archives of Anesthesia and Critical Care, Feb. 2026. doi: 10.18502/aacc.v12i2.20957. View source
- R. H. D. Santos and M. C. O. Costa, “Mechanisms of Blood–Brain Barrier Disruption and Subsequent Cellular Injury in Animal Models of Neurological Disorders,” Pathophysiology of Cell Injury Journal, Feb. 2024. doi: 10.18081/2378-5225/14.58. View source
- Z. Chong, M. Zhou, Y. Chen, X. Xu, S. Chen, J. Rao, C. Wang, and R. Liang, “Ganoderma lucidum low molecular weight polysaccharide promotes the repair of spinal cord injury through anti-inflammatory and antioxidant.,” International Journal of Biological Macromolecules, Aug. 2025. doi: 10.1016/j.ijbiomac.2025.146958. View source
- K. Shen, X. Li, H. Wang, H. Zhang, Z. Yuan, X. Zheng, L. He, and T. Chen, “Seleno‐Amino Acid Nanotherapeutics Upregulates Selenoproteins to Inhibit Oxidation and Neuroinflammation for Spinal Cord Injury Recovery,” Advanced Functional Materials, Mar. 2026. doi: 10.1002/adfm.74986. View source
- Y. Xing, Y. Xiao, M. Zhao, J. Zhou, K. Zhao, and C. Xiao, “The role of oxidative stress in spinal cord ischemia reperfusion injury: mechanisms and therapeutic implications,” Frontiers in Cellular Neuroscience, Jun. 2025. doi: 10.3389/fncel.2025.1590493. View source
- H. Zhang, W. Wang, X. Hu, Z. Wang, J. Lou, P. Cui, X. Zhao, Y. Wang, X. Chen, and S. Lu, “Heterophyllin B enhances transcription factor EB-mediated autophagy and alleviates pyroptosis and oxidative stress after spinal cord injury,” International Journal on Biological Sciences, Oct. 2024. doi: 10.7150/ijbs.97669. View source
- X. Yang, W. Li, M. Ding, K. Liu, Z. Qi, and Y. Zhao, “Contribution of zinc accumulation to ischemic brain injury and its mechanisms about oxidative stress, inflammation and autophagy: an update.,” Metallomics, Feb. 2024. doi: 10.1093/mtomcs/mfae012. View source
- M. D. Morsy, O. Mostafa, and W. N. Hassan, “A potential protective effect of α-tocopherol on vascular complication in spinal cord reperfusion injury in rats,” Journal of Biomedical Sciences, Jul. 2010. doi: 10.1186/1423-0127-17-55. View source
- P. Liu, X. Liu, Z. Wu, K. Shen, Z. Li, X. Li, Q. Wu, L. Chan, Z. Zhang, Y. Wu, L. Liu, T. Chen, and Y. Qin, “Size effect-based improved antioxidant activity of selenium nanoparticles regulating Anti-PI3K-mTOR and Ras-MEK pathways for treating spinal cord injury to avoid hormone shock-induced immunosuppression,” Journal of Nanobiotechnology, Jan. 2025. doi: 10.1186/s12951-024-03054-7. View source
- B. Ll, B. Hultgren, A. Craig, L. H. Appell, L. Ed, D. Mattson, and D. Duffield, “Clinical, viral, and genetic evaluation of equine degenerative myeloencephalopathy in a family of Appaloosas.,” Journal of the American Veterinary Medical Association, Mar. 1991. doi: 10.2460/javma.1991.198.06.1005. View source
- M. Nakazaki, T. Yokoyama, K. Lankford, R. Hirota, J. Kocsis, and O. Honmou, “Mesenchymal Stem Cells and Their Extracellular Vesicles: Therapeutic Mechanisms for Blood–Spinal Cord Barrier Repair Following Spinal Cord Injury,” International Journal of Molecular Sciences, Dec. 2024. doi: 10.3390/ijms252413460. View source
- Y. Ma, Y. Liu, D. Pan, J. Zhang, Z. Liang, Y. Wang, X. Hu, Z. Wang, and T. Ding, “Photobiomodulation repairs the blood-spinal cord barrier in a mouse model of spinal cord injury.,” Neural Regeneration Research, Mar. 2025. doi: 10.4103/NRR.NRR-D-24-01098. View source
- P. Gao, J. Yi, W. Chen, J. Gu, S. Miao, X. Wang, Y. Huang, T. Jiang, Q. Li, W. Zhou, S. Zhao, M. Wu, G. Yin, and J. Chen, “Pericyte-derived exosomal miR-210 improves mitochondrial function and inhibits lipid peroxidation in vascular endothelial cells after traumatic spinal cord injury by activating JAK1/STAT3 signaling pathway,” Journal of Nanobiotechnology, Nov. 2023. doi: 10.1186/s12951-023-02110-y. View source
- L. Liu, C. Yang, B. P. Lavayen, R. J. Tishko, J. Larochelle, and E. Candelario-Jalil, “Targeted BRD4 protein degradation by dBET1 ameliorates acute ischemic brain injury and improves functional outcomes associated with reduced neuroinflammation and oxidative stress and preservation of blood–brain barrier integrity,” Journal of Neuroinflammation, Jun. 2022. doi: 10.1186/s12974-022-02533-8. View source
- M. ScholarWorks, E. Zukowski, and E. Natale, “COVID-19 and Nutrition,” Current Developments in Nutrition, Jul. 2023. doi: 10.1016/j.cdnut.2023.100427. View source
- E. Fadila, A. R. Bamahry, and M. Said, “The Role of Micronutrients in Gastrointestinal Function and Protection in Stroke Patients: Literature Review,” Journal La Medihealtico, Dec. 2025. doi: 10.37899/journallamedihealtico.v6i6.2808. View source
- M. Hood, B. Darimont, M. Missinato, M. Diaz, K. Lemoine, Y. Shi, D. Arias, C. D. Miller, A. Bhattacharya, M. Jordan, G. Erdogan, M. Cochran, H. W. Kwon, M. J. Moon, Y. Chen, A. J. Geall, V. Doppalapudi, R. S. Burke, A. A. Levin, H. Huang, and B. Malecova, “Antibody‐Oligonucleotide Conjugates Reduce Trim63 Expression in Skeletal Muscle and Prevent Muscle Atrophy,” JCSM Communications, Jan. 2026. doi: 10.1002/rco2.70035. View source
- M. Dirks, B. Wall, and L. V. van Loon, “Interventional strategies to combat muscle disuse atrophy in humans: focus on neuromuscular electrical stimulation and dietary protein.,” Journal of applied physiology, Sep. 2018. doi: 10.1152/japplphysiol.00985.2016. View source
- S. P. Kilroe, Z. V. Ruff, H. Kalenta, V. Bugay, E. J. Arentson-Lantz, A. J. Murton, V. G. Rontoyanni, D. Abdelrahman, E. Volpi, D. Paddon-Jones, and B. B. Rasmussen, “Integrated Muscle Protein Synthesis During Disuse and Rehabilitation in Late-Midlife Adults.,” American Journal of Physiology. Regulatory Integrative and Comparative Physiology, Sep. 2025. doi: 10.1152/ajpregu.00072.2025. View source
- T. Kim, K. Park, and S. Choung, “Codonopsis lanceolata and its active component Tangshenoside I ameliorate skeletal muscle atrophy via regulating the PI3K/Akt and SIRT1/PGC-1α pathways.,” Phytomedicine, Mar. 2022. doi: 10.1016/j.phymed.2022.154058. View source
- H. Magne, I. Savary-Auzeloux, C. Migné, M. Peyron, L. Combaret, D. Rémond, and D. Dardevet, “Unilateral Hindlimb Casting Induced a Delayed Generalized Muscle Atrophy during Rehabilitation that Is Prevented by a Whey or a High Protein Diet but Not a Free Leucine-Enriched Diet,” PLoS ONE, Aug. 2013. doi: 10.1371/journal.pone.0070130. View source
- M. Lees, C. M. Prado, P. Wischmeyer, and S. M. Phillips, “Skeletal Muscle: A Critical Organ for Survival and Recovery in Critical Illness.,” Critical care clinics, Oct. 2024. doi: 10.1016/j.ccc.2024.08.011. View source
- E. E. Howard, S. Pasiakos, M. A. Fussell, and N. Rodriguez, “Skeletal Muscle Disuse Atrophy and the Rehabilitative Role of Protein in Recovery from Musculoskeletal Injury.,” Advances in Nutrition, Mar. 2020. doi: 10.1093/advances/nmaa015. View source
- H. B. Llano, R. Soares, L. Y. Acevedo-Gutiérrez, J. Rodas, G. Polo, W. Borges-Silva, R. F. Jesus, and L. Gondim, “Seroepidemiology of Sarcocystis neurona and Neospora spp. in Horses, Donkeys, and Mules from Colombia.,” Acta Tropica, May 2021. doi: 10.1016/j.actatropica.2021.105970. View source
- P. James, Z. Ali, A. Armitage, A. Bonell, C. Cerami, H. Drakesmith, M. Jobe, K. Jones, Z. Liew, S. Moore, F. Morales-Berstein, H. M. Nabwera, B. Nadjm, S. Pasricha, P. Scheelbeek, M. Silver, M. R. Teh, and A. Prentice, “The Role of Nutrition in COVID-19 Susceptibility and Severity of Disease: A Systematic Review,” Journal of NutriLife, May 2021. doi: 10.1093/jn/nxab059. View source
- R. Abbasciano, G. Olivieri, R. Chubsey, F. Gatta, N. Tyson, K. Easwarakumar, D. Fudulu, R. Marsico, M. Kofler, G. Elshafie, F. Lai, M. Loubani, S. Kendall, M. Zakkar, and G. J. Murphy, “Prophylactic corticosteroids for cardiopulmonary bypass in adult cardiac surgery.,” Cochrane Database of Systematic Reviews, Mar. 2024. doi: 10.1002/14651858.CD005566.pub4. View source
- E. V. D. Carvalho, G. Polido, R. H. Mendonça, and E. Zanoteli, “Access to rehabilitation for people with spinal muscular atrophy undergoing treatment with disease-modifying drugs,” Arquivos de Neuro-Psiquiatria, 2024. doi: 10.5327/cbn240062. View source
- K. NoorAlhudaA., M. Jewi, K. Jasim, and Mohammed, “Micronutrient-Driven Immunomodulation Against Streptococcus Pneumoniae: The Role of Vitamins and Minerals in Pulmonary Defense,” Ibom Medical Journal, Jan. 2026. doi: 10.61386/imj.v19i1.936. View source
- J. T. Emmerson, L. K. Murray, and N. M. Jadavji, “Impact of dietary supplementation of one-carbon metabolism on neural recovery,” Neural Regeneration Research, Jul. 2017. doi: 10.4103/1673-5374.211183. View source
- Y. Shen, C. Zhang, C. Dai, Y. Zhang, K. Wang, Z. Gao, X. Chen, X. Yang, H. Sun, X. Yao, L. Xu, and H. Liu, “Nutritional Strategies for Muscle Atrophy: Current Evidence and Underlying Mechanisms.,” Molecular Nutrition & Food Research, May 2024. doi: 10.1002/mnfr.202300347. View source
- V. López-Sebastiani, K. V. Quiroz-Cornejo, M. P. Arellano-Salazar, F. Monje-Bolivar, and V. Samillan, “Micronutrient balance and brain function: neuropsychological, metabolic, and clinical interactions,” Frontiers in Molecular Biosciences, Apr. 2026. doi: 10.3389/fmolb.2026.1748487. View source
This review is one of 34 in the Foxwatch research corpus. Methodology: PRISMA 2020-style systematic literature review against PubMed, CAB Abstracts, Scopus, Web of Science, and equine veterinary conference proceedings.